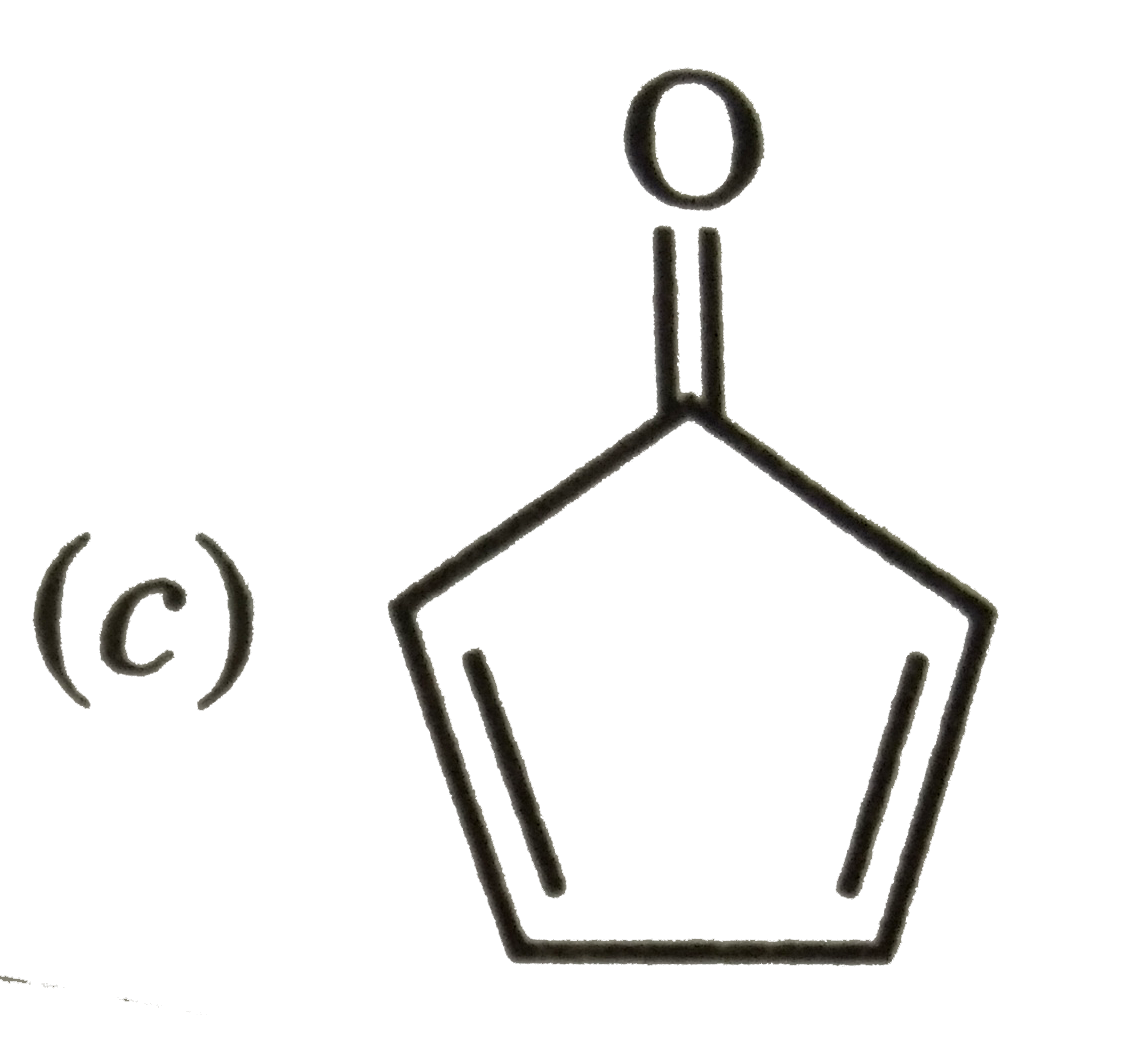Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which substance is known as .Calgon. ? |
|
Answer» `Na_6P_4O_18` |
|
| 2. |
What is the amount of Ca & Mg required by human body/bag? |
| Answer» SOLUTION :The HUMAN body requires 200-300 mg of Mg and 800 mg of CA per day. | |
| 3. |
Which of the following oxides dissolves in both hydrochloric acid and sodium hydroxide solution Boron and its compounds |
| Answer» ANSWER :D | |
| 4. |
What is the correct relationship between the two molecules shown below ? H-overset(Cl)overset(|)underset(H)underset(|)C-overset(H)overset(|)underset(H)underset(|)C-overset(H)overset(|)underset(H)underset(|)C-H and H-overset(H)overset(|)underset(H)underset(|)C-overset(Cl)overset(|)underset(H)underset(|)C-overset(H)overset(|)underset(H)underset(|)C-H |
|
Answer» There is no relationship |
|
| 5. |
What is troposphere? |
| Answer» Solution :The REGION which is extend UPTO the `~`10 KMS from sea level and in which the human beings and organisms can LIVE is called troposphere. | |
| 7. |
Write the structure of the tautomer of phenol and give its IUPAC name. which of the tautomers is more stable and why? |
Answer» SOLUTION :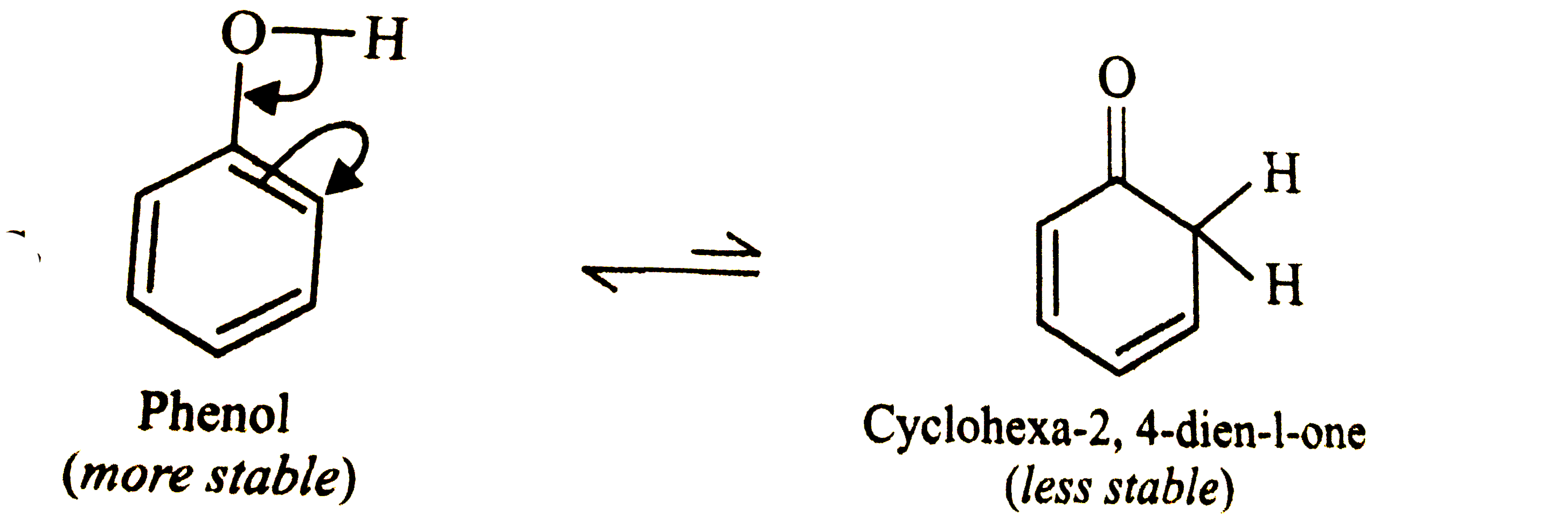 Phenol is more stable than cyclohexa-2, 4-dien-1-one because being an aromatic compound is stabilized by the resonance energy of the BENZENE RING `(151 kJ mol^(-1))` |
|
| 8. |
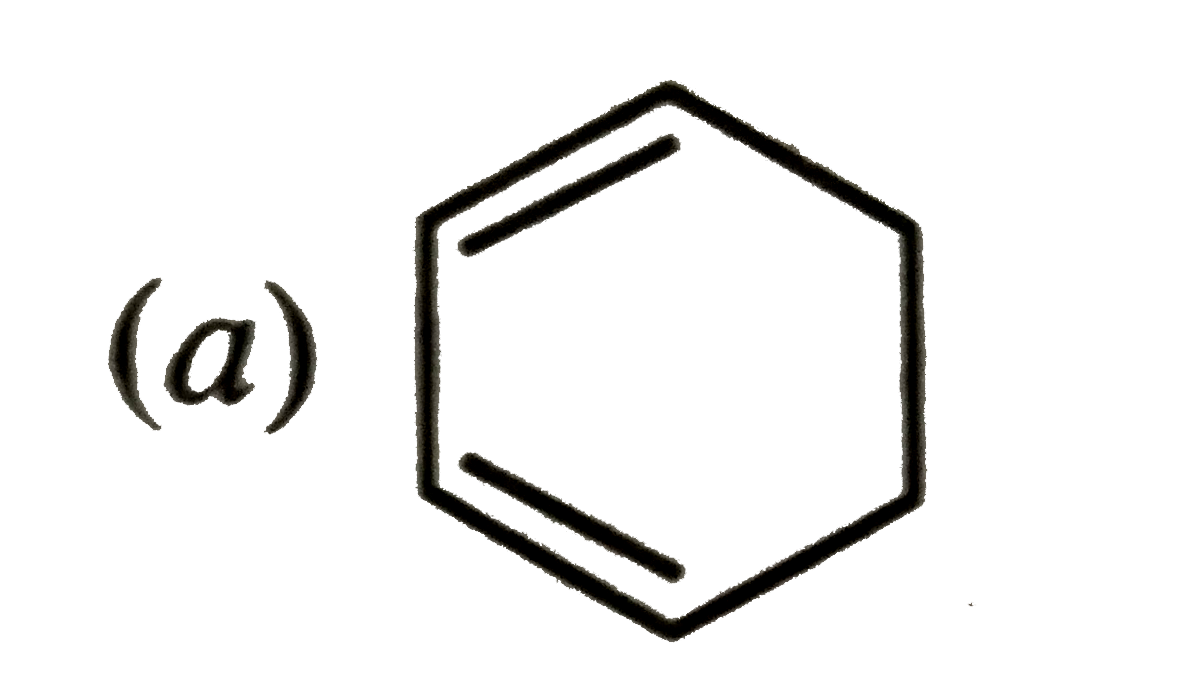
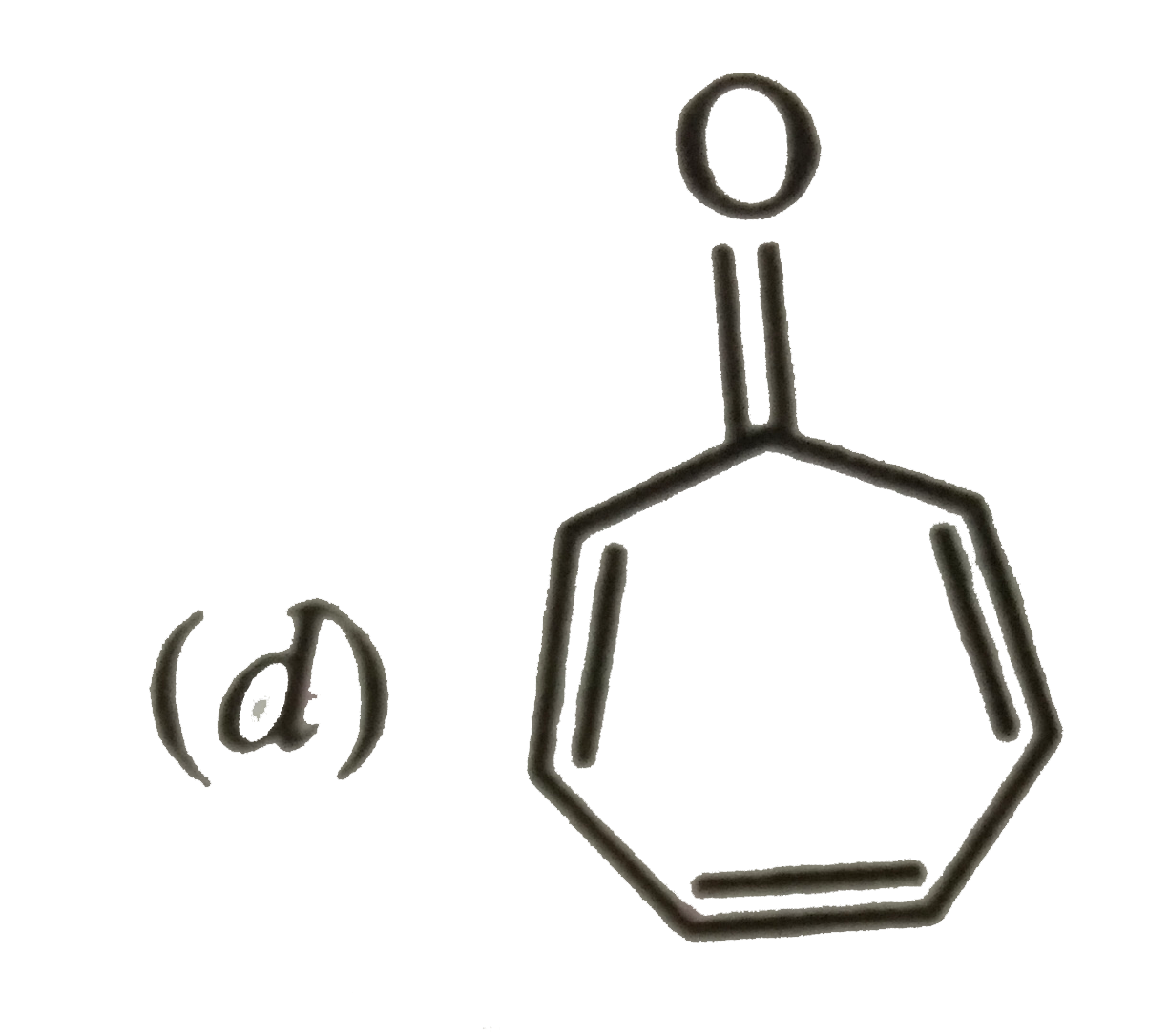
Which of the following molecules, in pure form, is unstable at room temperature ? |
|
Answer»
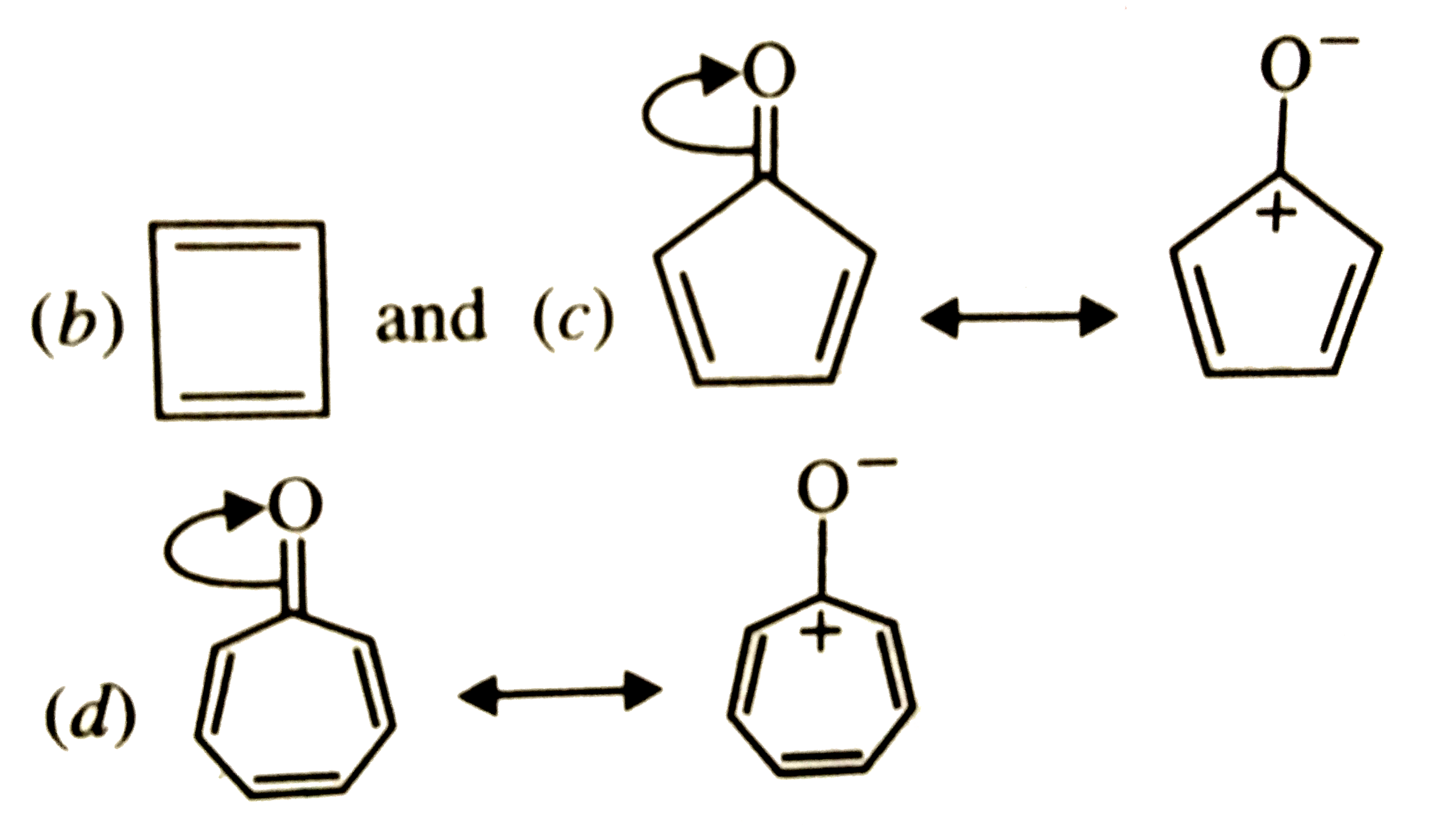 Both (b) and (c ) are PLANAR molecules and contain cyclic CLOUDS of `4pi`-electrons and HENCE are antiaromatic. Therefore these are unstable at room temperature. Compound under option (a) ALSO contions `4pi`-electrons but these are not delocalized over the entire ring. Therefore, it is non-aromatic. Compound under option (d) contains completely delocatlized `6pi`-electrons and is also planar. Therefore, it is aromatic. |
|
| 9. |
Write structures of different isomeric alkyl groups corresponding to the molecular formula C_(5)H_(11). Write IUPAC names of alcohols obtained by attachment of -OH groups at different carbons of the chain. |
Answer» SOLUTION :
|
|
| 10. |
Which pair is different for reaction with Fehling solution ? |
|
Answer» GLUCOSE, fructose |
|
| 11. |
Which group of element of periodic table has least ionization enthalpy ? |
|
Answer» I-A |
|
| 12. |
Which of the following molecules is not linear |
|
Answer» `C_2H_4` |
|
| 13. |
Write the IUPAC names of the compounds (i-iv) from their given structures (i) {:(overset(1)(C)H_(3)-overset(2)(C)H_(2)-overset(3)(C)H-overset(4)(C)H_(2)-overset(5)(C)H_(2)-overset(6)(C)H-overset(7)(C)H_(2)-overset(8)(C)H_(3)),("|""|"),(""OH""CH_(3)):} (ii)overset(6)(C)H_(3)-overset(5)(C)H_(2)-overset(O)overset("4||")(C)-overset(3)(C)H_(2) overset(2)(-)overset(O)overset(||)(C)-overset(1)(C)H_(3)(iii) overset(6)(C)H_(3)-overset(O)overset("5||")(C)-overset(4)(C)H_(2)-overset(3)(C)H_(2)-overset(2)(C)H_(2)-overset(1)(C)OOH (iv) Hoverset(6)(C)-=overset(5)(C)-overset(4)(C)H=overset(3)(C)H-overset(2)(C)H=overset(1)(C)H_(2) |
|
Answer» Solution :(i) Step 1. Here, the functional group is alcohol (OH). Hence, its secondary suffix is ol. Step 2. The longest carbon chain containing the functional group has EIGHT carbon atoms. THEREFORE, word root is oct. Further, since the longest carbon chain is saturated, therefore, its primary suffix is ane. Step 3. NUmber the carbon atoms of the chain from left to right. It gives lowest locant 3 to the functional group (OH) and locant 6 to the substituent methyl group (secondary prefix). Step 4. Since the secondary suffix 'ol' begins with a vowel 'o' therefore, terminal 'e' from the primary suffix is dropped. Therefore, the complete IUPAC name of the compound is : 6-methyl + octane-e + 3-ol = 6-methyloctane-3-ol. (ii) Step 1. Here, the functional group PRESENT is ketone `(gt C = O)`. Therefore, its secondary suffix is one Since there are two keto groups, therefore, the numerical prefix di is attached to the secondary suffix one. Hence the complete secondary suffix BECOMES 'dione'. Step 2. The longest carbon chain contains 6 carbons atoms, therefore, the word root is hex. Further, since the carbon chain is saturated, therefore, the priary suffix is ane. Step 3. Number the carbon atoms of the chain from right to left. It gives lowest locants 2 and 4 to the keto groups. Step 4. The complete IUPAC name is : hexane +2, 4-dione = hexane-2, 4-dione. Please note that the terminal 'e' from the primary suffix 'ane' has not be dropped here since the secondary suffic dione begins with a consonant 'd' and not with any vowel. (iii) Step 1. In this compound, two functional groups are present, i.e., a ketone and a carboxylic acid. Since carboxylic acid is the principal functional group, therefore, its secondary suffix is oic acid. Further, since keto group is the substituent functional group, therefore its prefix is oxo or keto. Step 2. The longest carbon chain contains atoms, therefore, its word root is hex. Further, since the chain is saturated, therefore, the primary suffix is ane. Step 3. Number the carbon chain from right to left since the principal functional group, i.e., `-COOH` is the chain terminating group, it must have the lowest locant 1. This gives the substituent functional group, i.e., oxo group locant 5. Step 4. Drop the terminal 'e' from the primary suffix 'ane' since the secondary suffix, 'oic acid' begins with a vowel. Therefore, the complete IUPAC name is : 5-oxo +hex + ane-e + oic acid = 5-oxohexanoic acid. iv Step 1. In this compound, there two C=C and one `C-=C` functional groups. The secondary suffixfor the two C=C groups is 'diene' while the secondary suffix for `C -= C` functional group is 'yne'. Step 2. The longest carbon chain contains six carbon atoms, therefore, the root word is hex. Further, since the carbon chain contains two double bonds and one triple bond, therefore, the complete secondary suffixes are diene and yne respectively. Step 3. Whenever there is a choice in numbering, double bond is given preference, therefore, locants for diene are 1 and 3, and for yne is 5. Step 4. The terminal 'e' from the suffix 'diene' is dropped because the first letter 'y' in the suffix 'yne' sounds like a vowel. Thus, the complete IUPAC name of the given compound is : hexa + 1, 3-diene+yne = hexa-1,3-dien-5-yne. Please note that here extra 'a' has been added to the root word hex since the primary suffix diene beigns with a consonant rather than a vowel. |
|
| 14. |
Which bicarbonate does not exist in solid state |
|
Answer» `LiHCO_(3)` |
|
| 15. |
The total number of valency electrons present in SO_(4)^(2-) ion |
|
Answer» 32 |
|
| 17. |
Which of the following statements regarding the above is correct ? |
|
Answer» C2 is more reactive then C1 towards phenyl hydrazine. |
|
| 18. |
Write important of organic chemistry |
|
Answer» Solution :ORGANIC compounds are vital for SUSTAINING life on earth. Organic compounds of body: (i) Genetic information bearing deoxyribonucleic acid (DNA) and (ii) Proteins that constitute essential compounds of our blood, MUSCLES and skin. Organic chemicals appear in materials like clothing, FUELS, polymers, DYES and mediciness. These are some of the important areas of application of these compounds |
|
| 19. |
Write product of the following reaction :- Fe_2O_3 + CO to |
|
Answer»
|
|
| 20. |
The vapour pressure of pure benzene(C_6H_6)at a given temperature is 640 mm Hg . 2.2 g of non -volatile solute is added to 40 g of benzene. The vapour pressure of the solution is 600mm Hg . Calcultaed the molar mass of the solute? |
|
Answer» <P> Solution :`P _(C _(6)H_(6))^(0) = 640mm Hg, W _(2) =2.2 g` (non volatile solute),` W _(1) = 40 g` (benzene)`P _("solution") = 600 mm Hg, M _(2)=` ? `(P ^(0) - P )/(P ^(0))= x _(2)` ` (640-600)/(640) = (n _(2) )/(n_(1) + n _(2)) [ because n _(1) GT gt n _(2) , n _(1) + n _(2) ~~n _(1) ]` `(40)/(640) = (n _(2))/( n _(1))` ` 0.0625 = (W_(2) xx M _(1))/( M _(2) xx W_(1))` `M _(2) = ( 2.2 xx 78)/( 0.0625 xx 40) = 68. 64 g mol ^(-1)` |
|
| 21. |
Which of the following species contains equal number of sigam and pi -bonds ? |
|
Answer» `(CN)_(2)` `CN_(2) (CN)_(2) : N-=C - underset(H)underset(|)overset(H)overset(|)C-C-=N (6sigma + 4pi)` 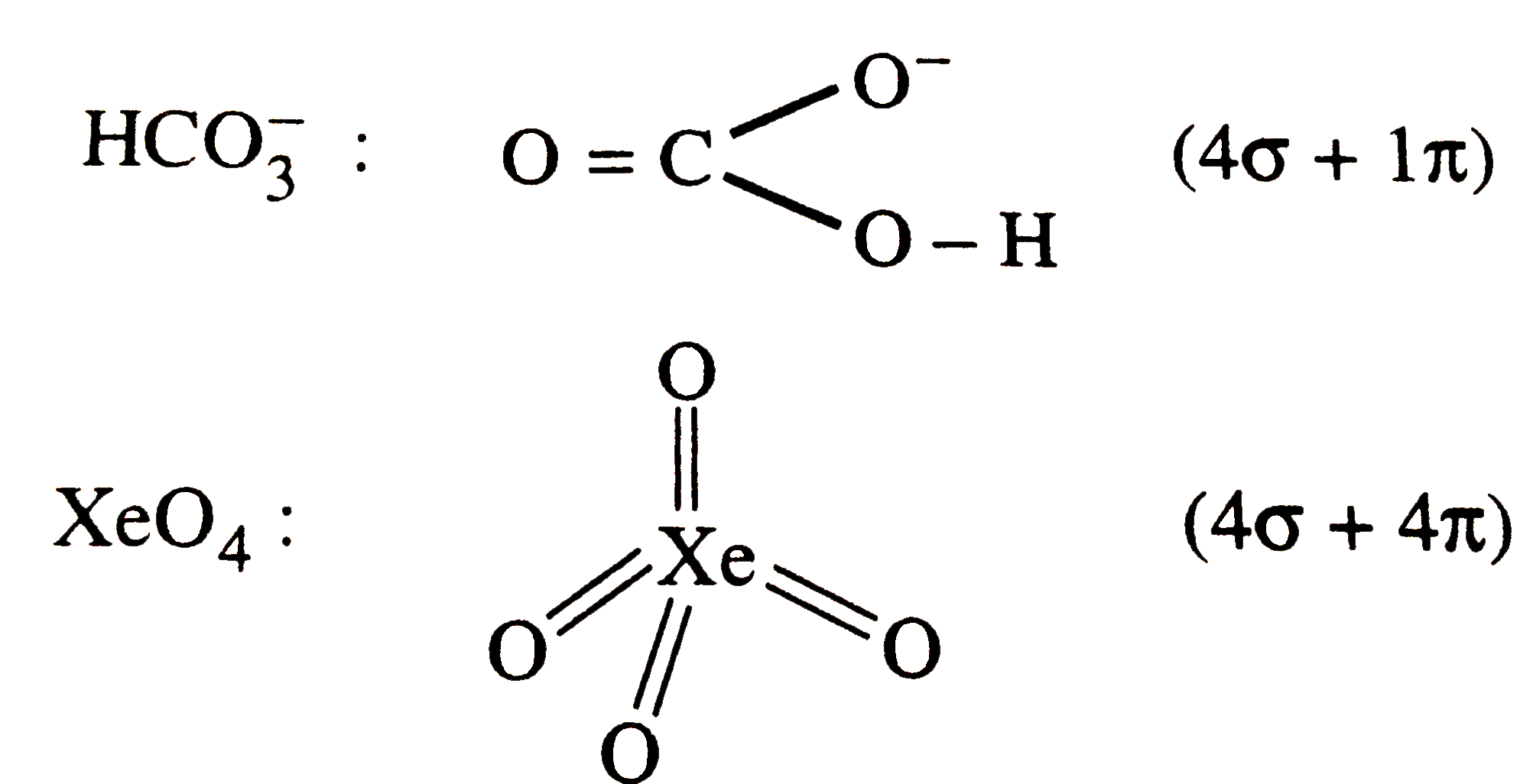
|
|
| 22. |
Which type of hybridization are shown by carbon atoms present in but-I-ene? |
|
Answer» `sp^(2)` |
|
| 23. |
What is the normality of 0.3 M H_(3)PO_(4) when it undergoes the reaction as ? H_(3)PO_(4) + 2OH^(-) to HPO_(3)^(2-) + 2H_(2)O |
|
Answer» 0.3 N |
|
| 24. |
WriteimportanceofPlanck'squantum theory. |
|
Answer» Solution :Blackbodyradiationcannot beexplainedby wave theoryof light .( Emitandabsorbwavefrequency and COLOUROF blackbodyradiationischangewithtemperature ) TheoryPlank suggestedthatatomsand moleculescouldemitand not isa continuousmannerPlankquantitiesnamequantumto thesmallestquantityofenergythatcan beemittedorabsorbed. Theenergy( E)or a QUANTUM of radiationisproportionalto itsfrequency( v ) and isexpressedby equation ( E `prop` v ) Important : WiththistheoryPlankwasable to explainthe DISTRIBUTION ofintensityin therediationfromblackbodyas afuntion offrequencyorwavelengthatdifferenttemperature . E= nhv(Eq. 2.11) wheren= 0,1,2 n=1 So E =HV =MINIMUM energy |
|
| 25. |
Using the standard electrode potential, find out the pair between which redox reactions is not possible ? E_(Fe^(+3)//Fe^(+2))^(Ө)=+0.77"volt",E_(I_(2)//I^(-))^(Ө)=+0.54"volt" E_(Cu^(+2)//Cu)^(Ө)=+0.34"volt",E_(Ag^(+)//Ag^(-))^(Ө)=+0.80"volt" |
|
Answer» `Fe^(+3)andI^(-)` 
|
|
| 26. |
The unit cell in a body centred cubic lattice is given in the figure. Each sphere has a radius ,r and the cube has a side ,a . What fraction of the total cube volume is empty ? |
|
Answer» `1-(8)/(3)PI(R^(3))/(a^(3))` |
|
| 27. |
Which of the following does not affect the degree of ionisation? |
|
Answer» Temperature |
|
| 28. |
What is CORRECT about surface tension and viscosity of liquids ? |
|
Answer» Both INCREASE the temperature |
|
| 30. |
When 3.0 mole of an ideal diatomic gas is heated and compressed simultaneously from 300K, 1.0 atm to 400K and 5.0atm, the change in entropy is (Use C_(P) = (7)/(2)R for the gas) |
|
Answer» `-20 JK^(-)` `= 3 xx (7)/(2) R xx "ln" (5)/(1) = -15` |
|
| 31. |
When two reactants A and B are mixed to give products C and D, the reaction quotient (Q) at the intiai stages of the reaction: |
|
Answer» zero |
|
| 32. |
Write formula for the following compound: Iron (III) Sulphate |
| Answer» SOLUTION :`Fe_2` (III) `(SO_4)_3` | |
| 33. |
Which of the following mixture is not a buffer solution |
|
Answer» `100 mL` of `0.5 N CH_(3)COOH + 100 mL ` of `0.05 N NaOH` |
|
| 34. |
Which of the following compound(s) give white precipitate with silver nitrate solution? |
|
Answer» `NaCI` |
|
| 35. |
The shape of AB_3E molecule (B = bond pair, E = lonepair) |
|
Answer» TETRAHEDRAL |
|
| 36. |
Write homo and heterolytic fission of C-Br bond in CH_(3)CH_(2)-Br |
Answer» SOLUTION :
|
|
| 37. |
What will be the number of monohalogen derivatives for |
|
Answer» |
|
| 38. |
Which are not correct ? |
|
Answer» `Ge(OH)_(2)` is amphoterica |
|
| 39. |
Two solution marked as A and B are seprated through semipermeable membrane as below . The phenomenon undergoing (##SUR_CHE_XI_V02_C09_E02_042_Q01.png" width="80%"> |
|
Answer» ` Na^(+) ` moves from solution A to solution B |
|
| 40. |
Which one of the following forms micelles in aqueous solution above certain concentration |
|
Answer» Urea |
|
| 41. |
Write the alkali metals in the increasing order of hydrogen energy. |
| Answer» SOLUTION :`Li^(+) GT Ma^(+) gt K^(+) gt Rb^(+) gt CS^(+)` | |
| 42. |
Which of the following property of water can be used to explain thespherical shape of rain droplets ? |
|
Answer» viscosity |
|
| 43. |
Which of the following statements are correct for above reaction . |
|
Answer» Reaction intermediate is carbocation |
|
| 44. |
Which of the following is not formed by the reaction of Cl_(2) on CH_(4) in sunlight ? |
|
Answer» `CHCl_(3)` `underset("Methyl free radical")(CH_(3)+CH_(3)) rarr underset("Ethane")(CH_(3)-CH_(3))` |
|
| 45. |
What is caused by UV rays? |
|
Answer» SKIN CANCER |
|
| 46. |
Which of the following compounds will not underge Friedal-craft's reaction easily ? |
|
Answer» NITRO benzene |
|
| 47. |
Which of the following is meta directing group? |
| Answer» Solution :`-COOH ` is META directing group | |
| 48. |
Which of the following alkynes show acidic charater?(i) Propyne (ii) But-1-yne (iii) But-2-yne (iv) Pent-2-yne |
| Answer» SOLUTION :(i) and (II) | |
| 49. |
The space or three-dimensional region round the nucleus where there is maximum probability of finding an electron of specific energy is called an____ |
| Answer» SOLUTION :ORBITAL | |
| 50. |
The shape of AB_(3)E type molecule is |
|
Answer» Pyramidal |
|