Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
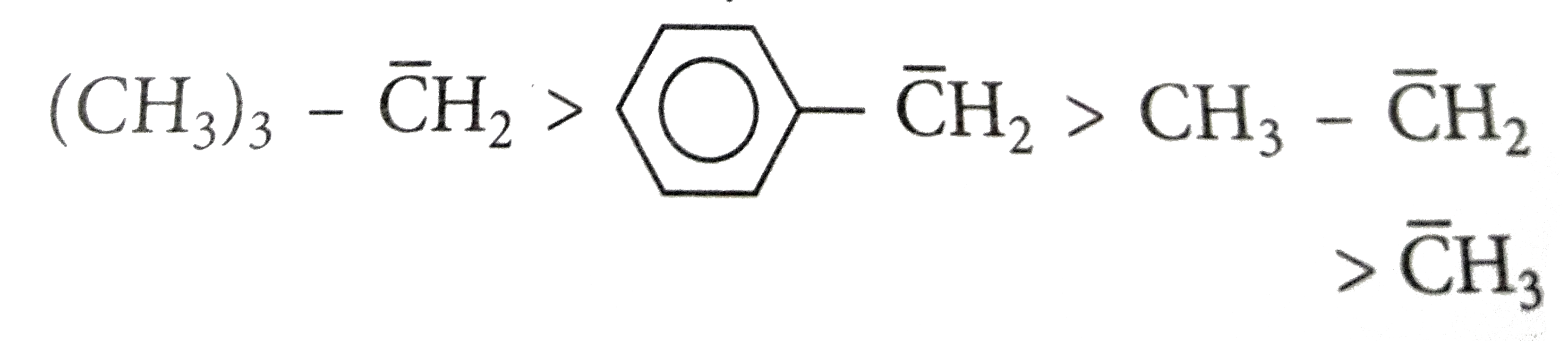
Which of the following orders correctly depicts the decreasing order of stability of carbanion? |
|
Answer»
|
|
| 2. |
What will be the percentage of dissociation in 1.0 M CH_(3)CO OH at equilibrium having dissociation constantof 1.8xx10^(-5)? |
|
Answer» % dissociation `= (4.24xx10^(-3))xx100=0.424` |
|
| 3. |
Which of the following is an extensive property? |
|
Answer» MOLAR volume |
|
| 4. |
White fumes appear around the bottle of anhydrous aluminium chloride. What is the reason ? |
| Answer» Solution :Anhydrous `AICI_(3)` is hydrolysed partially with atmospheric moisture to LIBERATE HYDROGEN CHLORIDE gas. Moist `HCl` APPEARS white in COLOUR. | |
| 5. |
What type of organic compounds cannot be Kjeldahlised ? |
| Answer» Solution :COMPOUNDS CONTAINING nitrogen atom in the ring and those compounds in which nitrogen is directly LINK either to OXYGEN or to another nitrogen atom such as in nitro `(-NO_(2))` and AZO (-N = N-) compounds. | |
| 6. |
Which of the following statements about petroleum is correct? |
|
Answer» PETROLEUM is a dark coloured OILY liquid with a characteristic smell |
|
| 7. |
What happens when HI reacts with teri-butyl alcohol ? |
| Answer» Solution :`CH_(3) underset("Tert, butyl alcohol")(-overset(CH_(3))overset(|)underset(CH_(3))underset(|)(C)-OH)+HI overset(KI)underset(H_(3) PO_(4))(to)CH_(3) underset("Tert. butyl IODIDE") (-overset(CH_(3))overset(|)underset(CH_(3))underset(|)(C)-I) + H_(2)O` | |
| 8. |
What is the work of salt bridge in Denial cell ? |
|
Answer» SOLUTION :Work : (i) CONNECTING TWO container. (II) MAINTAIN neutrality of electrolyte. |
|
| 9. |
Which of the following statements is not true for Ce^(3+) and Yb^(3+)? |
|
Answer» Both ABSORB in UV region |
|
| 10. |
Which of the following acid has the same molecular weight and equivalent weight |
|
Answer» `H_(3)PO_(2)` |
|
| 11. |
Which of the following variant of hydrogen spectrum results from nuclear spin interaction with that of electron? |
|
Answer» Fine spectrum |
|
| 12. |
Which one of the following law is used to explain gas-solution equilibrium processes? |
|
Answer» BOYLE's law |
|
| 14. |
Which of the following hydroxides is the strongest base. |
|
Answer» `Ba(OH)_(2)` |
|
| 16. |
Which elements of group 13, except B, can form tetrahedral and octahedral complex in aqueous medium ? |
|
Answer» Solution :TETRAHEDRA species : `[m(OH)_4]^(-)` OCTAHEDRAL species : `[m(H_2O)_4]^(+3)` |
|
| 17. |
Which of the following is right for CO_2 ? |
|
Answer» It is a LEWIS ACID. |
|
| 18. |
Which of the following molecule possess least resonance stability ? |
|
Answer»
|
|
| 19. |
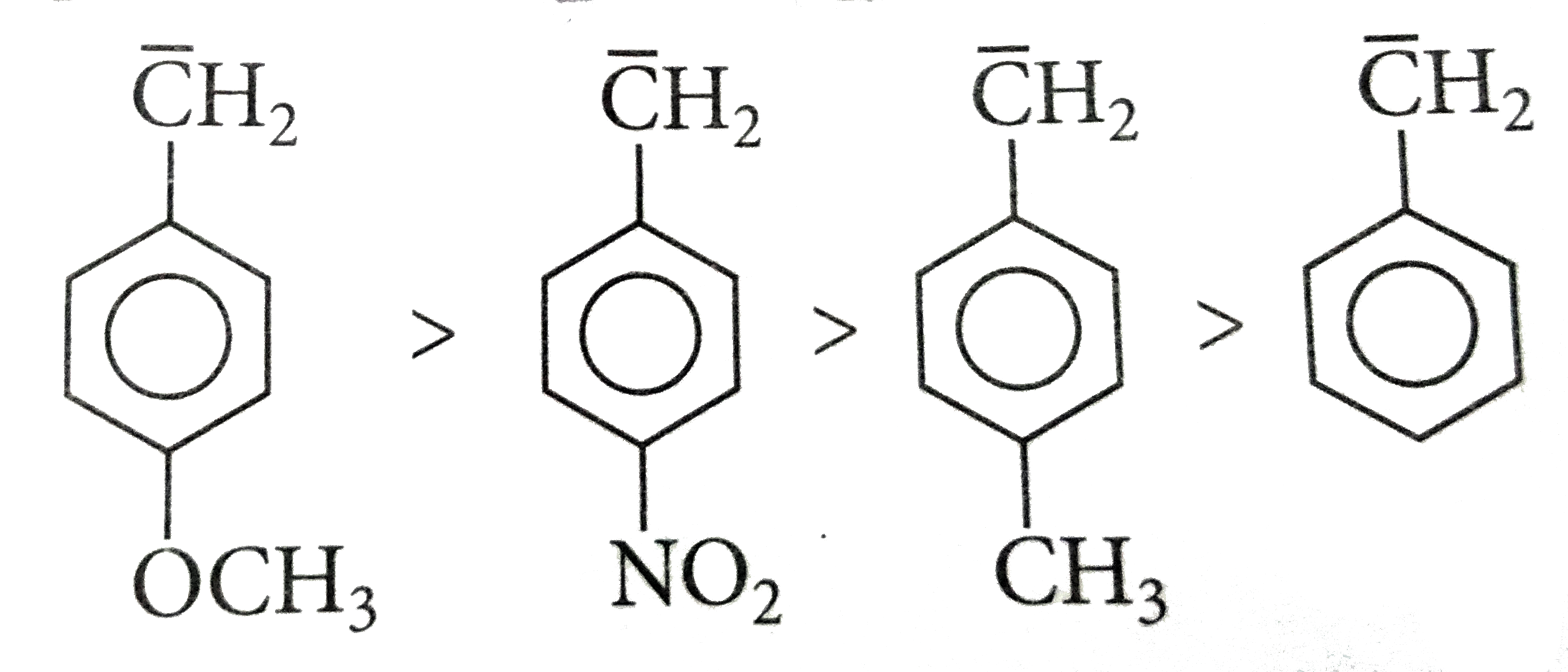
Which of the following statements is right regarding CH_(3)-, CH_(3)CH_(2)-, (CH_(3))_(2)CH-, (CH_(3))_(3)C- |
|
Answer» Increasing HYPER conjugation left to right `-CH_4 to " no " alpha - H`  `CH_3 - UNDERSET(CH_3)underset(|)overset(CH_3)overset(|)C- to ` Has 9 `-alpha - H` atoms |
|
| 20. |
Try all possible approaches to justify that the following reactions re redox reaction (a)CuO(s)+H_(2)rarrCu(s)+H_(2)O(g) (b)Fe_(2)O_(3)(s)+3CO(g)rarr2Fe(s)+3CO_(2)(g) (c )4BCI_(3)(g)+3 LiAIH(s)rarr2B_(2)H_(6)(g)+3LiCI(s)+3AICI(s) (d)2K(s)+F_(2)(g)rarr2K^(+)F^(-)(s) (e )4NH_(33)(g)+5O_(2)(g)rarr4NO(g)+6H_(2)O(g) |
|
Answer» Solution :(a) `CuO(s)+H_(2)(g)rarrCu(s)+H_(2)O(g)` here O is removed form CuO therefore it is reduced to Cu while o is added to `H_(2)`form `H_(2)` therefore it is oxidised furhter O.N of Cu DECREASES form +2 in CuO to 0 in Cu that of H increases from 0 in `H_(2)` to +1 `H_(2)O` therefore CuO reduceed to Cu but `H_(2)` is oixdised to `H_(2)O` thus this is REDOX reactoin (b) `Fe_(2)O_(3)(s)+3cO(g)rarr2fe(s)+3 CO_(2)(g)` here O.N of Fe decreases form +3 is `Fe_(2)O_(3)` to 0 in Fe while that of c increases form +2 in CO to + in `CO_(2)` further oxygen is aremoved from `Fe_(2)O_(3)`and added to CI therefore `Fe_(3)O_(3)` is reduced while CO is oxidised thus this is a redox reaction ltbnrgt (c ) `4B CI_(3)(g)+Li AI H_(4)(s)rarr 2B_(2)H_(6)(g)+3 Li CI (s) + 3AICI_(3)` here O.N of B decreases form + in `CrCI_(3)` to -3 Li `CI(s) +3 AICI_(3)(s)` here O.N of b decrease form +3 in `BrCI_(3)` to -3 in `B_(2)H_(6)` while that of H increases from -1 Li `AIH_(4)`to +1 in `B_(2)H_(6)` (d) `2K(s) +F_(2)rarr2F^(+)F^(-)(s)` here each k atom has lost one letron to form `K(+)` while `F_(2)` has gained two electrons to form two ions therefore k is oixdised while `F_(2)` is reduced thus it is a redox rection (e ) `4HN_(3)(g)+5O_(2)(g)rarr4NO(g)+6H_(2)O(g)` here O.N of N increases from -3 is `NH_(3)` ot +32 is NO while that O decreaess from 0 is `O_(2)` to -2 in NO or `H^(@)O` therefore `NH_(3)` is oxidised while `O_(2)` is reduced further H been removed has been oxidised while `O_(2)` is reduced thus this isa redox reaction |
|
| 21. |
Which of the following pairs of structures donot constitute resonating structures ? |
| Answer» SOLUTION :They DONOT CONTRIBUTE TOWARDS RESONANCE. | |
| 22. |
The weakest Lewis acid strength of BF_3 among boron halides can be explained on the basis of |
|
Answer» the most electronegative NATURE of F |
|
| 23. |
Write the functional group of (i) thiocyanate (in) isothiocyanate (iii) thiols (iv) thioether |
|
Answer» Solution :(i) Thio CYANATE -SCN (II) Isothiocyanate -NCS (iii) thiols -SH (iv) THIOETHERS -S- |
|
| 24. |
What is vicinal dihalides ? Give examples. |
|
Answer» Solution :VICINAL dihalide means attachment of two halogen ATOS to ADJACENT (neihgbour) CARBON ATOMS of compound. e.g.. `CH_(2)Br CHBrCH_(3), CH_(3) CHBrCH_(2)Br`….. |
|
| 25. |
Which of the following is water repellent ? |
|
Answer» Silicone |
|
| 26. |
Which of the following substances can be used for drying gases ? |
|
Answer» CALCIUM carbonate |
|
| 27. |
Write the structure of the following compounds (i) 1-Bromo-4-ethyl cyclohexane (ii) 1,4-Dichlorobut-2-ene (iii) 2-Chloro-3-methyl pentane. |
Answer» SOLUTION :(i)  (ii) `H_(2)underset(Cl)underset(|)(C)-CH=CH-underset(Cl)underset(|)(C)H_(2)` (iii) `H_(3)C-CH_(2)-underset(CH_(3))underset(|)(C)H-underset(Cl)underset(|)(C)H-CH_(3)`. |
|
| 28. |
Which of the following molecule does not exist due to its zero bond order? |
|
Answer» `H_(2)^(-)` |
|
| 29. |
Which one of the following is not a covalent hydride? |
| Answer» Solution :`[Fe(CN)_(6)]^(4-)` | |
| 30. |
Which of the following statement are correct I) Now -a days syngas is produced from sewage , saedust , scrap wood ,news paper etc. (II) The processs of proucing syngas from coal is called coal gassification .III) The producation of dihdration can be increased by the presence of iron chromate catalyst. IV) 77% of the industrial s=dihyrogen is propduced from perto chemicals |
|
Answer» I,II |
|
| 31. |
Which is the following is the strongest acid ? |
|
Answer» 3,5-dinitrophenol |
|
| 32. |
What are the main features of quantum mechanical model of an atom |
|
Answer» Solution :(1) The energy of electrons in an ATOMS is quantised. (2)The EXISTENCE of quantized electrons energy levels is a direct results of the like properties of electrons.The solutions of schrodinger WAVE equation gives the allowed energy levels(orbitals). (3)According to Heisenberg.s uncertainty principle,the exact position and momentum of an electron cannot be determined with absolute accuracy.As a consequence,quantum mechanics introduced the concept of orbital.Orbital is a three dimensionalspace in which the probality of finding the elctron is maximum. (4)The solution of Schrodinger wave equation for the allowed energies of an atom gives the wave FUNCTION `Psi`,which represents an atomic orbital.The wave nature of electron present in an orbital can be well defined by the wave function `Psi`. (5)The wav e function `Psi` itself has no physical meaning.However,the probability of finding the electron in a small volume dx,dy,dz around a point(x,y,z) is proportional to`|Psi(x,y,z)|^(2)`dxdydz`|Psi(x,y,z)|^(2)` is known as probability and is always POSITIVE. |
|
| 33. |
Which of the following is correct IUPAC ? |
|
Answer» 1-ethyl-3-isopropyl-5-propyl CYCLO HEXANE |
|
| 34. |
What are the conditions under which the relation between 'V' and 'n' are plotted ? |
|
Answer» <P>At CONSTANT P |
|
| 35. |
Which one is used as an purifier in space craft? |
|
Answer» QUICK lime |
|
| 36. |
Which is a natural colloidal |
|
Answer» SODIUM chloride |
|
| 37. |
Weight of H_(2)O_(2) present in 560 ml. of 20 vol. H_(2)O_(2) solution is approximately |
|
Answer» 69 g 20 vol _______ ? = 6% 100 ML ________ 6 grams 560 ml _________ ? = 33.6 GMS |
|
| 38. |
When Br_(2) is mixed with phosphorous, first PBr_(3) is formed till all phosporous has reacted. If Br_(2) still remains, PBr_(5) is formed as far as possible. If equal masses of Br_(2) and P are mixed, which of the following will surely remain in the reaction mixture |
|
Answer» P If equal MASSES of PHOSPHOROUS and bromine are mixed, phosphorous is excess REAGENT. |
|
| 39. |
Which of the following can be used in Friedel Crafts acylation reactions ? |
|
Answer» `CH_(3)COOCH_(3)` |
|
| 40. |
Which of the following compound does not react with metallic sodium? |
|
Answer» `CH_(3)CH(OH)CH_(3)` |
|
| 41. |
Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure P_(i) and temperature T_(1)are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to T_(2). The final pressure P_(f) is : |
|
Answer» `p_(i) ((T_(1)T_(2))/(T_(1) + T_(2)))` `:.` Initial moles `=` FINAL moles `(p_(i)V)/(RT_(1)) + (p_(i) V)/(RT_(1)) = (p_(f) V)/(RT_(2)) + (p_(f)V)/(RT_(1))` `(2p_(i))/(T_(1)) = p_(f) [(1)/(T_(1)) + (1)/(T_(2))]` `p_(f) = 2p_(i) [(T_(2))/(T_(1) + T_(2))]` |
|
| 42. |
Which of the following ratio of volume of positive and negative ions are minimum ? |
| Answer» Answer :C | |
| 43. |
Which of the following statement(s) is/areincorrect fora reaction ,A_(2)(g) + B(g) to A_(2) B(g) assuming ideal gas behaviour if Delta H_(R)^(@)=-100 Kcal //"mole"andDelta S_(R)^(@)=- (1)/(3) kcal //" mole"K attemperature T=300 K. |
|
Answer» The reaction is at EQUILIBRIUM when occuredat 1 BAR pressure of each gas and 300 K. |
|
| 44. |
Which salt is significantly more soluble in a strong acid than in water? |
|
Answer» `PbF_(2)` |
|
| 45. |
Write the products P, Q, R and S in the given reaction sequence. |
|
Answer» |
|
| 46. |
Which substance are present in industrial effluents ? |
|
Answer» HEAVY METALS |
|
| 47. |
Which of the hydrolysis resction of halides of III - A group elements is correct. |
|
Answer» `AlCl_(3)+3H_(2)O rarr Al(OH)_(3)+3HCl` |
|
| 49. |
What is the effect of reducing the volume on the system described below ? 2 C (s) + O_(2) (g) hArr 2 CO (g) |
| Answer» Solution :On reducing the volume, the pressure will increase. By LE Chatelier's PRINCIPLE , equilibrium will SHIFT to the side accompanied by decrease of pressure, i.e., decrease in the number of gaseous MLES, i.e., BACKWARD sirection. | |