Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which alkali metal is radioactive element? |
|
Answer» Cesium |
|
| 2. |
Which of the followingg reactions of methane is incomplete combustion? |
|
Answer»
|
|
| 3. |
Which are having less volume between Mg and Mg^(+2) ? |
| Answer» SOLUTION :`Mg^(+2)` having LESS volume than Mg because effective nuclear charge of `Mg^(+20` ion is more. | |
| 4. |
Which of the following compounds gives curdy white precipitate in Lassaigne's test? |
|
Answer» `CH_(3)Br` |
|
| 5. |
When an ideal gasexpands into vaccum, there is neither absorption nor evolution of heat. Why? |
| Answer» SOLUTION :In an IDEAL gas, there are no intermolecular forces of attraction. Hence, no energy is required to overcome these forces,. Moreover, when a gas expands against vacuum,work doneis zero( because`P_(ext) =0)`. Hence, INTERNAL energy of the SYSTEM does not change,i.e., there is no ABSORPTION or evolution of heat. | |
| 6. |
The spotsof thecompounds havingthepropertyof fluoresencearedetectedbyplacing theTLCplateunder________. |
| Answer» Answer :A | |
| 7. |
Upon heating phthalic anhydride with phenol in the presence of a little conc. H_(2)SO_(4)........is formed |
|
Answer» PHTHALIC acid |
|
| 8. |
Which of the following can be included in the category of colloids? |
|
Answer» Milk |
|
| 9. |
Which of the following practices will not come under green chemistry ? |
|
Answer» If possible, making use of soap made of vegetable oils INSTEAD of using synthetic detergents. |
|
| 10. |
What is COD? |
| Answer» Solution :Chemieal oxygen demand (COD) is detined as the amount of oxygen required by the organic MATTER in a sample of WATER for its OXIDATION by a strong oxidising agent like `K_(2)Cr_(2)O_(7)` ACIDIC medium for a period of 2 HOURS. | |
| 11. |
Write the reaction of thermal decomposition of sodium azide. |
|
Answer» SOLUTION :Sodium AZIDE decomposes to form sodium METAL and `N_(2)` gas. `2NaN_(3)OVERSET(Delta)to2Na+3N_(2)`. |
|
| 12. |
When orthoboric acid (H_(3)BO_(3)) is heated strongly the residue left is |
|
Answer» boron |
|
| 13. |
This section contains questions each with two columns-I and II .Match the itemsgiven in columnI with that in column II. |
|
Answer» (b-p,r,s,) `C_(4)` axis (tetrad axis) PASSES through two face-centered ions, body -centred ION (OV). (c-r) Rectangular plane passes through 4 face-centreed ions, 4 edge-centered ions, body-centered ion. |
|
| 14. |
Which of the following will have most negative electron gain enthalpy? P,S,Cl,F |
|
Answer» |
|
| 15. |
Which or the following molecular orbitals has maximum number of nodal planes ? |
|
Answer» `sigma^(**) 1S` NODAL PLANE are , `sigma^(**) 1s = 1, sigma^(**) 2p_(z) = 1 , pi 2p_(x) = 1 , pi^(**) = 2 ` 
|
|
| 16. |
Which of the ions has largest ionic radius? |
|
Answer» `Be^(2+)` |
|
| 17. |
Which of the following covalent ions are highly present in biological compound ? |
| Answer» ANSWER :A | |
| 18. |
Which one of the following is independent to the mass of the system? |
| Answer» Answer :C | |
| 19. |
Which elements does not give Hydride ? |
|
Answer» Te |
|
| 20. |
The well known mineral fluorite is chemically calcuim fluride. It is well known that in one unit of this mineral, there are4 Ca^(2+)ions and8 F^(-) ions, and thatCa^(2+)ions are arranged in a fcc lattice. TheF^(-)ions fill all the tetrahedral holes in the face - centred cubic lattice ofCa^(2+) ions. The edge of the unit cell is5.46 xx 10^(-8) cm in length . The density of the solid is 3.18 " g cm"^(-3) . Use this information to calculate Avogadro's number ( Molar mass ofCaF_(2)= 78.08 " g mol" ^(-1)) |
|
Answer» |
|
| 21. |
Which stoichiometric defect in crystals increases the density of a solid ? |
| Answer» SOLUTION :Intersitital DEFECT. | |
| 22. |
What is hydrolysis ? Give difference between unhydrolyse and hydrolyse ion. |
|
Answer» Solution :Definition : The process of interaction between WATER and cations/anions or both of salts is CALLED hydrolysis. Salts FORMED by the REACTIONS between acids and bases in definite proportions, undergo ionization in water . The cations/anions formed on ionization of salts (i) Ether exist as hydrated ions in aqueous solutions or (ii)interact with water to reform corresponding acids/bases depending UPON the nature of salts. The pH of the solutiongets affected by this interaction. 
|
|
| 23. |
What is the formula of yellow ppt. (E)? |
|
Answer» `HgSO_(4)` |
|
| 24. |
Which reaction is known as Kharash reaction ? |
| Answer» Solution :REACTION of `Br_(2)` with ASYMMETRICAL ALKENE in presence of peroxide is known as KHARASH reaction. | |
| 25. |
Two pure substances of equal molar masses are alpha and beta (where alpha = AHCO_(3) and (beta = BCO_(3)] and they decomposes as shown: 2alpha overset(Delta)to H_(2)O + CO_(2) + A_(2)CO_(3)……………….(i) beta overset(Delta)to BO + CO_(2) ...................(ii) 6.2g of mixture of H_(2)O and CO_(2) is obtained by 16.8g of alpha. Find out the molar mass of alpha and beta and value of ((alpha + beta)/(2))^(2). |
|
Answer» 84 each, 168.0 |
|
| 26. |
Write four species which act both as Bronsted acid as well as base. Write their corresponding conjugate acids and bases. |
| Answer» SOLUTION :`{:("Conjugate acid ","SPECIES","Conjugate base",,),(H_(3)O^(+)" "OVERSET(+H^(+))larr,H_(2)O,overset(-H^(+))rarr" " OH^(-),,),(NH_(4)^(+)" "overset(+H^(+))larr,NH_(3),overset(-H^(+))rarr" " NH_(2)^(-),,),(H_(2)CO_(3)" "overset(+H^(+))larr,HCO_(3)^(-),overset(-H^(+))rarr" " CO_(3)^(2-),,), (H_(2)SO_(4)" "overset(+H^(+))larr,HSO_(4)^(-)," "overset(-H^(+))rarr" " SO_(4)^(2-),,):}` | |
| 27. |
Which element do you thick would have been named by (a) Lawrence Berkeley Laboratory (b) Seaborg's group |
|
Answer» |
|
| 29. |
Which of the carbonates given below is unstable in air and is kept in co, atmosphere to avoid decomposition ? |
|
Answer» `BeCO_(3)` |
|
| 30. |
What is the effect of added inert gas on the reaction at equilibium at constant volume ? |
| Answer» Solution :When an inert GAS (i.e., a gas which does not react with any other species invoved in equilibium) is ADDED to an equilibrium system to constant volume, the total number of moles of gases present in the CONTAINER increases, that is, the total pressure of gases increases, the partial pressure of the reactants and the PRODUCT are unchanged. HENCE at constant volume, addition of inert gas has no effect on equilibrium. | |
| 31. |
Which of the following is non bio-degradable? |
| Answer» Solution :glass | |
| 32. |
The value of n in the equation, Cr_(2)O_(7)^(2-) +14 H^(+)+ne^(-) to 2Cr^(3+) + 7H_(2)O is : |
|
Answer» 2 |
|
| 33. |
Which of the following statement(s) are correct? |
|
Answer» `MnO_(2)` oxidizes halide ions into dihalogen |
|
| 34. |
Write the structural formula , bond line formula common and IUPAC name of C_(5)H_(12). |
Answer» SOLUTION :
|
|
| 35. |
Write expression for molar mass, M ( in kgmol^(-1) )of a body-centred cubic crystal of an ionic compound if it has an edge length of'a' metre and a density of 'd' kg m^(-3) |
| Answer» Solution :`d = (Z xx M)/( a^(3) xx N_(A)) or M = dxx a^(3) xx N_(A)`(as for ionic compound having BCC structure , Z =1) | |
| 36. |
Which of the following does not change during compression of a gas at constant pressure- |
|
Answer» DENSITY of a gas |
|
| 37. |
Which of the following molecules are homoatomic ? (i) H_(2)O, (ii) O_(3), (iii) NH_(3), (iv) Cl_(2) |
|
Answer» |
|
| 38. |
Which metal alloy tanks are used for storage of dihydrogen |
| Answer» Answer :D | |
| 39. |
Which is the correct statement ? |
|
Answer» Minimum BOILING AZEOTROPIC MIXTURE boils at temperature lower than either of the two pure components. |
|
| 40. |
Under what conditions, hydrogen behaves as a metal? |
| Answer» SOLUTION :Under very high PRESSURE, hydrogen BEHAVES as a METAL. | |
| 41. |
Which of the following statement is not true for benzene? |
|
Answer» It is planar molecule |
|
| 42. |
What type of graph will you get when PV is plotted against P at constant temperature. |
| Answer» SOLUTION :A STRAIGHT line parallel to PRESSURE AXIS. | |
| 43. |
Which of the following does not represent polymerisation of alkenes and alkynes? |
|
Answer» `nCH-=Chto(--CH=CH-CH=CH--)_(n)` A linear polyethene polymer is FORMED when ethene polymerises. |
|
| 44. |
Which reaction indicates the oxidising behaviour of H.... |
|
Answer» `2PCl_(5) +H_(2)SO_(4) rarr 2POCl_3 +2HCl +SO_(2)Cl_2` |
|
| 45. |
The structural formula of lithium tetrahydrido-aluminate is |
|
Answer» `Al[LiH_(4)]` |
|
| 46. |
Which of the following minerals contain aluminium ? |
|
Answer» Fluorspar Feldspar : `KAlSiO_(3)O_8` Mica : `K_(2) O.3 Al_(2) O_(3) . 6SiO_(2) . 2H_(2) O` Carborundum : Sic |
|
| 47. |
The volume of O_2 gas evolved from 40 gram per litre H_2O_2 at STP is..... |
|
Answer» 22.4 `V(O)_2=11.2xxM` =11.2 x 1.176 =13.18 litre `O_2` RELEASE |
|
| 48. |
Which one of the following has coordinate covalent bond? |
| Answer» Solution :Non metals | |
| 49. |
What is Z in the following sequence of reactions ? Phenol overset("Zn dust")rarr X underset("anhyd." AlCl_(3))overset(CH_(3)Cl)rarr Z |
|
Answer» Toluene 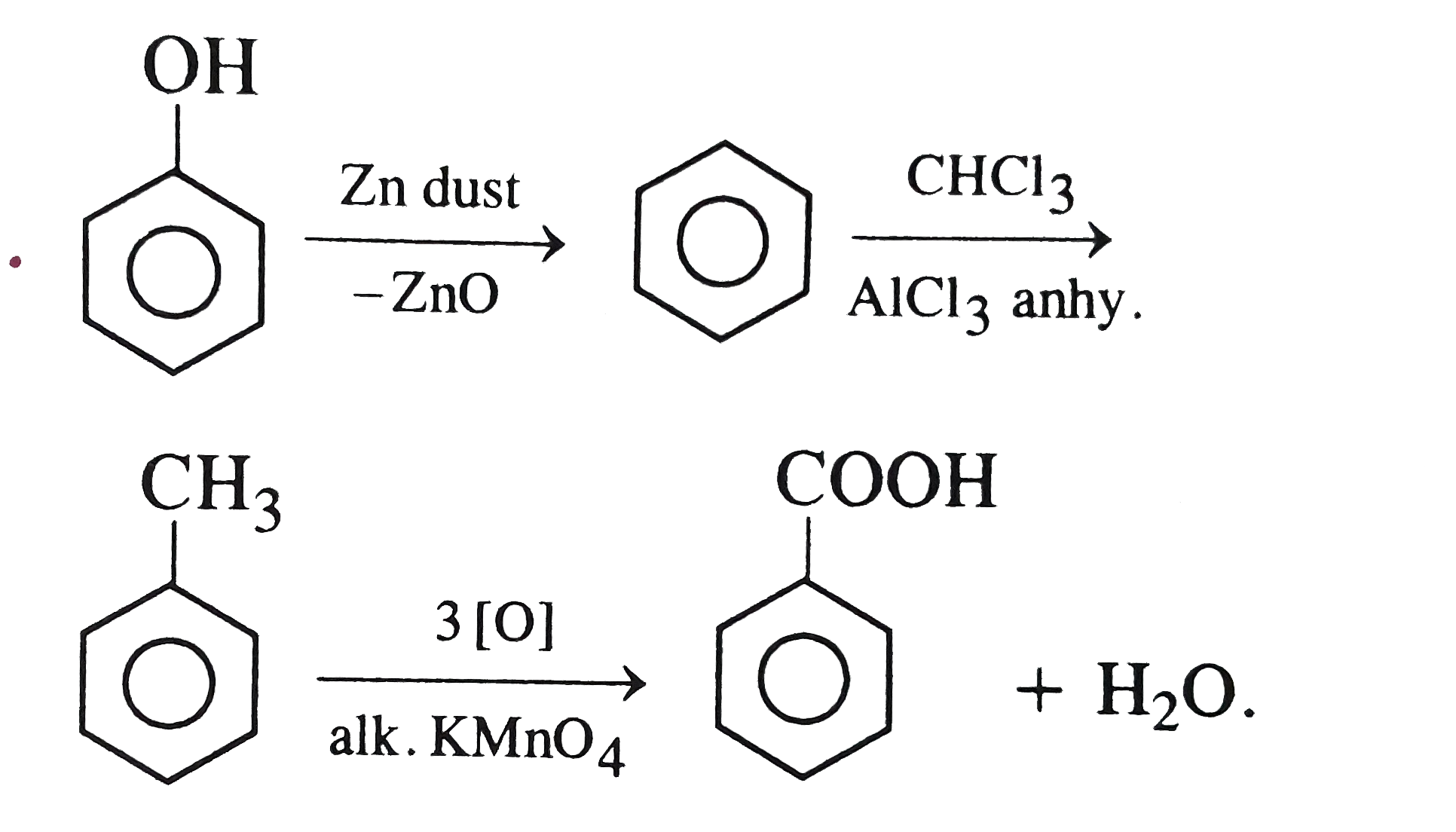
|
|
| 50. |
Which of following statement(s) point is maximum. (P) Slope of isotherm at critical point is maximum. (Q) Larger is the value of T_(c) easier is the liquification of gas. (R) van der Waal's equation of state is applicable below critical temperature at all pressure. |
|
Answer» Only P |
|
