Saved Bookmarks
| 1. |
An atom of anelement 'X' has three completely filled shell . Theratio of electrons in 1 and 4 shells is 1:1. Find the atomic number of 'X' . Alsowrite the formulae of its bicarbonate , chromate and phosphide. |
Answer» Solution :The electronic arrangement of X must be 2 , 8 , 18 , 2 So the ATOMIC NUMBER of X ahould br 30 . The element X is Zn which can form`Zn^(+2)`ion by LOSING 2 electrons from N shell.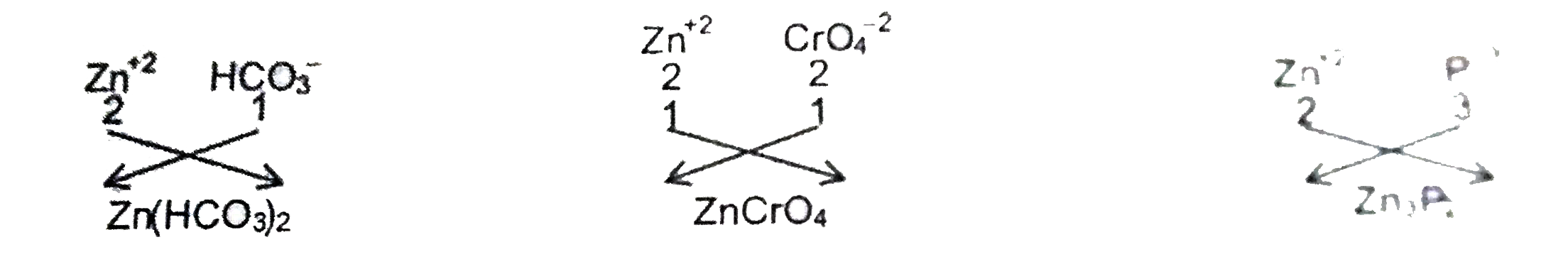
|
|
Discussion
No Comment Found
Related InterviewSolutions
- Which of the following phenomenon is an effect acid rain ?
- Which of the following can be used to reduce suspended particulate matter in atmosphere in mine areas ?
- What are noble gases ? Mention their uses.
- What will be the formula of the sulphate and sulphite of a trivalent metal that isM^(+3) ?
- Which among the following is an element ?
- Xenon and krypton is used in electric bulbs to slow down the sublimation of tungsten.
- Whichof the following substances does not form a curdy precipitate when it is added to hard water ?
- Why are droplets of water observed on the walls of a glass tumbler containing ice ?
- What are homogeneous and heterogeneous mixtures ? Give one example for each.
- Which among the following is true regarding aqueous solution of sulphur trioxide and sodium oxide?