Saved Bookmarks
| 1. |
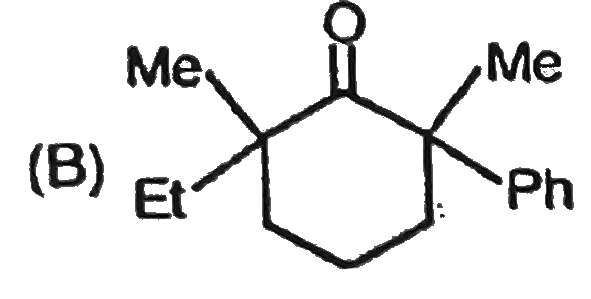
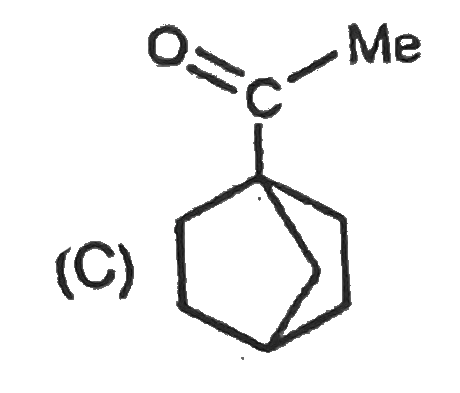
In the presence of excess base and excess halogen a methylketone is converted first into a trihalo substituted ketone and then into a carboxylic acid.After the trihalo substituted ketone is formed hydroxide ion attacks the carboxyl carbon because the trihalo methyl ion is the group more easily expelled from the tetrahedral intermediate.The conversion of a methyl ketone to a carboxylic acid is called a haloform reaction because one of the product is haloform (CHCl_3) or CHI_3 or CHBr_3. Which of the following compound show haloform reaction and racemisation in OD^(-) //D_2O |
|
Answer» `CH_3CH_2OH` If H, present on the chiral carbon, undergoes TAUTOMERISATION we will get racemic MIXTURE. |
|
Discussion
No Comment Found
Related InterviewSolutions
- Which of the following compounds is not cleaved by HI even at 525 K ?
- To a 25 mL H_(2)O_(2) solution excess of an acidified solution of potassium iodide was added. The iodine liberated required 20 " mL of " 0.3 N sodium thiosulphate solution Calculate the volume strength of H_(2)O_(2) solution.
- The suggested mechanism of a reaction is : (a) A+BhArrD("fast) "(b)A+Drarr2C("slow")Write the balanced equation of the reaction if its experimentally deduced rate equation is , rate k=[A]^(2)[B] Find the intermediate formed during the course of the reaction . Does the predicted rate law from the mechanism match the experimental rate law ?
- Which of these changes with time for a first-order reaction A Rate of reaction B . Rate constant C . Half-life
- What is the hybridisation of central atom in the product obtained along with hydrofluoric acid when complete hydrolysis of Xenon Hexa Fluoride takes place ?
- Which of the following amino acid forms sulphide bond in polypeptide
- Which of following pair is Diastereomers:
- What is the major product of the following reaction CH_3C-=C-CH_2-CH_3overset("1 mole of " Cl_2)to
- Which polymer is used in petrol tank linings ?
- Which of the following carbohydrates are branched polymer of glucose ?