Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Result of 3.421-0.12 is |
|
Answer» 3.301 |
|
| 2. |
Respiration involves "_________________" reaction. |
|
Answer» only OXIDATION |
|
| 3. |
Resonating structures of a molecule should have |
|
Answer» IDENTICAL arragmentof atoms |
|
| 4. |
Resonance structures of propenat are give below. Which of these resonating structures is more stable? Give reason for your answer. underset((I))(CH_(2)=CH-CH=O)harr underset((II))(overset(oplus)(C )H_(2)-CH=CH- overset(Θ)(O)) |
Answer» SOLUTION :STRUCTURE (I) is more STABLE because, 
|
|
| 5. |
Resonance structures of propenal are given below. Which of these resonating structures is more stable ? Give reason for your answer. underset(I)(CH_(2)=CH-CH=O) harr underset(II)(overset(o+)(C)H_(2)-CH=CH-overset(Θ)(O)) |
Answer» Solution :Resonance STRUCTURES of PROPENAL are shown below : 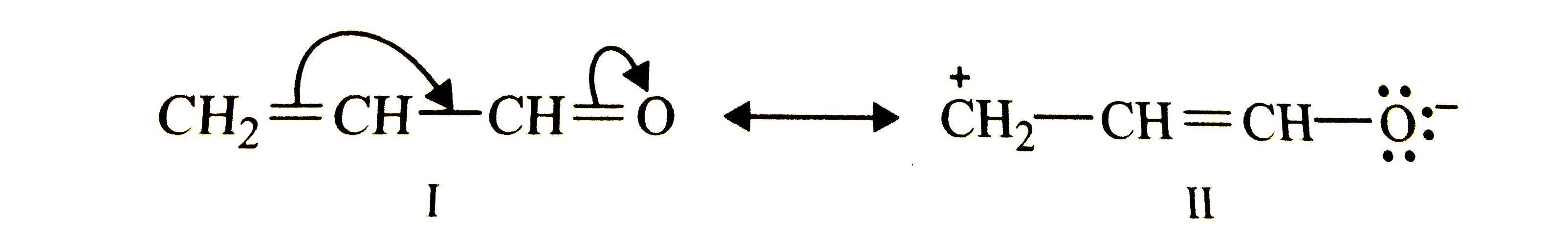 Structure II INVOLVES separation of +ve and -ve charges. Furthermore, the terminal carbon has only a sextet of electrons. Due to these two FACTORS, structure (II) is less stable than structure (I). |
|
| 6. |
Resonance structures of a molecule should have |
|
Answer» IDENTICAL arrangement of atoms |
|
| 7. |
Resonance structure of the molecule does not have |
|
Answer» higher ENERGY than their HYBRID structure. |
|
| 8. |
Resonance phenomenon is generally involved when single valence bond structure could not represent the actual structure of the molecules or species. Based on the actual bond lengths, bond energies etc, of the molecules or species, resonance phenomenon will help to achieve at the correct structure of the species or molecules. Which of the following is correct |
|
Answer»
|
|
| 9. |
Resonance is not exhibited by |
|
Answer» PHENOL |
|
| 10. |
Resonance involves : |
|
Answer» DELOCALIZATION of `PI`-electrons along a CONJUGATED system. |
|
| 11. |
Resonance in benzene is accompanied by delocalisation of pi electrons. Each pi electron is attached with |
|
Answer» FOUR CARBON electrons |
|
| 12. |
Resonance energy per benzene ring is maximum for |
|
Answer» BENZENE |
|
| 13. |
Resonance energy will be more if |
|
Answer» canonical strutures are equivalent than if canonical STRUCTURES are non-equivalent. B) molecule is aromatic than if molecule is not aromatic. C) more number of p-bonds are involved in resonance than less number of p-bonds are involved. D) Resonance with 2p-2p overlapping than resonance with 3p-2p overlapping. |
|
| 14. |
Resonance energyof benzene is |
|
Answer» canonical strutures are equivalent than if canonical STRUCTURES are non-equivalent. |
|
| 16. |
Represent the potential energy/enthalpy change in the following processes graphically. (a) Throwing a stone from the ground to roof. (b) (1)/(2) H_(2) (g) + (1)/(2) Cl_(2) (g) hArr HCl (g) Delta_(r) H^(Θ) = - 92.32 kJ mol^(-1) In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity ? |
|
Answer» Solution :Representation of potential energylenthalpy change in the following PROCESSES (a) Throwing a stone from the ground to roof. 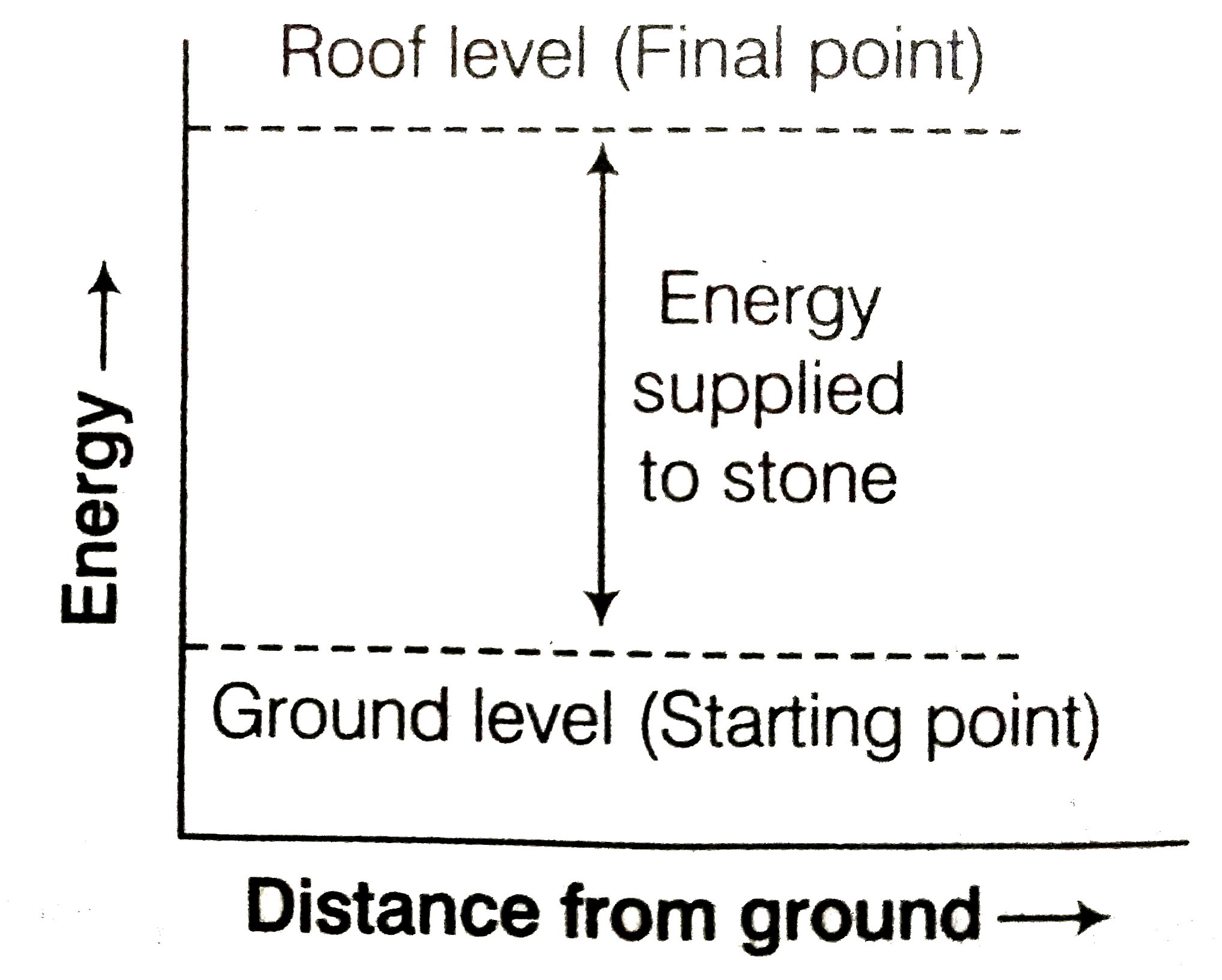 (b) `(1)/(2) H_(2) (g) + (1)/(2) Cl_(2) (g) hArr HCl (g) , Delta_(f) H^(Θ) = - 92.32 kJ MOL^(-1)` 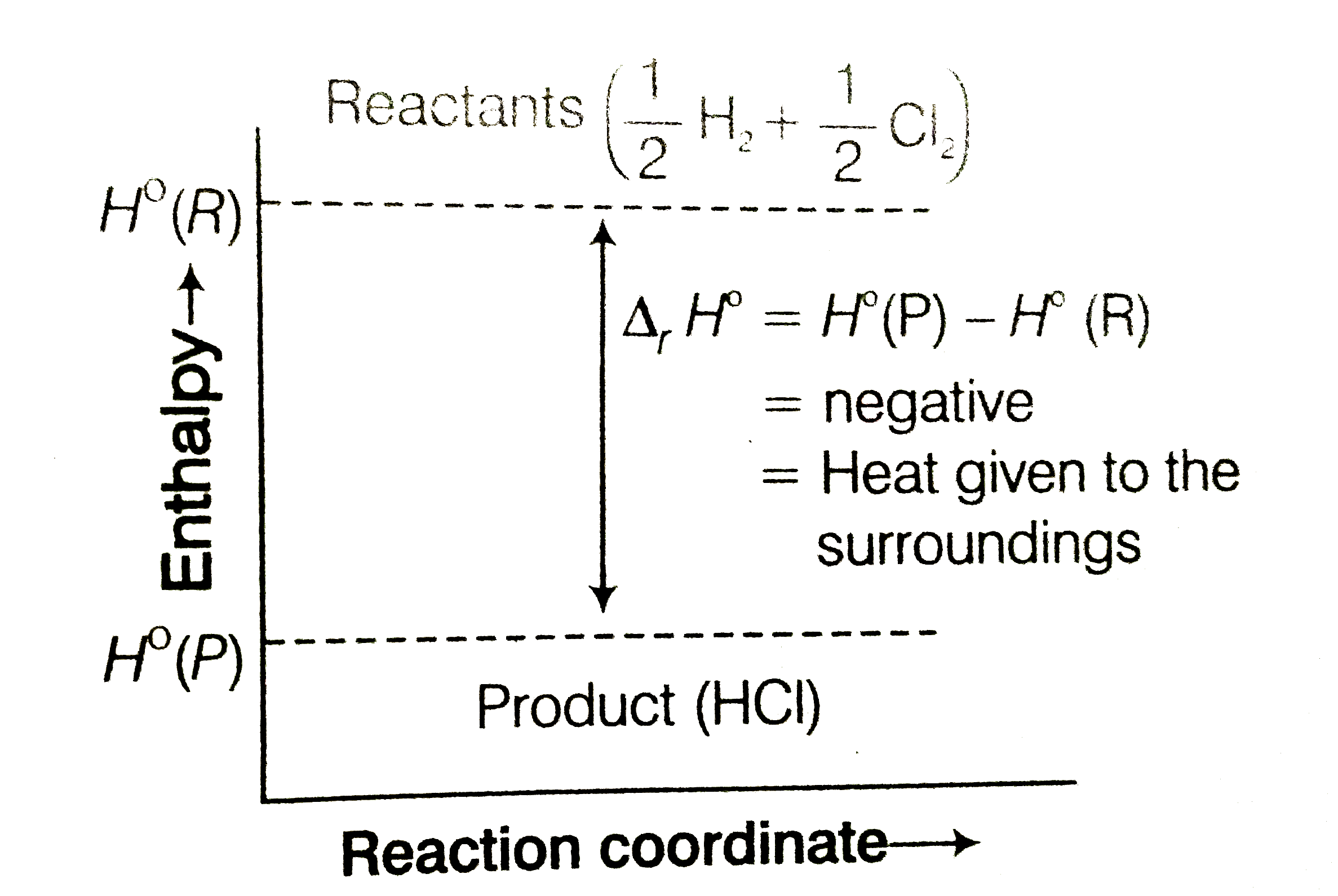 Energy increases in (a) and it decreases in (b) PROCESS. Hence, in process (b), enthalpy change is the contributing FACTOR to the spontaneity |
|
| 17. |
Representative elements are those which belong to |
|
Answer» <P>s and d- blocks |
|
| 18. |
Representation of the orbital with quantum numbers n = 3,1 = 1 is |
|
Answer» 3s |
|
| 19. |
Represent the three type of electron movement in organic reaction |
Answer» SOLUTION :(i) The electron from `pi` bond to ADJACENT bond position  (ii) Electron PAIR from the `pi` bond SHIFT on that atom  (III) From the atom (Y) on its conjugated bond 
|
|
| 20. |
Represent the potential energy // enthalpychange in the followingprocesses graphically : (a) Throwing astone from the ground to roof. (b)(1)/(2)H_(2)(g)+(1)/(2)Cl_(2)(g)hArr HCl(g) Delta_(r)H^(c-) = - 92.32kJ mol^(-1) In which of the following processpotential energy // enthalpy change is contributing factor to the spontaneity ? |
Answer» Solution :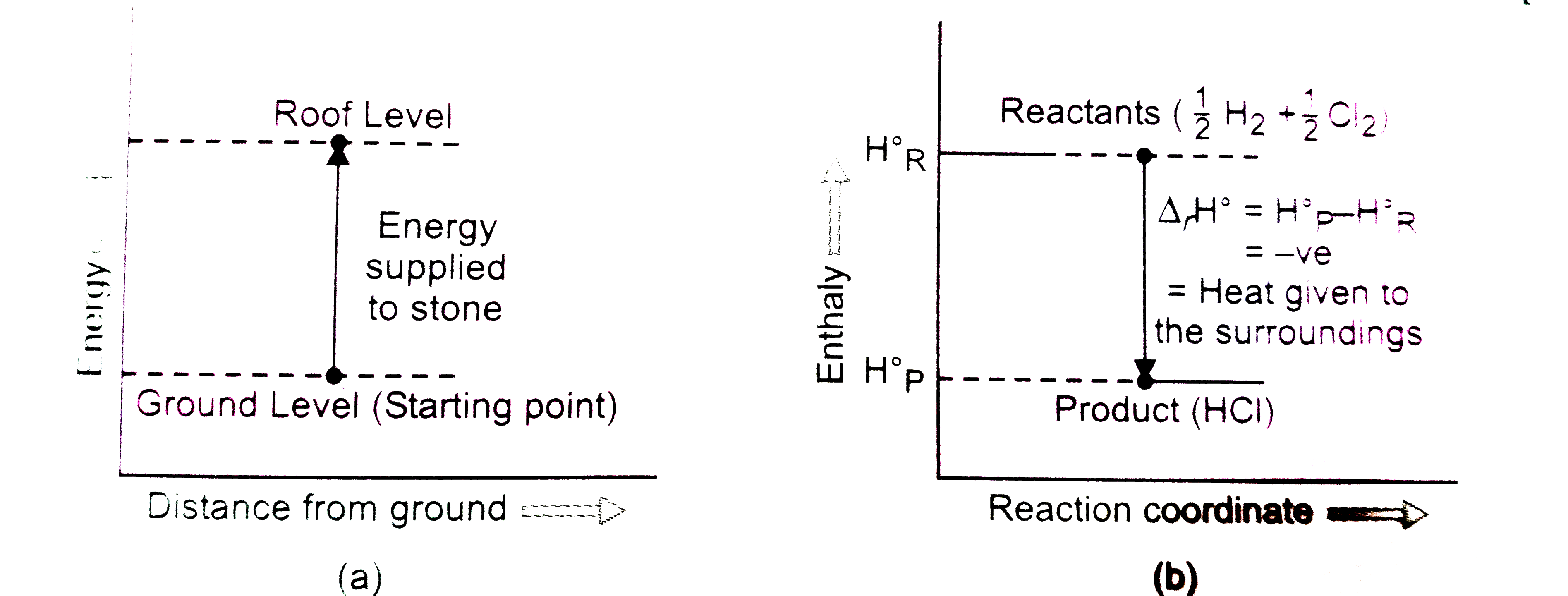 ENERGY INCREASES in (a) and it decreases in (b). HENCE, in process (b), ENTHALPYCHANGE is the CONTRIBUTING factorto the spontaneity. |
|
| 21. |
Represent the potential energy / enthalpy change in the following processes graphically. (a) Throwing a stone from the ground to roof. (b) (1)/(2) H_(2(g)) + (1)/(2) CI_(2(g)) hArr HCI_((g)) Delta_(r) H^( Theta ) - 92.32 "kJ mol"^(-1) In which of the processes potential energy/ enthalpy change is contributing factor to the spontaneity ? |
|
Answer» Solution :Representation of potential energy/ENTHALPY CHANGE in the following process : (a) Throwing as stone from the ground to roof.  (B) `(1)/(2) H_(2(g)) + (1)/(2) CI_(2(g)) hArr HCI_((g)) , Delta_(R) H^( Theta ) = -92.32 "kJ mol"^(-1)`  Energy is increased in (a) and it is decreased in (b) process. Hence, in process (b), enthalpy change is the contributing factor to the spontaneity. |
|
| 22. |
Represent the single electron movement of CH_(3)- Cl bond |
Answer» Solution :The movement of SINGLE electron is indicated by a single BARBED fish hookd (i.e., half headed curved arrow). The curved arrow starts fro the point from where an electron PAIR is SHIFTED and it ends at a location to which the pair of electron MAY move 
|
|
| 23. |
Represent the inductive effect in: (a) CH_(3)COOH, (b) C Cl_(3)COOH and (c ) CH_(3)COOH |
Answer» Solution :(a) `CH_(3)COOH`  There is ELECTRON denoting INDUCTIVE effect (+I) of `-CH_(3)` group There is ELECTRON denoting INDUCTIVE effect (+I) of `-CH_(3)` group (b) `ClCl_(3)COOH`:  There is three `-Cl` are electron attractive group so electron is AWAY from `-COOH` There is three `-Cl` are electron attractive group so electron is AWAY from `-COOH`  There is electron attractive effect of TWO `-Cl`. So, `C-COOH` electron density is more, away from `-COOH` There is electron attractive effect of TWO `-Cl`. So, `C-COOH` electron density is more, away from `-COOH`
|
|
| 24. |
Represent the following equation in ionic form K_(2)Cr_(2)O_(7)+7H_(2)SO_(4)+6FeSO_(4)=3Fe(SO_(4))_(3)+Cr_(2)(SO_(4))_(3)+7H_(2)O+K_(2)SO_(4) |
|
Answer» SOLUTION :In this equation except, `H_(2)O`, all ionic in NATURE. Representing these compounds in ionic forms, `2K^(+)+Cr_(2)O_(7)^(2-)+14H^(+)+7SO_(4)^(2-)+6Fe^(2+)+6SO_(4)^(2-)to6Fe^(3+)+9SO_(4)^(2-)+2Cr^(3+)+3SO_(4)^(2-)+2K^(+)+SO_(4)^(2-)+7H_(2)O` `2K^(+)` ions and `13SO_(4)^(2-)` ions are common on both SIDES, so these are cancelled. The desired ionic equation reduces, to `Cr_(2)O_(7)^(2-)+14H^(+)+6Fe^(2+)=6Fe^(3+)+2Cr^(3+)+7H_(2)O` Total charge are equal on both sides, thus, the balanced ionic equation is the same as above. |
|
| 25. |
Represent the bond formation in [Fe(CN)_(6)]^(4-) and BF_(3)-NH_(3) |
|
Answer» In certain , molecules having a long pair of electrons such as ammonia donates its pair to an ELECTRON deficient molecules such as`BF_(3)*`to form a co-ordinate bond `(##SUR_CHE_XI_VO2_S_MQP_02_E01_041_A01##)` `(##SUR_CHE_XI_VO2_S_MQP_02_E01_041_A02##)` |
|
| 26. |
Represent the cell corresponding to the following redox reaction and identify the anode and the cathode. Cd(s)+2Ag^(+)(aq)toCd^(2+)(aq)+2Ag(s) |
| Answer» SOLUTION :`CD(s)|Cd^(2+)(AQ)||AG^(+)(aq)|Ag` | |
| 27. |
Represent the bond formation in [Fe(CN)_(6)]^(4-)" and " BF_(3) - NH_(3) |
|
Answer» SOLUTION :In ferrocynide ion `[Fe(CN)_(6)]^(4-)`,eachcyanide ion `(CN^(-))` donates a pair of electrons to form a co-ordinate BOND with iron `(Fe^(2+))` and these electrons are shared STRUCTURE of Ferrocyanide ion by `Fe^(2+)` and `CN^(-)`. In CERTAIN cases, molecules having a lone pair of electrons such as ammonia donates its pair to an electron deficient molecules such as `BF_(3)` . to form a co-ordinate bond. 
|
|
| 28. |
Represent hyperconjugation in CH_(3) overset(+)(C )H_(2) and CH_(3)CH= CH_(2) by only figure |
Answer» SOLUTION :
|
|
| 29. |
Represent structure of the free radical, carbocation and carbanion from methane. |
Answer» SOLUTION :
|
|
| 30. |
Represent hydrogen bonding between two molecular of (i) acetic acid (CH_(3) COOH)(ii) acetamide (CH_(3) CONH_(2)). |
Answer» SOLUTION :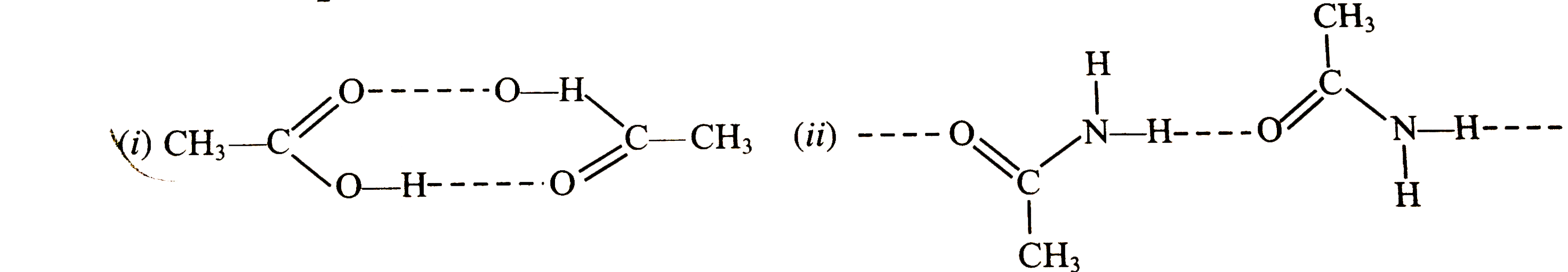
|
|
| 31. |
Represent different type of periodic properties in periodic table. |
|
Answer» Solution : When we go left to right in PERIOD then electron gain ENTHALPY. Ionization enthalpy and ELECTRONEGATIVITY increases. When atomic number increases atomic RADIUS decreases in period. In top to BOTTOM atomic radius increases in group. In top to bottom in group electronegativity ionisation enthalpy, electron gain enthalpy decreases. When we go left to right in periodic table nonmetallic diagonal properties increases and metallic properties decreases. 
|
|
| 32. |
Represent diagrammatically the bond moments and the resultant dipoJe moment in CO_(2), NF_(3) and CHCl. |
Answer» SOLUTION :
|
|
| 33. |
Report your answer as XY |
Answer» 
|
|
| 34. |
Report the volume at STP accupied by(a) 6.022xx10^(22) H_(2)S molecules and (b) 0.25 mol of nitrogen |
|
Answer» |
|
| 35. |
Report the area of a rectangle to approprite number of significant figures. Given that the edge lengths of the rectangle are 6.221 cm and 5.2 cm. |
|
Answer» |
|
| 36. |
Report the value of gas constant in cal K^(-1) molecule |
|
Answer» SOLUTION :Gas CONSTANT per molecule of a gas is called Boltzmann constant (k). It is GIVEN as ` K= ( R)/(N_0) = ( 1.987)/( 6.023 xx 10^(23)) = ( 19.87 )/( 6.023 ) xx 10^(-24)` Gasconstantpermolecule `= 3.3 xx 10^(-24) CALK^(-1)" molecule"^(-1)` |
|
| 37. |
Report the prouduct of 4.5 and1.25 in significant figures. |
|
Answer» |
|
| 38. |
Removal of CO_2from a -COOH group is called decarboxylation. |
|
Answer» |
|
| 39. |
Release of oxides of nitrogen and hydrocarbons into the atmosphere by motor vehicles is prevented by using........ |
|
Answer» GRIT chamber |
|
| 40. |
Relationship between formed products is : |
|
Answer» ENANTIOMER 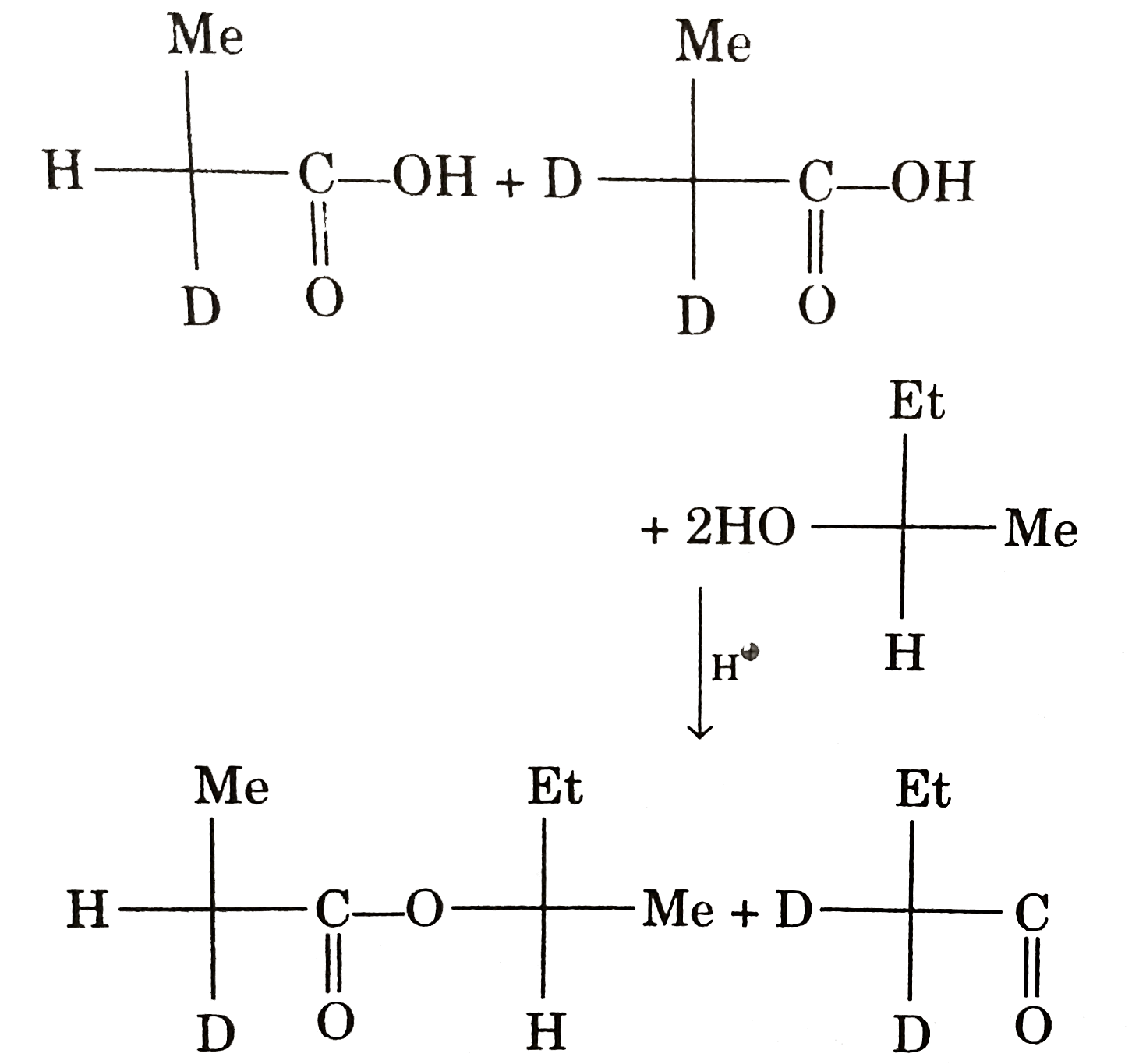
|
|
| 41. |
Relationship between different types of speeds. |
|
Answer» Solution :Most PROBABLE speed `(u_(mp))=sqrt((2RT)/(M))` Average speed, `(u_(av))=sqrt((8RT)/(pi M))` ROOT mean square speed, `(u_(RMS))=sqrt((3RT)/(M))` RELATION between three speeds are as under. `u_(rms)gt u_(av)gt u_(mp)`  ratio between three speeds are as under, `{:(,u_(mp),:,u_(av),:,u_(rms)),(,1,,1.128,,1.224),("OR",0.82,,0.92,,1.00):} ""`......(EQ. -i) `u_(rms)=sqrt((3RT)/(M))=sqrt((3pV)/(M)) = sqrt((3p)/(d)) ""`.....(Eq. -ii) `u_(av)=0.92133xx u_(rms)` `u_(mp)=0.816xx u_(rms)` |
|
| 42. |
Relation between oxidation numbers and electronegativity is given by |
|
Answer» PAULING's SCALE |
|
| 43. |
Relation between K_(1)andK_(2) is : |
|
Answer» `K_(1)=K_(2)` |
|
| 44. |
Relation between given pair is : |
|
Answer» enantiomer 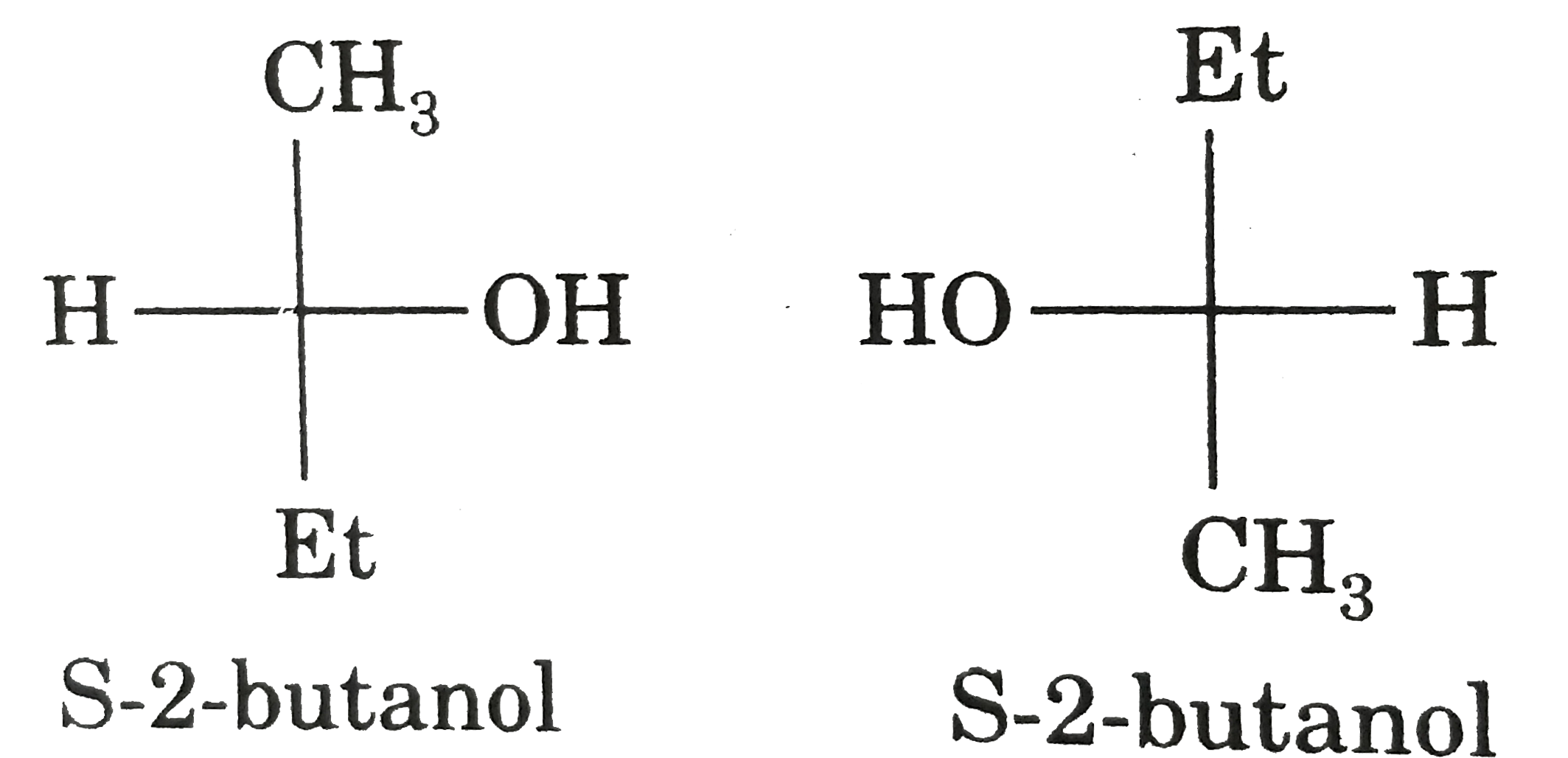
|
|
| 45. |
Relation between 1 L = _____ dm^3 |
|
Answer» 1 |
|
| 46. |
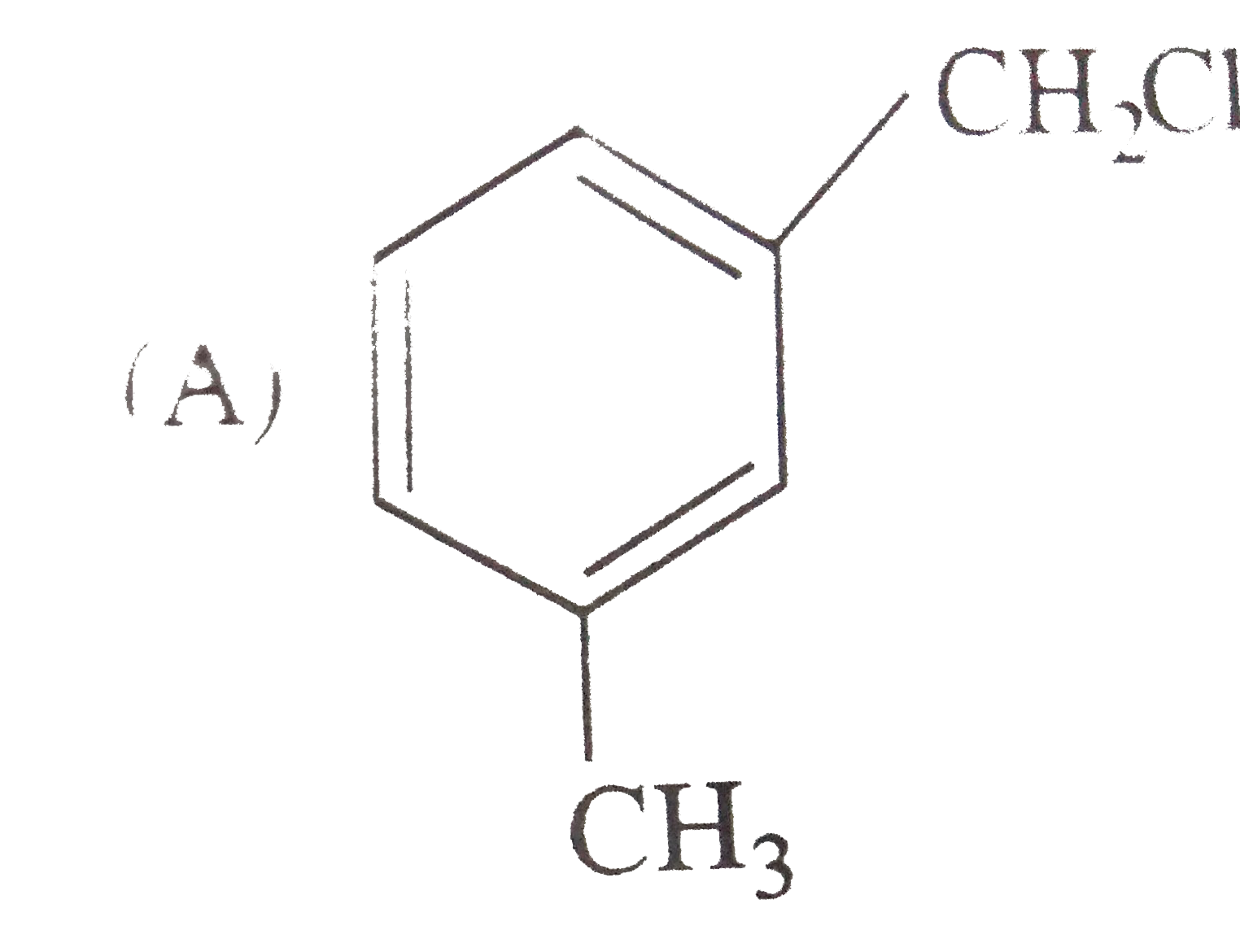
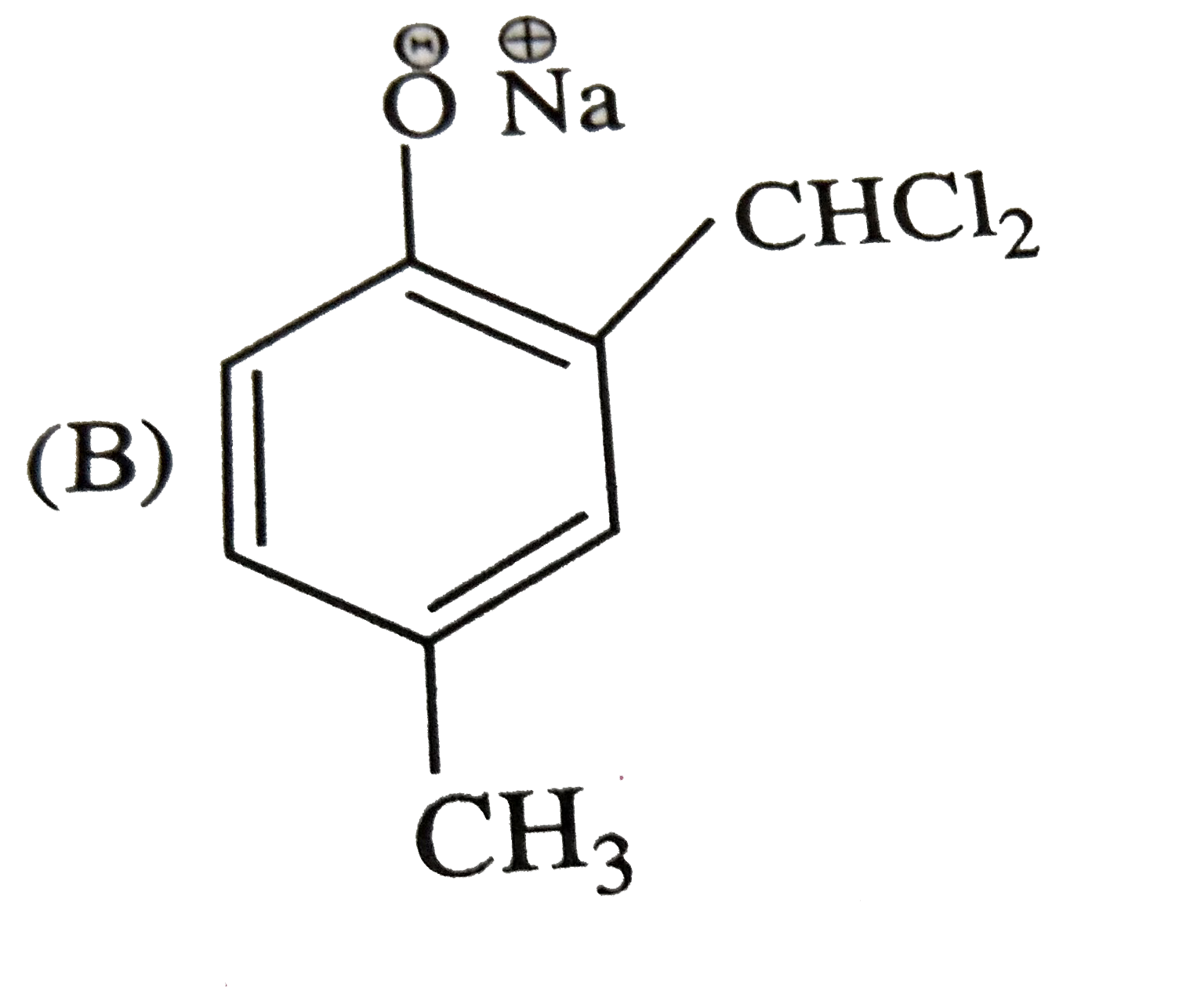
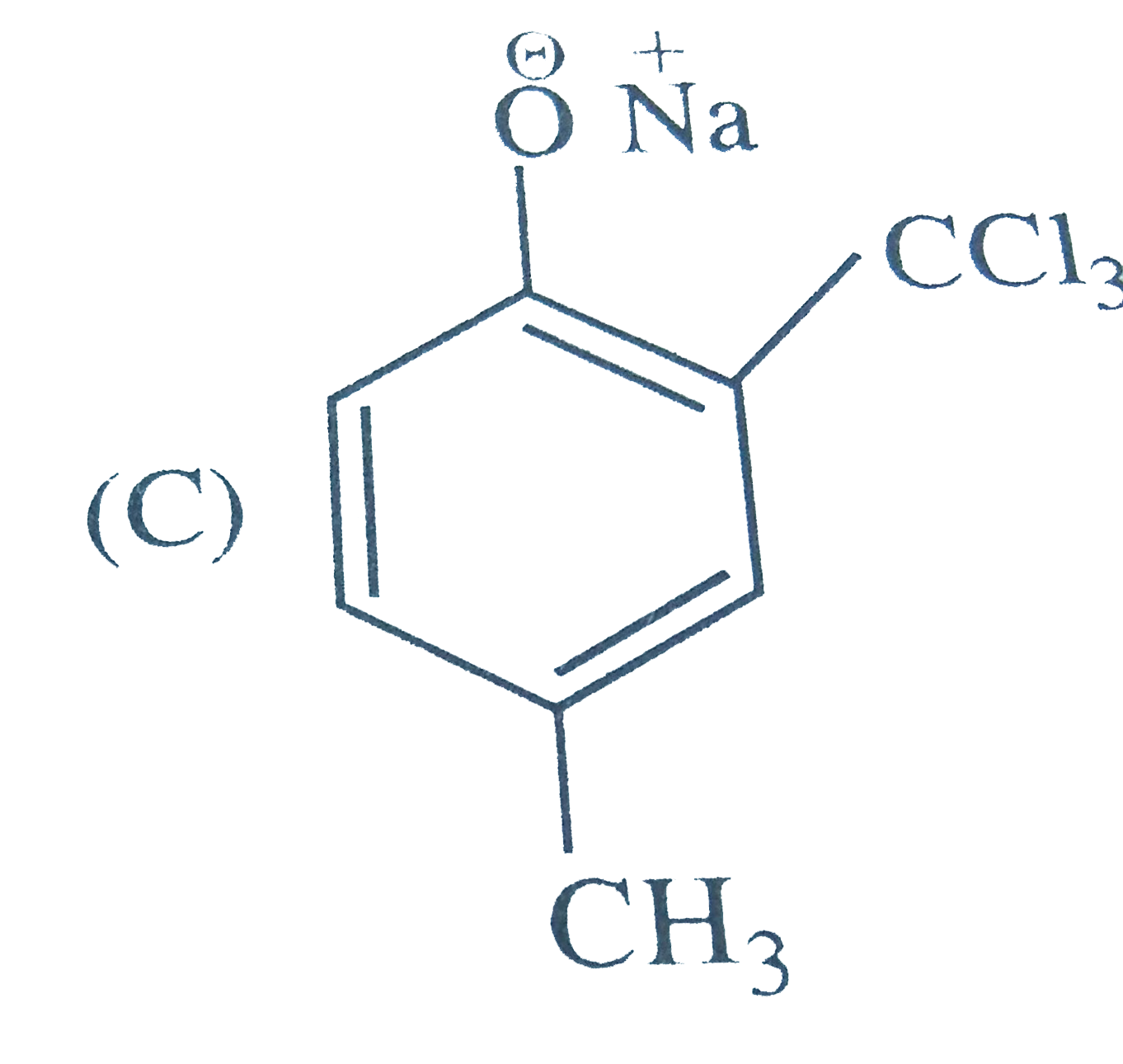
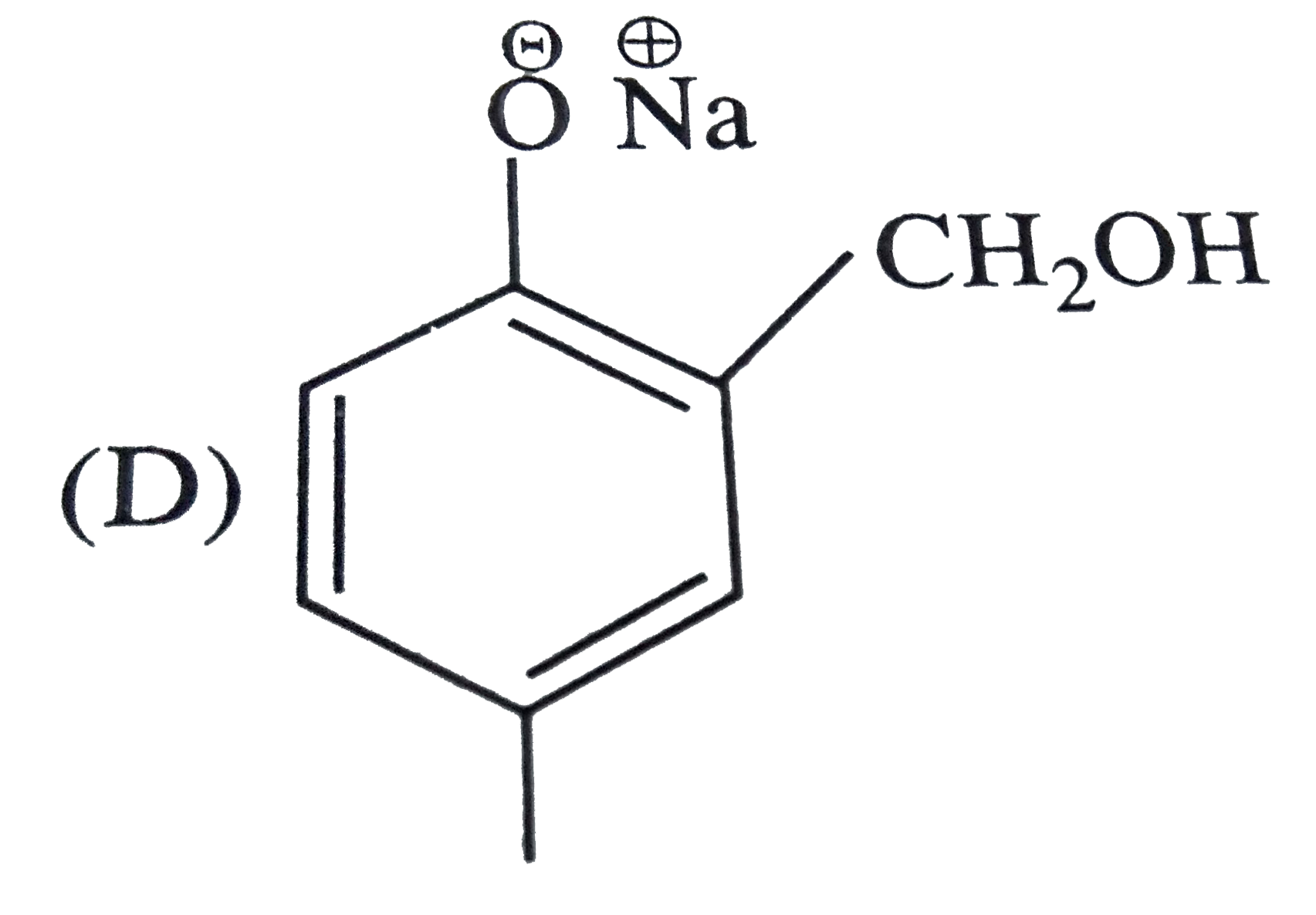
Reimer Tiemann reaction introduces an aldehyde group, on to the ring of phenol, ortho to hydroxyl group. The reaction involves electro-philic aromatic substitution. This is a general method for synthesis of substituted salicyladehyde as depicted below The structure of the intermediate is |
|
Answer»

|
|
| 47. |
Reimer Tiemann reaction introduces an aldehyde group, on to the ring of phenol, ortho to hydroxyl group. The reaction involves electro-philic aromatic substitution. This is a general method for synthesis of substituted salicyladehyde as depicted below Which one of the following reagents is used in the above reactions |
|
Answer» aq `NAOH+CH_(3)Cl` `Cl_(3)C-H UNDERSET(-H_(2)O)overset(OH^(-))rarr Cl_(3)C^(-)` |
|
| 48. |
Reimer Tiemann reaction introduces an aldehyde group, on to the ring of phenol, ortho to hydroxyl group. The reaction involves electro-philic aromatic substitution. This is a general method for synthesis of substituted salicyladehyde as depicted below The electrophile in this reaction is |
|
Answer» `:CHCl` `underset("base")(OH^(-))+CHCl_(3) underset(-H_(2)O)rarr : overset(-)(C) Cl_(3)underset(-Cl^(-))rarr underset(("ELECTROPHILE"))underset("chlorocarbene") ( :C Cl_(2))` |
|
| 49. |
Regions of the atmosphere showing decrease in temperature with altitude are ___ |
|
Answer» Troposphere |
|
| 50. |
Regarding the surface tension property of liquids, the correct statements is/are a) surface tension decreases with increase of temperature b) at 20^@ C the surface tension of ethanol is greater than that of water c) the dimensions of surface tension is SI units is Nm^(-1) d) in the fine polishing of the glass, the edges become smooth due to surface tension |
| Answer» Answer :B | |