Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Show that cylopentadienyl cation is a diradical. Strategy: Use the polygon-and-circle method for deriving the relative energies of the pi MOs |
Answer» SOLUTION : Cyclopentadienyl CATION, has four `pi` ELECTRONS its electron DISTRIBUTION is: 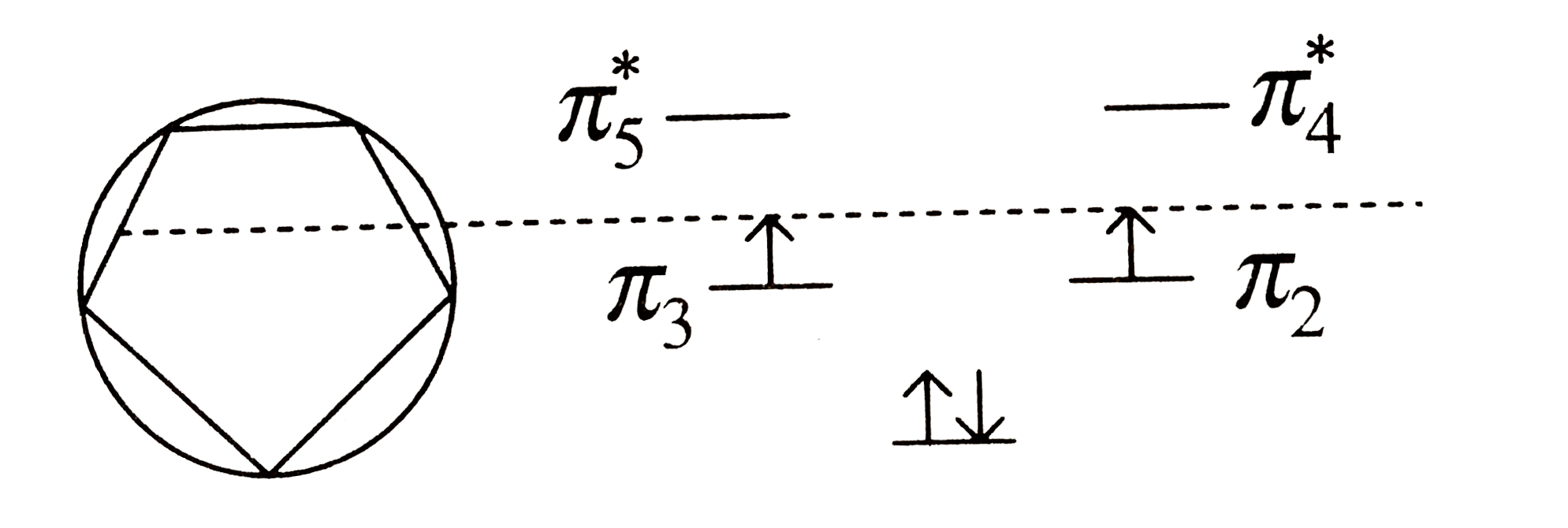
|
|
| 2. |
Show that ethylene dichloride undergoes (i) Hydrolysis (ii) Dehalogenation (iii) Dehydrohalogenation. |
|
Answer» Solution :(i) Hydrolysis: `underset("Ethylene DICHLORIDE")(underset(Cl)underset(|)(C)H_(2)-underset(Cl)underset(|)(C)H_(2)) underset((aq))OVERSET(2KOH)to` `underset("Ethylene glycol")(underset(OH)underset(|)(C)H_(2)-underset(OH)underset(|)(C)H_(2))+2KCl` (ii) Dehalogenation: `underset("Ethylene dichloride")(underset(Cl)underset(|)(C)H_(2)-underset(Cl)underset(|)(C)H_(2))+Zn underset(Delta)overset("Methanol")tounderset("Ethylene")(CH_(2)=CH_(2))+ZnCl_(2)` (iii) Dehydrohalogenation: `underset("Ethylene dichloride")(H-underset(H)underset(|)overset(Cl)overset(|)(C)-underset(Cl)underset(|)overset(H)overset(|)(C)-H)+2KOH underset(Delta)overset("Ethanol")to underset("Acetylene")(HC-=CH)+2KCl+H_(2)`. |
|
| 3. |
Show that entropy as a state function. |
|
Answer» Solution :Consider a cylinder fitted with a frictionless and weightless piston, which contains a gas and is in contact with a large heat reservoir. During isothermal and REVERSIBLE expansion of the gas from volume `v_(1)` to `v_(2)`, let there be absorption of heat q at temperature T. `:.` Change in ENTROPY of the system. `Delta S_(sys) = (q_(rev))/(T)` Since an EQUIVALENT amount of hest will be lost by reservoir, change in entropy of reservoir will be `Delta S_(res) = Delta S_(sys) + Delta S_(res) = (q_(rev))/(T) + ((-q_(rev))/(T)) = 0` If we now, compress the gas isothermally from a volume `v_(2)` to `v_(1)`, heat given by the system is `-q_(rev)`. `:. Delta S_(sys) = (-q_(rev))/(T)` and `Delta S_(rev) = (q_(rev))/(T)` Total change in entropy `Delta S_(2) = (-q_(rev))/(T) + (q_(rev))/(T) = 0` Total change in entropy for the complete cycle `Delta S_(1) + Delta S_(2) = 0` At the END of the cycle, the entropy of the system is the same as it initially. Therefore, entropy is a state function. |
|
| 4. |
Show that banzene is aromatic by using Huckel's rule. |
|
Answer» Solution :In BENZENE, `6PI` electrons are PRESENT . `therefore` n = 1 wkt Huckel's rule is `(4N + 2) pi` electrons. `therefore (4 XX 1 + 2)pi = 6pi` electrons Benzene is aromatic because it obeys Huckel rule and it is planar. |
|
| 5. |
Show steps to prepare (E) -pent-2-ene from acetylene. |
Answer» SOLUTION :
|
|
| 6. |
Show how the carbanion overset(ɵ)CH_(2)-overset(O)overset("||")(C)_CH_(3) is stabilised by resonance. |
Answer» SOLUTION :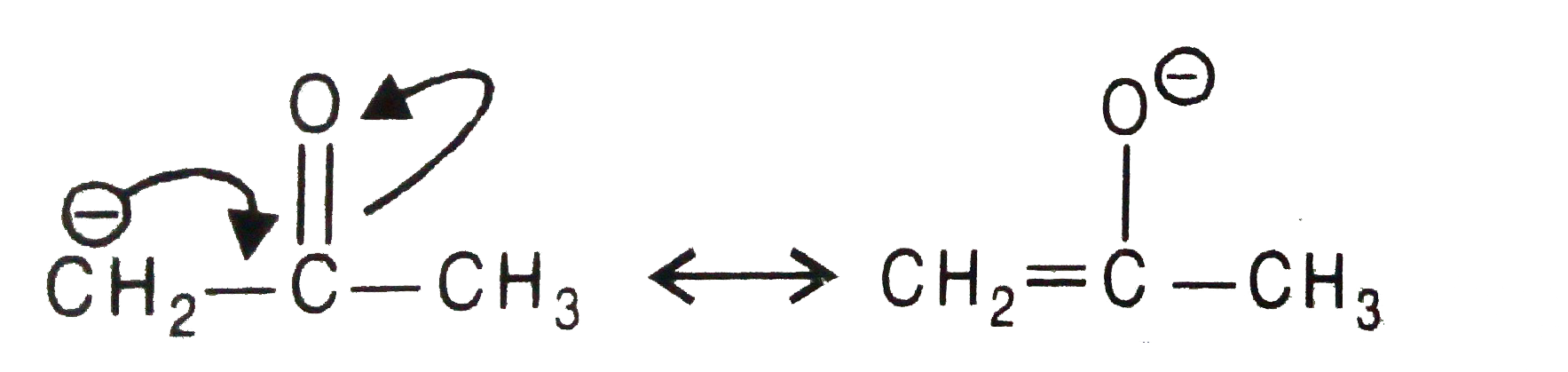
|
|
| 7. |
Show how the bond order is related to stability and bond length of the molecule. |
|
Answer» Solution :Bond order `alpha` Stability Greater the bond order , more stable the molecule is EG : `N_(2)` (B.O = 3) is much stable than `O_(2)` (B.O = 2) Bond order ` alpha1/("Bond LENGTH ")` Greater the bond order SHORTER is the bond length . Eg : Nitrogen: B.O3 , Bond length = 110 pm OXYGEN : B.O . = 2, Bond length = 121 pm. |
|
| 8. |
Show dipole moment of H_(2)O and BeH_(2)by figure . |
Answer» Solution :`H_(2)`O MOLECULE is ANGULAR and bond angleH - O- H is `104.5^(@)` . `BeH_(2)` is leaner and bond ANGLE H - Be - H is leaner. 
|
|
| 9. |
Show difference of polarity of N - 11 bond and N - F bond . |
Answer» SOLUTION : 
|
|
| 10. |
Show by an arrow the preferred product of reaction with E^(+) of each of the three isomeric, (a) nitrotoluenes, (b) methoxy toluenes and ( c) methoxy acetanilides. Explain your choices in each case. |
Answer» Solution :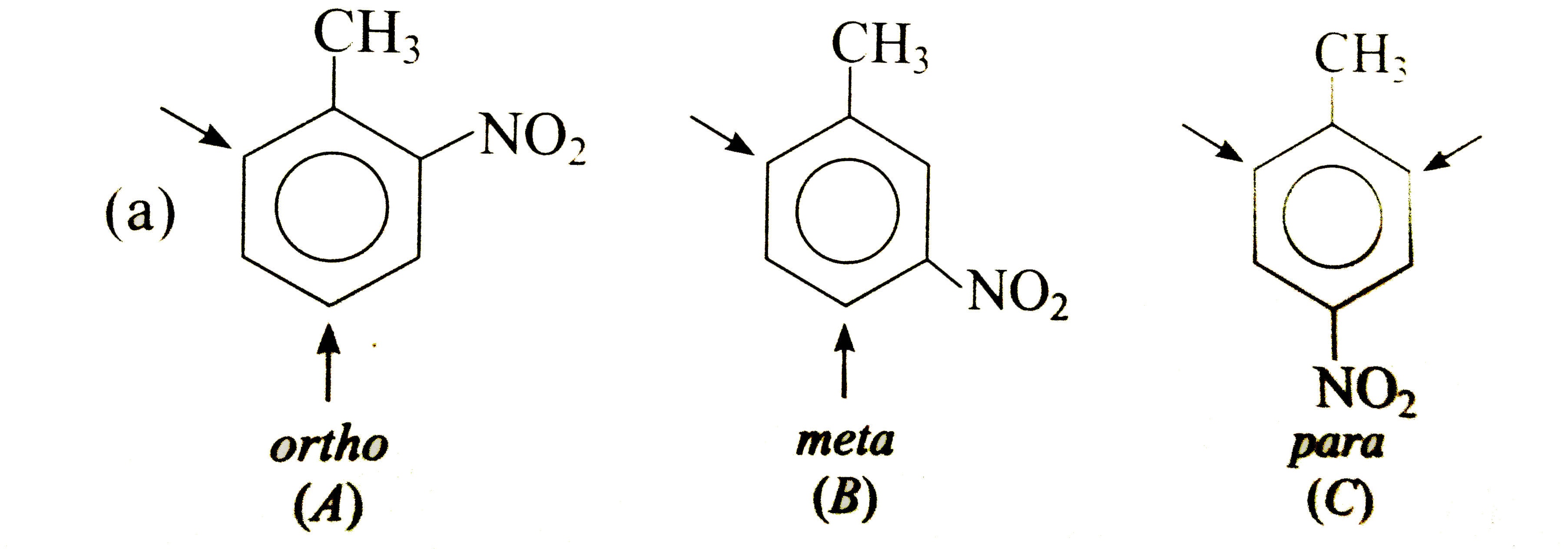 In (A), `CH_(3)` DIRECTS `E^(+)` to ORTHO and para-positions which are also meta to `NO_(2)`, i.e., substituents reinforce each other. In (B), the orientation is in opposition. The o-p-director controls the orientation, i.e., `E^(+)` enters para to `CH_(3)` and ortho to `NO_(2)`. In (C), both groups are reinforcing, i.e., ortho to `CH_(3)` and meta to `NO_(2)`. 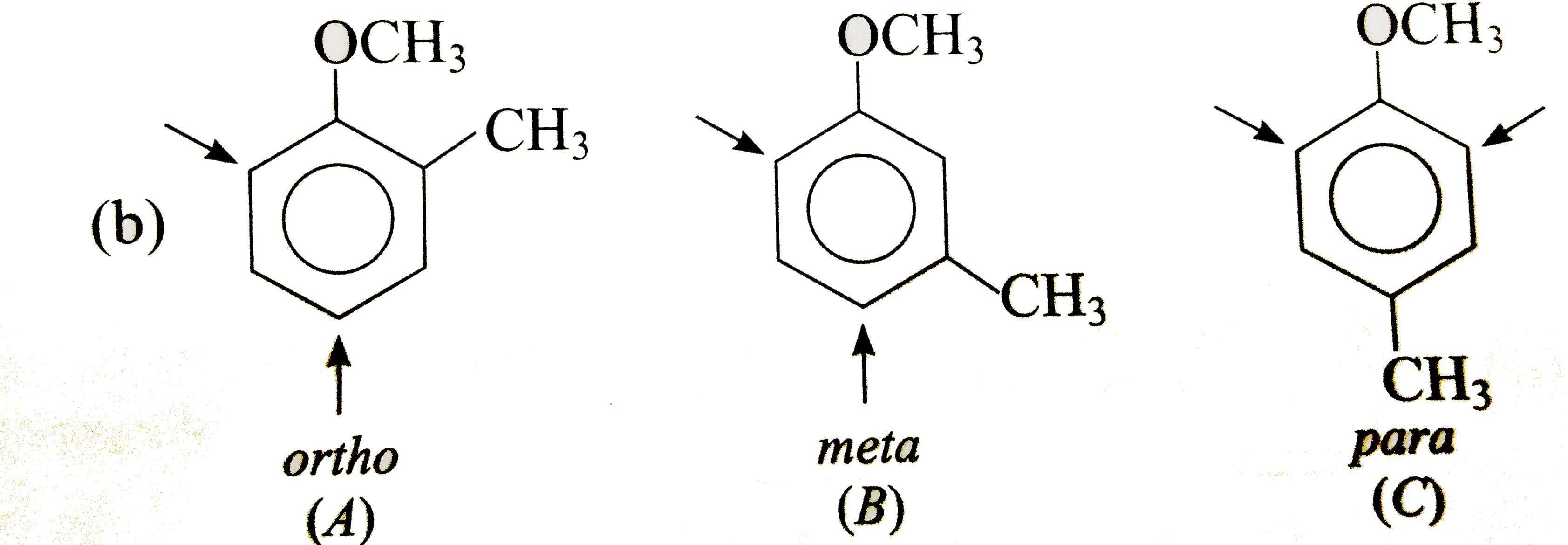 Both groups are o- p-directing. In (B), the substituents reinforce each other, very little substitution occurs between the groups due to steric HINDRANCE. In (A)and(C), the two groups ar in opposition. The more strongly activating `-OCH_(3)` controls the orientation. 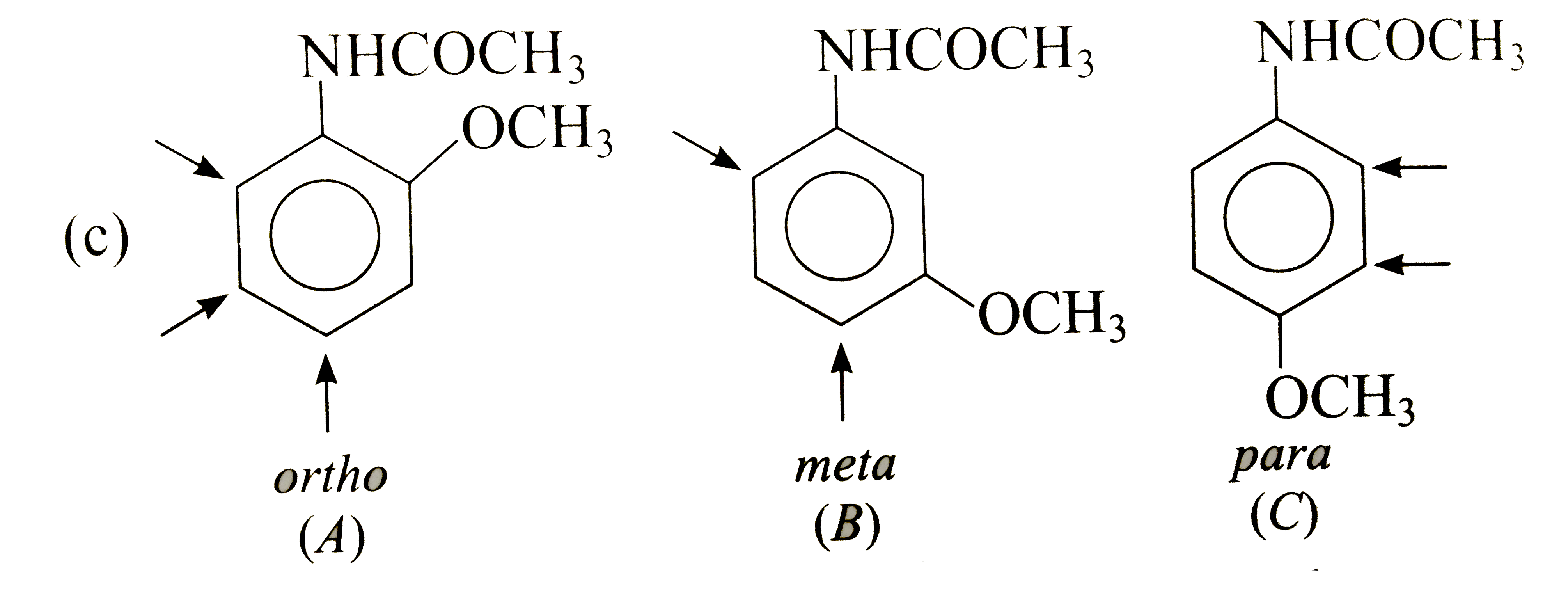 Both the substituents are o- p-directing and equally MODERATE STRONG activators. In (B), both the groups are reingorcing and a mixture of two isomers is obtained. In (A) and (C), two groups are in opposition. In (A), Substantial amounts of all isomers are obtained while in (C), substitution occurs ortho to both the groups. |
|
| 11. |
Show by a chemical reaction with water that Na_(2) Ois a basic oxide and Cl_(2)O_(7)is an acidic oxide. |
|
Answer» SOLUTION :`Na_(2)O` with water forms a strong base whereas `Cl_(2)O_(7)` forms strong acid. `Na_(2) O + H_(2) O to NaOH` `Cl_(2)O_(7) ` REACT with `H_(2)O` and forms strong acid `HClO_(4)`. `Cl_(2)O_(7) + H_(2)I to 2HClO_(4)` ` :. Cl_(2) O_(7)` is an acidic oxide. Their BASIC or acidic nature can be qualitatively tested with litmus paper. |
|
| 12. |
Showby achemicalreaction withwaterthat Na_(2) O isa basicoxideand CI_(2)O_(7) is anacidicoxide . |
|
Answer» Solution :`Na_(2)O` reactswithwater to fromsodiumhydroxidewhichturnsredlitumsblue, `{:(Na_(2)O + H_(2)O ,,to ,,2NaOH ),("Sod oxide ",,,,"Sod . Hydroxide "):}` Therefore`Na_(2)O` is a basicoxide In CONTRAST `C1_(2)O_(7)` reacts withwaterto formperchloric acidwhichturmsbluelitmus red `{:(Cl_(2) O_(7)+H_(2)O ,,to ,,2HClO_(4)),("Chlorine (VII) oxide ",,,,"Perchloric acid "):}` Therefore `Cl_(2) O_(7)` isanacidicoxide. |
|
| 13. |
Show aromatic structure having 1, 2 and 3 rings with suitable example. |
|
Answer» Solution :Benzene was considered as parent .AROMATIC. compound. Now the name is applied to all the ring system whether or not having benzene ring, possessing FOLLOWING characteristics. (a) Requirements for aromatic character in compound : (i) Compound must havr cyclic (ring) structure. (ii) Cyclic compound must have all atoms in one planer structure. (iii) Complete delocalisation of the `pi` electronsin the ring. (iv) Presence of `(4n+2)pi` electrons in the rinf where .n. is an inetger (n=0, 1, 2,...). This is often referred to as Huckel Rule. (b) Huckel.s rule for aromaticity : If the number of electrons in planar cyclic compound equals to (4n+2), then these compounds possess aromatic properties, (aromaticity) where n = number of cyclic STRUCTURES, (4n+2) = total delocalized electron of atoms present in aromatic ring). (c) Aromatic hydrocarbon compounds : (i) Benzene is aromatic in nature. Number of rings in benzene = n =1 NUmber of double bonds in benzene = 3 so, number of `pi2(p)` electrons = 6 `therefore` According to Huckel rule, if (4n+2) `pi` electrons are present in structure then it is considered as aromatic. So for benzene , n=1 and (4n+2) = 4(1) + 2 = 6, so benzen obeys Huckel rules and benzene is aromatic in nature and it possess aromatic characteristics.  (ii) Cyclopentadienyl anion aromatic in nature. According to Huckel rule, n = 1 and hence (4n+2) = 6 In cyclopentadienail anion two double BOND have `2 xx 2 = 4pi` + two lone `pi` electrons, so it is aromatic in nature.  (iii) Cycloheptatrienyl cation is aromatic in nature. Number of ring = n = 1 `therefore (4n+2) = 6`  In this compound three double bonds are present, so no. of `pi` electrons `= (3 xx 2) = 6`. So it follows Huckel rule, so cycloheptatriene, n = 1(4n+2)=6, cation is aromatic in nature. It possess positive charge `sp^(2)` carbon so it also possess planarity. (iv) Nepthalene is aromatic in nature.  Number of ring = n= 2 It having five double bonds so no. of `pi` electrons `= (5 xx 2) = 10` electrons Napthalene follows Huckel rules and so it is aromatic innature. (v) Anthrecene and Phenanathrene are aromatic compounds. 
|
|
| 14. |
Shifting of electrons of a multiple bond under the influence of reagent is called |
|
Answer» I-effect |
|
| 15. |
Sharp glass edges are heated for making them smooth (polishing of glass) which is due to its |
|
Answer» VISCOCITY |
|
| 16. |
Shape-selective catalysis is a reaction catalysed by:- |
|
Answer» Zeolites |
|
| 18. |
Shape of PCl_(3) ....... |
|
Answer» Trigonal pyramidal `OVERSET(..)(PCl_(3))` is `AB_(3)` Etype molecule DUE to non-bonding pair on P it has trigonal pyramidal shape. |
|
| 19. |
Shape of O_(2)F_(2) is similar to that of |
|
Answer» `C_(2) F_(2)` They , HOWEVER, differ in BOND anglesand bond lengths . |
|
| 20. |
Shape of ClF_(3) is |
|
Answer» PLANAR triangular |
|
| 21. |
Shape of the methyl carbonium ion is |
|
Answer» Planar |
|
| 22. |
Shape of methyl carbanion is |
|
Answer» Planar |
|
| 23. |
Shape of C_60 fullerene is ………. |
|
Answer» Tennisball |
|
| 24. |
Shape of an orbital is given by |
|
Answer» Principal quantum NUMBER |
|
| 25. |
Shape and hybridisation of IF_5 are................. |
|
Answer» TRIGONAL bipyramidal , `sp^3 d^2` |
|
| 26. |
Shape and hybridisation of IF_(5) are_______ |
|
Answer» Trigonal bipyramidal, `sp^(3)d^(2)` |
|
| 27. |
Shape and hybridisation of IF_(5) are |
|
Answer» TRIGONAL bipyramidal, `SP^(3)d^(2)` 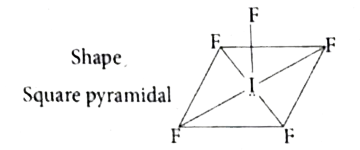
|
|
| 28. |
SF_(2), SF_(4) and SF_(6)have the hybridisations at sulphur atom respectively as |
|
Answer» `sp^(2), sp^(3), sp^(3), d^(2)` (c)+ Charge on polyatomic anion (a) For `SF_(2) , X= (1)/(2) (6 + 2 - 0 ) = 4 ` , Hybridisation = =`sp^(3)` For `SF_(2) , X = (1)/(2) (6 + 4 - 0 ) = 5 ` Hybridisation = ` sp^(3) d ` For ` SF_(6), X = (1)/(2) (6 + 6 - 0 ) = 6 ` Hybridisation = ` sp^(3) d^(2)` |
|
| 29. |
SF_(4) has _____shape. |
|
Answer» T-shape |
|
| 30. |
Sewage mostly constitutes |
|
Answer» Effluents |
|
| 31. |
Sewage containing organic waste should not be disposed in water bodies because it causes major water pollution. Fishes in such a polluted water die because of |
|
Answer» Large number of mosquitoes |
|
| 32. |
Sewage water is purified by |
|
Answer» MICROORGANISMS |
|
| 33. |
Sewage containing organic waste should be disposed in water bodies because it causes major water pollution . Fishes in such a polluted water die because of |
|
Answer» Large number of MOSQUITOES |
|
| 34. |
Several sodium compounds find use in industries. Which of the following compounds are used for textile industry ? |
|
Answer» `Na_(2)CO_(3)` NaOH is used in Soap industry, PAPER industry. |
|
| 35. |
Several sodium compounds find use in industries . Which of the following compounds are used for textile industry ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 36. |
Several blocks of magnesium are fixed to the bottom of a ship to |
|
Answer» KEEP AWAY the sharks |
|
| 37. |
Several blocks of magnesium are fixed into the bottom of ship to : |
|
Answer» PREVENT action of water and slat |
|
| 38. |
Settling of cement is which types of reaction ? |
|
Answer» EXOTHERMIC reaction |
|
| 39. |
Setting time for di calcium and tricalcium silicate are.......days respectively. |
|
Answer» 28 and 1 |
|
| 40. |
Setting of plaster of paris involves |
|
Answer» Dehydration |
|
| 41. |
Setting time can be reduced by addition of ...... in plaster of Paris. |
|
Answer» Borex |
|
| 42. |
Setting time can be increase by addition of ...... in plaster of Paris. |
|
Answer» NaCl |
|
| 43. |
How is plaster of paris prepared ? What is setting of plaster of paris ? |
|
Answer» OXIDATION with atmospheric `O_(2)` |
|
| 44. |
Set of elements with the following atomic numbers belong to the same group |
| Answer» Answer :B | |
| 45. |
{:("Set-I ","Set-II "),("A) Sodium benzoate ","1) Red hot iron tube "),("B) Phenol ","2) dil. HCI "),("C) Acetylene ","3) anhydrous" AICI_3),("D) Benzene sulphonicAcid","4) Zinc dust, "Detla),(,"5) Soda lime,"Delta):} Correct matching to get benzene is |
|
Answer» A-3, B-4, C-1, D-5 |
|
| 46. |
{:(,"Set-I",,"Set-II"),("(A)","10 vol H"_(2)"O"_(2),1.,"Perhydrol"),("(B)","20 vol H"_(2)"O"_(2),2.,5.358N),("(C)","30 vol H"_(2)"O"_(2),3.,1.785N),("(D)","100 vol H"_(2)"O"_(2),4.,3.30%):} The correct match is : |
|
Answer» A-4, B -3, C-2, D-1 |
|
| 47. |
Sesquioxide of lead is |
|
Answer» PBO |
|
| 48. |
Separation of two substances by crystallisation depends upon their differences in |
|
Answer» densities |
|
| 49. |
Separation of substance of a mixture over adsorbent coated on glass tube is ____ chromatography. |
| Answer» ANSWER :B | |
| 50. |
Separation of Lanthanides from their mixture is not easy because of |
|
Answer» SHIELDING effect |
|