Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-1. Acetylene is more reactive than ethylene towards electrophilic addition reactions Statement-2. Acetylene containstwo pi-bonds |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation for Statement-1 |
|
| 2. |
Statement 1 a substance which gets reduced can act as an oxidising agent Statement 2 In the reaction 3 CIO^(-)rarrCIO_(3)^(-)+2cI^(-),CI atom is oxidised as well as reduced |
|
Answer» STATEMENT 1 is TRUE statement 2 is true , statement 2 is a corrrect EXPLANATION for statement 1 |
|
| 3. |
statement-1 : A reaction with K_(P)=(1)/(1.005)atm^2 is expected to be spobtaneous at standard conditions. statement-2: Reactions with negative Delta^circ will be spontaneous at standard condition. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 4. |
statement-1 : A net reaction can occur only if a system is not a equilibrium . statement-2 : All reversible reactions occur to reach a state of equilibrium. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 5. |
STATEMENT-1: (A) is optically active and (B) is optically inactive. STATEMENT-2: (A) has a centre of symmetry. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 6. |
Statement -1. A compound is formed from its elements only if free energy of formation of the compound is positive . Statement -2. Many photochemical reactions have positive valeu for the free energy change. |
|
Answer» STATEMENT -1 is True, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION of Statement-1 |
|
| 7. |
statement-1 :A catalyst does not influence the values of equilibrium constant. statement-2 : Catalyst influence the rate of both forward and backward reactions equality. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 8. |
Statement 1 2CuCIrarrCuCI_(2)+Cu is a disprotionation reaction Statement 2 all transitoin metals show disproportioination reactions |
|
Answer» Statement 1 is true statement 2 is true , statement 2 is a corrrect explanation for statement 1 |
|
| 9. |
Statement-1. 2p orbitals do not have any spherical node. Statement.2 The number of nodes in p-orbitals is given by (n -2) where n is the principal quantum number |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 |
|
| 10. |
STATEMENT -1: 2,3-dimethyl butane is a meso compound. STATEMENT-2: It has plane of symmetry so it is sptically inactive due to internal compensation. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 11. |
Statement-1 : 1 mol of H_(2)SO_(4) is neutralised by 2 mol of NaOH, however, 1 equivalent of H_(2)SO_(4) is neutralised by 1 equivalent of NaOH. Statement-2: Equivalent mass of H_(2)SO_(4) is half of its moelcular mass, however, the equivalent mass of NaOH is equal to its moelcular mass. |
|
Answer» Statement-1 is TRUE, statement -2 is true, statement-2 is a correct explanation for statement-1. |
|
| 12. |
STATEMENT-1: 2-butanol is a chiral molecule. STATEMETN-2: 2-butanol have chiral carbon in it. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 13. |
Statement I: Salt solution is an aqueous solution. Statement II: If water is used as the solvent, the resultant solution is called an aqueous solution. |
|
Answer» Statements I and II are CORRECT but II is not the correct EXPLANATION of I. |
|
| 14. |
State with equation what happens when borax is heated on a platinum wire loop and to the results tranparent mass, a minute of CuOis added and the mixture is again heated first in oxidisingflame and then in the reducingflame of a Bunsen burner ? |
|
Answer» Solution :Whenborax is HEATED on a platinumwire loop. Atransparent glassy beadis heated with `CuO` in the oxidisingflame,it impartsblue colour to the beaddue tothe formationof cupricmetaborate `CuO + B_(2)O_(3) OVERSET("Oxidising flame")rarr underset("CUPRIC metaborate(blue)")(Cu (BO_(2))_(2))` However, when cupric metaborate is heatedin the reducing flame of the Bunsen burner, the blue cupric metaborate isreducedby carbonpresent in the flamefirst to colourless currous metaborate and the then to metal andproduces red scales. `{:(underset(("blue"))underset("Cupric metaborate")(2Cu(BO_(2))_(2))+Crarrunderset(("colourless"))underset("Cuprous metaborate")(2CuBO_(2))+B_(2)O_(3)+CO),(2CuBO_(2) + C rarr underset(("red"))underset("COPPER metal")(Cu)+B_(2)O_(3)+CO):}` |
|
| 15. |
State zeroth low of thermodynamics. |
| Answer» SOLUTION :If twosystem at different temperatures are separately in THERMAL equilibrium with a third one, then they tend to be in thermal equilibrium with themselves'. | |
| 16. |
State with equations what happens when borax is heated on a plantium wire loop and then to the resulting transparent mass a minute amount of CuO is added and the mixture is again heated first in an oxidising flame and then in the reducing flame of a Bunsen Burner . |
|
Answer» Solution :When borax is heated on a platinum wire LOOP a transparent glassy bead is formed `Na_(2)B_(4)O_(7).10H_(2)O underset(-10H_(2)O) OVERSET("heat")rarrNa_(2)B_(4)O_(7)overset("heat")rarr underset("Transparent glassy bead")ubrace("Sod.metaborate Boric anhydride")2NaBO_(2)+B_(2)O_(3)` When this transparent glassy bead is heated with CuO in the oxidising flame it IMPARTS blue colour to the bead due to the FORMATION of cupric metaborate `CuO+B_(2)O_(3)underset("flame") overset("Oxidising") (rarr)underset("Cupric metaborate (blue)")(Cu(BO_(2))_(2))` However whencupric metaborate is heated in the reducing flame of the Bunsen burner the blue cupric metaborate is reduced by carbon present in the flame first to colourless cuprous metaborate and then to copper METAL which produces a red colour. `underset(("Blue"))underset("Cupric metaborate")(2Cu(BO_(2))_(2))+Crarr underset(("Colourless"))underset("Cuprous metaborate")(2CuBO_(2))+B_(2)O_(3)+CO` `2CuNO_(2)Crarrunderset("Red")underset("Copper metal") (Cu)+B_(2)O_(3)+CO` |
|
| 17. |
State, whether the following statements is True or False: The elements with higher electron affinity have higher ionization potential also. |
|
Answer» |
|
| 18. |
State, whether the following statements is True or False: An anion is larger than a cation if they are iso-electronic |
|
Answer» |
|
| 19. |
State whether the existence of equilibrium is possible in our lungs or not. Give reason. |
| Answer» Solution :The state of equilibrium exist in our lungs because, the three SPECIES NAMELY haemoglobin, OXYGEN (REACTANTS) and oxyhaemoglobin (product) are said to co-exist in our lungs. | |
| 20. |
State whether the following compounds are inorganic or organic : (i) caustic soda (ii) sugar (iii) nitric acid (iv) blue vitriol (v) vegetable ghee(vi) mustard oil (vii) baking soda. |
|
Answer» SOLUTION :(i) CAUSTIC soda (NaOH) : inorganic (ii) Sugar (`C_12H_(22)O_(11)`) : organic (III) Nitric acid (`HNO_3`) : inorganic (iv) Blue VITRIOL (`CuSO_4`) : inorganic (V) Vegetable ghee : organic (vi) Mustard oil: organic (vii) Baking soda (`NaHCO_3`) : inorganic. |
|
| 21. |
State water pollutant from followings. |
|
Answer» MICROORGANISMS |
|
| 22. |
State unit of velocity ? |
|
Answer» `m^(2)` |
|
| 23. |
State tpe order of bond dissociation enthalpy in O_(2), O_(2)^(+), O_(2)^(-) and O_(2)^(2-). |
|
Answer» SOLUTION :As the bond ORDER is greater the stability and dissociation ENTHALPY is greater. `O_(2)^(2-) lt O_(2)^(-) lt O_(2) lt O_(2)^(+)` `RARR`dissociation enthalpy INCREASING `rarr` 
|
|
| 24. |
StateTrue (T ) or False(F ) forfollowingstatements |
|
Answer» As the FREQUENCYIS morewavelengthis LESS |
|
| 25. |
State three conditions which govern the combination of atomic arbitals to from molecular orbitals. |
|
Answer» |
|
| 27. |
Statethe valueof n,l, m_(l) andm_(s) for 4f. |
|
Answer» SOLUTION :`n=4,l=3` `m_(1) ` has 7 VALUE: =-3 -2,-1 ,0+ 1+2 +3 `m_(1)` has 7valuehas + `(1)/(2) ` and `-(1)/(2)` |
|
| 28. |
State the various statements of second law of thermodynamics. |
|
Answer» Solution :(i)Entropy statement: Whenever a SPONTANEOUS process TAKES place, it is accompanied by an increase in the total entropy of the universe" (II)Kelvin-Planck statement: It is impossible to take heat from a hotter reservoir and convert it completely into WORK by a cyclic process without transferring a part of heat to a cooler reservoir (iii)Efficiency statement: Even an ideal, frictionless engine cannot convert 100% of its input heat into work. Efficiency =`(T_1-T_2)/T_1` % Efficiency =`[(T_1-T_2)/T_1]xx100` % Efficiency =`["Output"/"Input"]xx100` % Efficiency < 100% (iv)Clausius statement: Heat flows spontaneously from hot objects to cold objects and to get it FLOW in the opposite direction, we have to spend some work. |
|
| 29. |
Statethe usesof linespectrumor atomicspectra. |
| Answer» SOLUTION :lincspectrumare USEDTO DETECT element.Eachelement HASA uniquelineemissionspectrumThecharacteristiclinesin atomicspectrumcanbe usedin chemicalanalysisto identifyunknownatoms. | |
| 30. |
State the unit of temperature with explanation. |
|
Answer» |
|
| 31. |
State the unit of concentration. |
|
Answer»
|
|
| 32. |
State the unit of molar entropy. |
|
Answer» JOULE/ KELVIN |
|
| 33. |
State the type of hybrid orbitals associated with (i) P in PCl_(5) and (ii) S in SF_(6) . |
|
Answer» <P> SOLUTION :(i) `SP^(3) d `of P in `PCl_(5)(II) sp^(3) d^(2)` of S in `SF_(6)` . |
|
| 34. |
State the total increase in the oxidation number of central atom of reducing agent in following reaction. Reaction : N_(2)H_(4)+Cu(OH)_(2)toN_(2)+Cu |
|
Answer» 2 |
|
| 35. |
State the trends in the variation of electronegativity in group and periods. |
Answer» Solution :Variation of electronegativity in a period: The Variation of Electronegativity along I period electronegativity increases across a period from left to RIGHT. Since the ATOMIC radius DECREASES in a period, the ATTRACTION between the valence electron and the nucleus increases. HENCE the tendency to attract shared pair of electrons increases. Therefore, electronegativity increases in a period.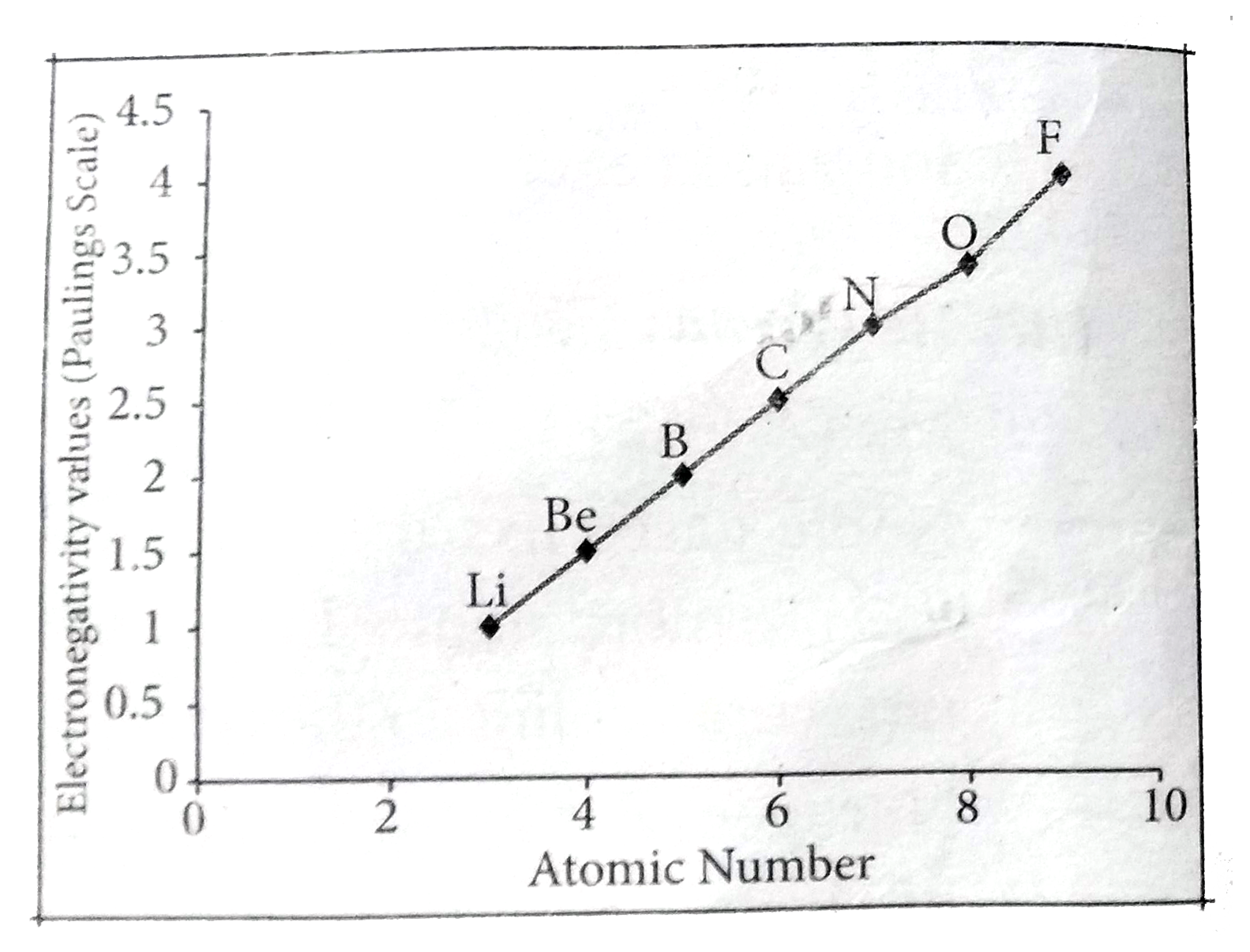 Variation of electronegativity in a group: The electronegativity decreases down a group. As we move down a group, the atomic radius increases and the nuclear attractive force on the valence electron decreases. Hence electronegativity decreases in a group. 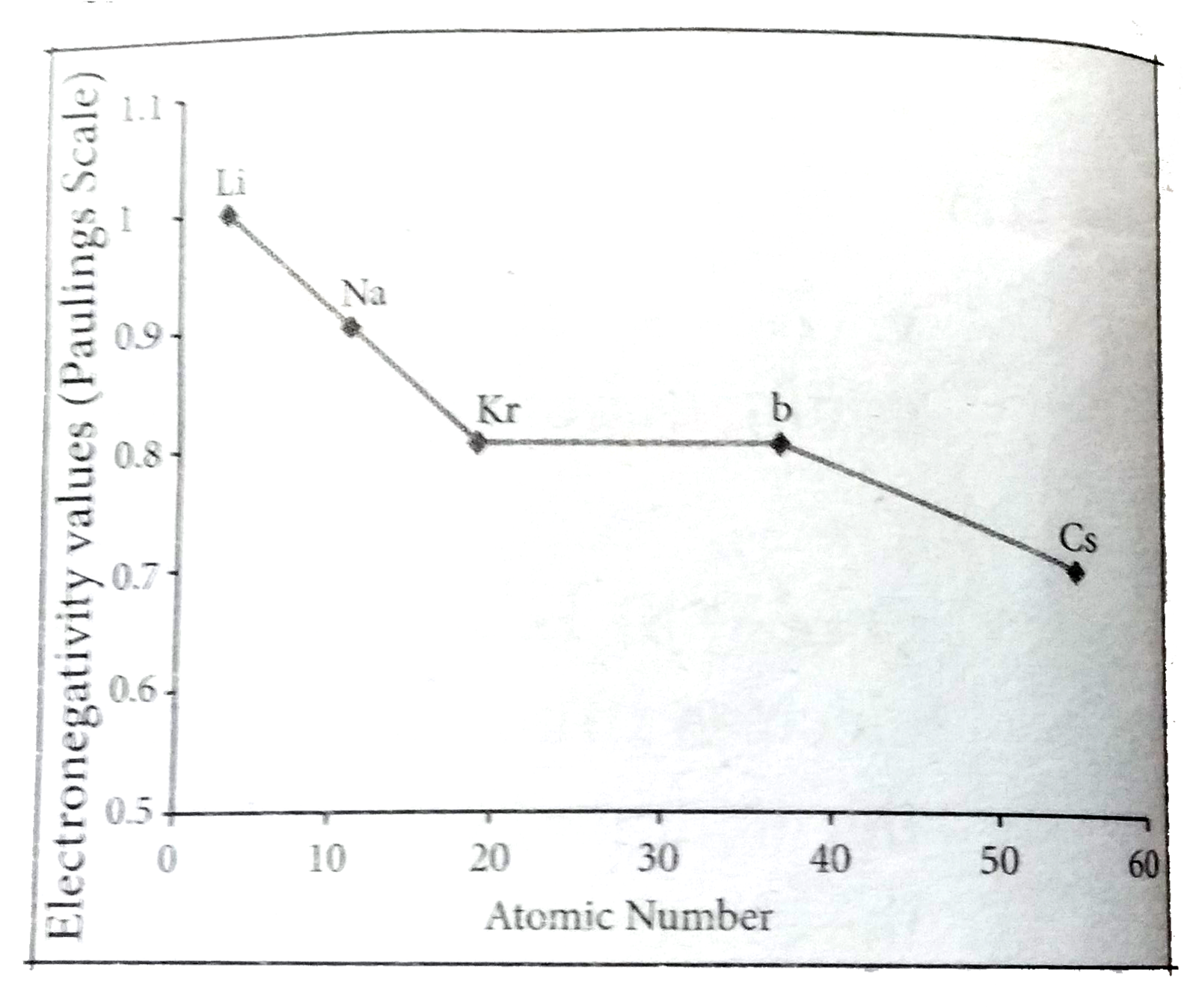
|
|
| 36. |
State the term "Isotonic solution |
| Answer» Solution :(i) Two solutions having same OSMOTIC pressure at a given temperature are called isotonic solutions. (II) When such solutions are separated by a semipermeable membrane, solvent flow between one to the other on either DIRECTION is same. i.e., the NET solvent flow between two isotonic solutions is zero. | |
| 37. |
State the thermodynamicconditions of spontaneous occurence of a process. |
|
Answer» Solution :For spontaneous occurrence,`DeltaG` of the processmustbe `( lt 0 , i.e., -ve`..This can be so under the following conditions `:` (i) `DeltaH ` is negative and`DeltaS` is POSITIVE( at any temperature). (ii) If `DeltaH` and`DeltaS` both are positive, then T should be so HIGH that`TDELTA Sgt Delta H`. (iii) If both `Delta H `and`DeltaS` are negative, then T should be solow that `T Delta ltDeltaH ` |
|
| 38. |
State the structure and shape of SF_(4) and CIF_(3). |
Answer» SOLUTION :`SF_(4)` See SAW and shape of `CIF_(3)` is T shaped. 
|
|
| 40. |
Statethe speciesin whichelectronprotonandneutron are10 ,8 , 8respectively ? |
|
Answer» `._(8)^(12) O^(2-)` |
|
| 41. |
Statethe second law of thermodynamics. |
| Answer» SOLUTION :NATURALLY occurringprocesses are accompanied by increase in entropy. Hence, entropyofthe UNIVERSE is CONTINUOUSLY increasing. | |
| 42. |
State the second law ofthermodynamics. |
| Answer» SOLUTION :"The net entropy of the universe is continuously increasing" (or) " TOTAL entropy of the system and the surroundings increases in any spontaneous process i.e., `Delta S_("system") + Delta S_("surroundings") gt 0`, for any spontaneous process. | |
| 43. |
State the relative stabiliry of N_(2) , N_(2)^(+), N_(2)^(-) and N_(2)^(2+) . |
|
Answer» Solution :stability `PROP` BOND ORDER `N_(2) gt N_(2)^(+) = N_(2)^(2+) = N_(2)^(-)` 
|
|
| 44. |
State the requirements to form hydrogen bond. |
| Answer» Solution :(i) The SIZE of negative atom must be smaller as possible. (II) The electronegativity of atom COMBINE with HYDROGEN should be more as possible. | |
| 46. |
State the product of reaction : 2SO_(2(g)) + O_(2(g)) + 2H_2 O_((1)) to |
|
Answer» `2H_2 SO_3` |
|
| 47. |
State the number of significant figures in each of the following: (i) 136.7 m (ii) 105.67 kg (iii) 3.4 xx 10^(6)s (iv) 0.0078 km (v) 0.00650 xx 10^(6) m |
|
Answer» |
|
| 48. |
State the number of significant figures in each of the following numbers : (i) 2.653 xx 10^4 ,(ii) 0.00368 (iii) 65.3 (iv) 0.368 (v) 0.0300. |
|
Answer» Solution :(i) `2.653 XX 10^4` has four significant figures because the EXPONENTIAL term does not ADD to the number of significant figures. (ii) 0.00368 has three significant figures because zeros placed left to the first non-zero DIGIT are insignificant. (iii) 65.3 has three significant figures. (iv) 0.368 has three significant figures. (v) 0.0300 has three significant figures because zeros placed after a non-zero digit are significant. |
|
| 49. |
State the number of significant figures in each of the following numbers : (i) 3.56 (ii) 35.6 (iii) 0.356 (iv) 0.0356. |
|
Answer» Solution :(i) 3.56 has three SIGNIFICANT figures. (II) 35.6 also has three significant figures because decimal POINT does not determine the NUMBER of significant figures. (iii) 0.356 has three significant figures. (iv) 0.0356 also has three significant figures because the zero placed LEFT to first non-zero digit is not significant. |
|