Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The alkali metal which can emit its outermost electron under the influence of even candle light is |
|
Answer» Li |
|
| 2. |
The alkali metal that can react directly with carbon to form ionic carbide is |
|
Answer» Na |
|
| 3. |
The alkali metal that does not participate in the reaction MI + I_2 to MI_3 is |
| Answer» Answer :A | |
| 4. |
The alkali halide which is soluble in pyridine is |
|
Answer» NaCl |
|
| 5. |
The alkali metal that can react differently in many reactions is |
| Answer» ANSWER :D | |
| 6. |
The alicyclic compound is |
|
Answer» Cyclohexane |
|
| 7. |
The air oxidation of which of the following organic compound produces hydrogen peroxide ? |
|
Answer» 2-Ethylanthracene |
|
| 8. |
The air contains 78% and 22% O_(2) by volume. The volume occupied by 40g air at 20^(@)C and 745 mm Hg pressure approximately: |
|
Answer» 34L |
|
| 9. |
The aggregation of social, biological, physical and chemical factors existing around us is called .......... |
|
Answer» ENVIRONMENT |
|
| 10. |
The additions of sodium acetate to 0.1 M acetic acid will cause : |
|
Answer» increase in its pH VALUE ` therefore [H^(+)] downarrow rArr pH uparrow ` |
|
| 11. |
The addition of NaCl to AgCI decreases the solubility of AgCl, because |
|
Answer» `K_(sp) of `AgCl DECREASES |
|
| 12. |
The addition of oxygen gas to reaction mixture of chlorine and methane (photochemical chlorination) |
|
Answer» accelerates the REACTION |
|
| 13. |
The addition of Na_(2)CO_(3), to the aqueous solution of an oxide produces CO_(2). This reaction indicates that |
|
Answer» OXIDE is BASIC |
|
| 14. |
The addition of HCl to an alkene proceeds in two steps. The first step is the attack of H^(+) ion to portion which can be shown as…….. |
|
Answer»
 (A) Not correct because in this `H^(+)` attack on `pi` bond (C ) Wrong, because in this `H^(+)` ATTACH with carbon and `pi` bond electron shift on carbon |
|
| 15. |
The addition of HCl to an alkene proceeds in two steps. The first step is the attack of H^(+) ion to gt C = C lt portion which can be shown as |
|
Answer»

|
|
| 16. |
The addition of HBr to an unsymmetrical alkene takes place in accordance with …….. Rule. |
|
Answer» |
|
| 17. |
The addition of HBr to propene in the presence of peroxides follows ..........rule. |
|
Answer» |
|
| 18. |
The addition of HBr is easiest with |
|
Answer» `Cl_(2)C=CHCl` |
|
| 19. |
The addition of dilute HCI acid to a solid nitric in cold yields a transient, pale-blue liquid is obtaine due to presence to presence of the following compound(s): |
|
Answer» FREE NITROUS acid |
|
| 20. |
The addition of carbonyl compound to HCN is an example of |
|
Answer» NUCLEOPHILIC substitution |
|
| 21. |
The addition of a catalyst during a chemical reaction alters which of the following quantities? |
|
Answer» INTERNAL energy |
|
| 22. |
The acurate value of pH is measure by which instrument ? |
|
Answer» Ameter |
|
| 23. |
The actual product of 4.327 and 2.8 is 12.1156. The correctly reported answer will be |
|
Answer» 12 |
|
| 25. |
The active mass of 7.0 g of nitrojan in a 2.0 L container would be |
|
Answer» 0.25 |
|
| 26. |
The active mass of 5.6 litres of O_2 at STP is |
|
Answer» 5.6/22.4 |
|
| 27. |
The active constituent of bleaching powder is..... |
|
Answer» `CA(O Cl)_(2)` |
|
| 28. |
The acids which do not contain a -COOH group are |
|
Answer» Propionic acid 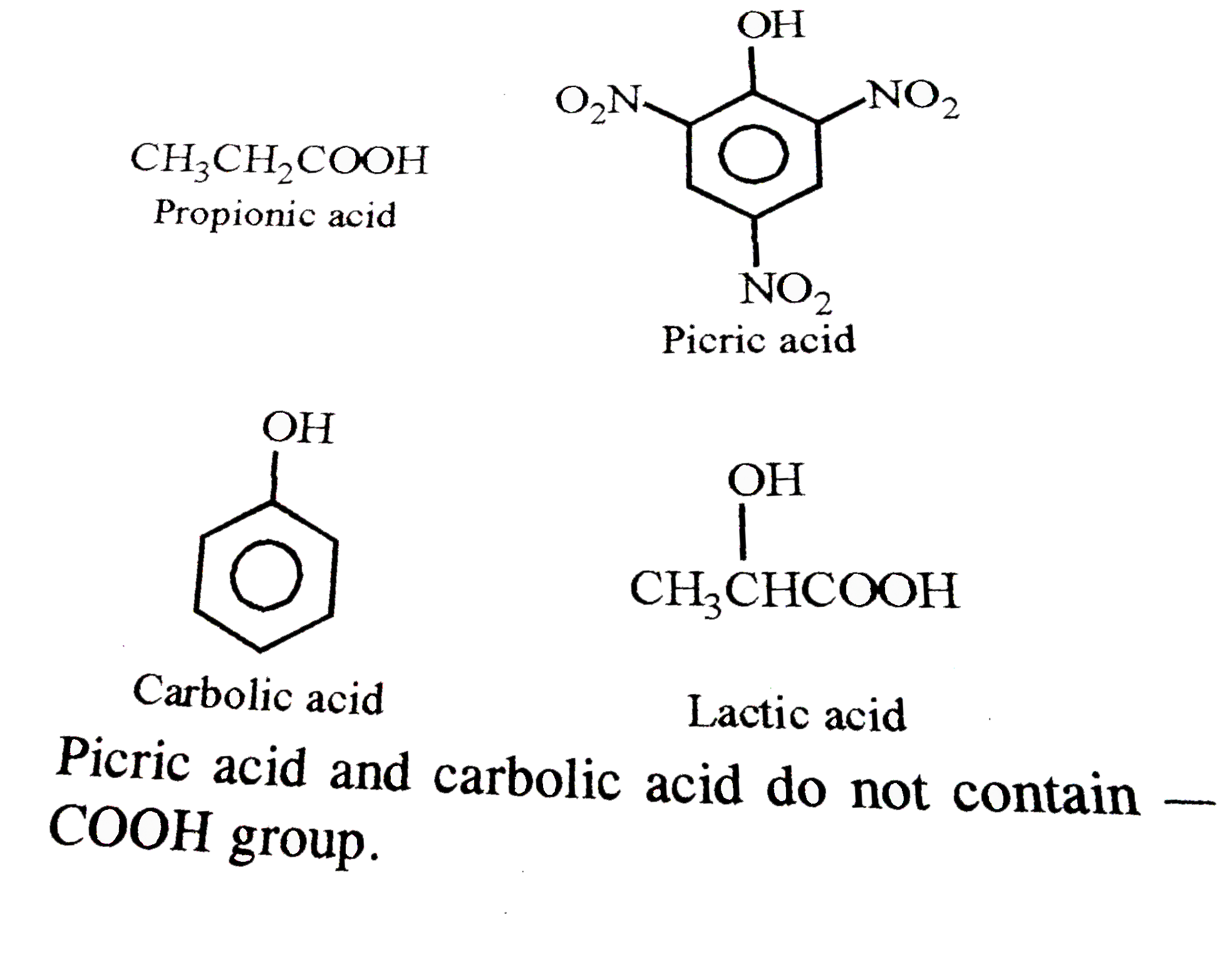
|
|
| 29. |
The acids present in acid rain are ______ |
|
Answer» Peroxyacetylnitrate |
|
| 30. |
The acids present in acid rain are ………. |
|
Answer» Peroxyacetylnitrate `CO_2 + H_2 O to H_2 CO_3` The oxides of NITROGEN undergo oxidation REACTION followed by reaction with water VAPOURS to form nitric acid. `NO + O_2 to H_2 CO_3` `2NO_2 + H_2 O to HNO_3 + HNO_2` The oxidation of `SO_2 and SO_3` occurs in the PRESENCE of dust particles or metal ions. The `SO_3` then reacts with water vapours to form `H_2 SO_4` `2SO_2 + O_2 underset("or metal ions")overset("Dust particles")to 2SO_3` `SO_3 + H_2 O to H_2 SO_4` |
|
| 31. |
The acidity of diprotic acids in aqueous solutions increases in the order |
|
Answer» `H_(2)S lt H_(2)Se lt H_(2)Te ` |
|
| 32. |
The acidic nature of hydrogens in acetylene cannot be explained by the reaction with |
|
Answer» SODIUM metal |
|
| 33. |
The acidic nature of hydrogens in acetylene can be explained by the reaction with |
|
Answer» Sodium metal |
|
| 34. |
The acidic character of three types of hydrocarbons follows the order alkanes gt alkenes gt alkynes. |
|
Answer» |
|
| 35. |
The acid which does not form an anhydride when treated with P_(4)O_(10) is |
| Answer» Solution :Formic acid | |
| 36. |
The acid used for etching the glass is |
|
Answer» SULPHURIC acid |
|
| 37. |
The acid S obtained through the following sequence of reactions is : C_(2)H_(5)Br overset(Alc. KOH)rarr P underset(C Cl_(4))overset(Br_(2))rarr Q underset(("excess"))overset(KCN)rarr R overset(H_(3)O^(+))rarr S |
|
Answer» <P>Succinic acid |
|
| 38. |
The acid showing salt like character in aqueous solution is |
|
Answer» Acetic acid `underset(alpha-"Amino acid")underset(NH_(2))underset(|)(R - CH -) COOH rarr underset("internal salt STRUCTURE")underset("Zwitter ion or")underset(+NH_(3))underset(|)(R - CH -) COO^(-)` |
|
| 39. |
The acid produced (A) in the sequence given below is {:(C_(2)H_(5)I underset(KOH)overset("Alcoholic")rarr X underset(C Cl_(4))overset(Br_(2)) rarr Y),(""darrKCN),(""A underset(H_(3)O^(+))larrZ):} |
|
Answer» Succinic acid |
|
| 40. |
The acid not suitable for the preparation of H_(2) by action with metals is |
|
Answer» HCl |
|
| 41. |
The acid ionization hydrolysis constant of Zn^(2+)is 1.0xx 10^(-9)which of the following statement are incorrect ? |
|
Answer» The `[H_3O^(+) ]`ion concentration in 0.001 M `Zn Cl_2` solution is `1.0 xx 10^(4) ` ` pH = 7 - ( P^(K_(b_2)))/( 2) - (1)/(2) log C= 7-(5)/(2) - (1)/(2) log 10 ^(-3)= 6` ` THEREFORE [H^(+) ] = 10 ^(-6) ` |
|
| 42. |
The acid catalysed hydrolysis products in the following reactions are - |
|
Answer» `{:("CHOCHO"),("|" &" |"),("CHOCHO"):}` |
|
| 43. |
The acid catalysed hydration follows following order (CH_3)_2C=CH_2 gt CH_3CH=CH_2 gt CH_2=CH_2 Explain this order of reactivity. |
|
Answer» `3^(ul@)` carbocation > `2^(ul@)` carbocation > `1^(ul@)` carbocation (stability order ) |
|
| 44. |
The accumulation of gases on surface of solids is called as |
| Answer» Solution :Occlusion | |
| 45. |
The acceptable level of carbon monoxide gas(CO) in the atmosphere in ppm level is |
|
Answer» 250 |
|
| 46. |
The absrobtion of hydroen by palladium is called: |
|
Answer» Hydrogenation |
|
| 47. |
The abundance of hydrogen gas in earth's atmosphere is lesser due to |
|
Answer» Lesser no. of hydrides present in earth's crust |
|
| 48. |
The abundance of alkaline earathmetails in the earth crust is in the order of |
|
Answer» `Ca gt Mg gt BA gt Sr gt Be` |
|





