Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The amount of energy released when an electron is added to an isolated gaseous atom to produce a monovalent anion is called electron affinity or first electron affinity or electron gain enthalpy. The first electron is given a negative sign as the addition of an electron to a neutral atom is an exoergic process. The addition of electron to A^(-1) requires energy to overcome the force of repulsion. Thus, the second electron affinity is an endoergic process. The magnitude of electron affinity depends on a number of factors such as (i) atomic size (ii) effective nuclear charge (iii) screening effect (iv) half and fully filled orbitals and (v) shape of orbital. In general, electron affinity increases as the atomic radii decrease in a period. However, there are exceptions when the atoms have stable configurations. In a group, electron affinity decreases as the size increases. However, the members of 3rd period have somewhat higher values than the members in the 2nd period of the same subgroups. Which one of the following arrangements represents the correct order of electron gain enthalpy (with negative sign) of the given atomic species ? |
|
Answer» `F LT CL lt O lt S` |
|
| 2. |
The amount of energy released when an electron is added to an isolated gaseous atom to produce a monovalent anion is called electron affinity or first electron affinity or electron gain enthalpy. The first electron is given a negative sign as the addition of an electron to a neutral atom is an exoergic process. The addition of electron to A^(-1) requires energy to overcome the force of repulsion. Thus, the second electron affinity is an endoergic process. The magnitude of electron affinity depends on a number of factors such as (i) atomic size (ii) effective nuclear charge (iii) screening effect (iv) half and fully filled orbitals and (v) shape of orbital. In general, electron affinity increases as the atomic radii decrease in a period. However, there are exceptions when the atoms have stable configurations. In a group, electron affinity decreases as the size increases. However, the members of 3rd period have somewhat higher values than the members in the 2nd period of the same subgroups. Which of the following processes is endoergic in nature ? |
|
Answer» `O^(-) + BARE to O^(2-)` |
|
| 3. |
The amount of heat released, when 20 ml of 0.5 M NaOH is mixed with 100 mL of 0.1M HCl, is x kJ. The heat of neutralisation is |
|
Answer» `-100 X` |
|
| 4. |
The amount of electrolytes required to coagulate a given amount of AgI colloidal solution (-ve charge) will be in the order ___________. |
|
Answer» `NaNO_3gtAl(NO_3)_3gtBa(NO_3)_2` |
|
| 5. |
The amount of dissolved oxygen required for oxidation of all the organic material present in liquid waste is known as ........ |
|
Answer» BOD |
|
| 6. |
The amount of a caustic soda required for complete neutralisation of 100 mL 0.1 N HCl is : |
|
Answer» 4.0 G |
|
| 7. |
The amont of water produced by the combustion of 16 g of methane is |
| Answer» Answer :B | |
| 8. |
The amorphous form of silica is |
|
Answer» KIESELGUHR |
|
| 9. |
The ammonia obtain from 0.50g organic compound is absorb in 50mL 0.5M, H_(2)SO_(4). In titration of excess H_(2)SO_(4), 60mL, 0.5M NaOH is used. Calculate the % of nitrogen |
|
Answer» |
|
| 10. |
The ammonia evolved from the treatment of 0.30 g of an organic compound for the estimation of nitrogen was passed in 100 mL of 0.1 M sulphuric acid. The excess of acid required 20 mL of 0.5 M sodium hydroxide solution for complete neutralization. The organic compound is |
|
Answer» acetamide = v mL of 0.1 `M H_(2)SO_(4)` Applying molarity equation `n_(a)M_(a)V_(a) = n_(b)M_(b)V_(b)`, we have, `2 XX 0.1 xx v = 1 xx 0.5 xx 20` or v = 50 mL `:.` Vol. of acid used = 100 - 50 = 50 mL of `0.1 M H_(2)SO_(4)` `%N = (1.4 xx n_(1)M_(a)V_(a))/("wt. of substance taken") = (1.4 xx 2 xx 0.1 xx 50)/(0.3)` `= 46.6` % N in urea `(NH_(2)CONH_(2)) = (28//60) xx 100 = 46.6%`, in acetamide `(CH_(3)CONH_(2)) = (14//59) xx 100 = 23.72%`, in benzamide `(C_(6)H_(5)CONH_(2)) = (14//21) xx 100 = 11.57%` and in thiourea `(NH_(2)CSNH_(2)) = (28//76) xx 100 = 36.84%` Thus, OPTION (c) is correct. |
|
| 11. |
The ammonia evolved from the treatment of 0.30 g of an organic compound for the estimation of nitrogen was passed in 100 mL of 0.1 M sulphuric acid. The excess of acid required 20 mL of 0.5 M sodium hydroxide solution for complete neutralization. The organic compound is : |
|
Answer» acetamide `(M_(1)V_(1))/(1) ("excess" H_(2)SO_(4))=(M_(2)V_(2))/(2)(KOH)` `(0.1xxV_(1))/(1)=(0.5xx20)/(2)` `V_(1)=50 mL` Volume of sulphuric acid USED up to absorb `NH_(3)=50 mL` `% N=(2.8xxMV)/(W)=(2.8xx0.1xx50)/(0.3)=46.6` Thus, the organic compound will be urea, which has 46.6% nitrogen. |
|
| 12. |
The ammonia evolved from 0.21 g of an organic compound by Kjeldahl method neutralised 15 ml of N/20 sulphuric acid solution. Calculate the percentage of nitrogen. |
|
Answer» Solution :`"weight of organic compound = 0.21 G"` `"Normality of ACID "=(N)/(20)` Volume of standard acid neutralised by ammonia = 15 ML 1000 ml of 1N ammonia contains = 14 g of nitrogen 15 ml of ammonia of normality `(N)/(20)" conains nitrogen "=(14xx15xx1)/(1000xx20)` 0.21 g of compound contains nitrogen `=(14xx15)/(1000xx20)` 100 g of compound contains nitrogen `=(14xx15xx100)/(1000xx20xx0.21)=5g` `therefore"Percentage of nitrogen = 5"` |
|
| 13. |
The ammonia evolved form 0.20 g of an organic compound by kjeldahl method neutralised 15ml of N // 20 Sulphuric acid solution. Calculate the percentage of Nitrogen. |
|
Answer» Solution :WEIGHT of organic compound = 0.20 g VOLUME of sulphuric acid taken= 15 ml `"Strength of sulphuric acid taken" = (N)/(20)=0.05 N` `"Percentage of NITROGEN"=(14 xx NV)/(1000 xx 10) xx 100` `=(14 xx 0.05 xx 15)/(1000 xx 0.20) xx 100=(1050)/(200)=5.25%` `IMPLIES`% of nitrogen=5.25% |
|
| 14. |
The ammonia evolved by the treatment of 0.30 g of an organic compound for the estimation of nitrogen was passed in 100 ml of 0.1 M sulphuric acid. The excess of acid required 20 mL of 0.5 M solution hydroxide solution for complete neutralization. The organic compound is |
|
Answer» acetamide Acid taken initially `= 100xx0.1xx2` m.e.q. Acid used for neutralisation `(100xx0.1xx2)-(20xx0.5xx1)` `20-10=10` m.eq. `rArr NH_(3)` liberated = 10 m.eq. `% N = (NH_(3)" liberated (m.eq)")/("wt.of ORG. compound")xx1.4` `=(10xx1.4)/(0.3)=46.66% rArr` Urea has 46.66% N. |
|
| 15. |
The alternate solvent used instead of tetrachloroethylene in dry cleaning is............* |
| Answer» SOLUTION :LIQUEFIED `CO_2` | |
| 16. |
The allotropic form of sulphur for which the standard enthalpy of formation is taken as zero is "…..................." |
| Answer» SOLUTION :RHOMBIC SULPHUR | |
| 17. |
The allotropic forms of carbon showing conductivity are |
|
Answer» Diamond |
|
| 18. |
The allotrope of Carbon not used in the making of electrodes is |
|
Answer» GAS Carbon |
|
| 19. |
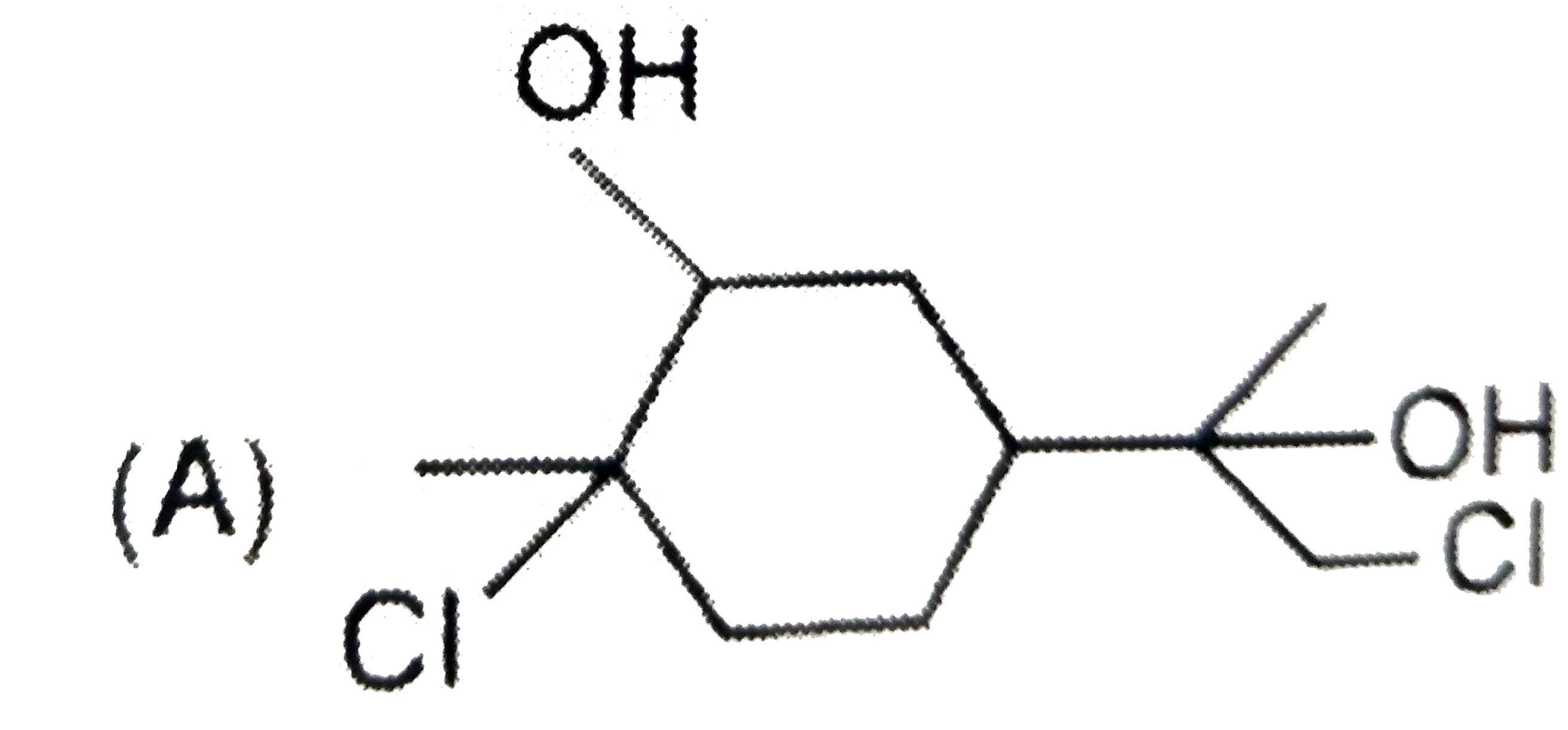
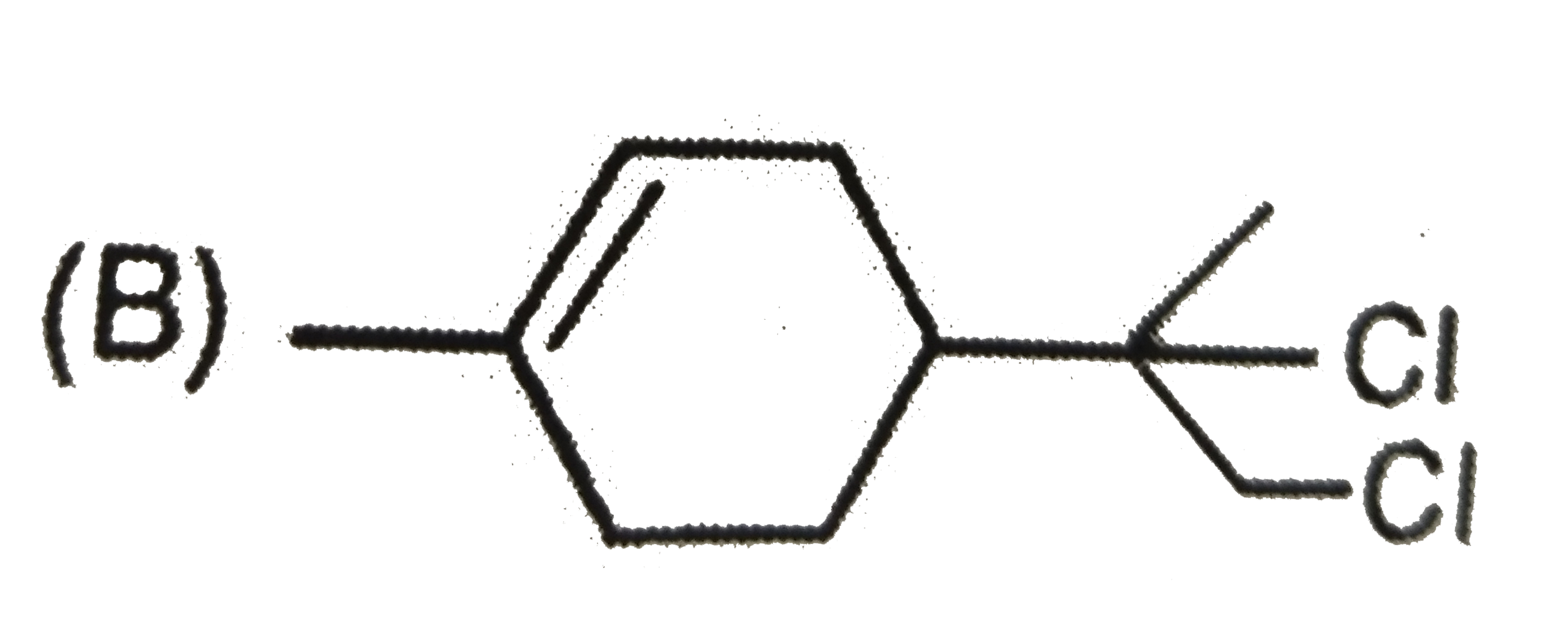
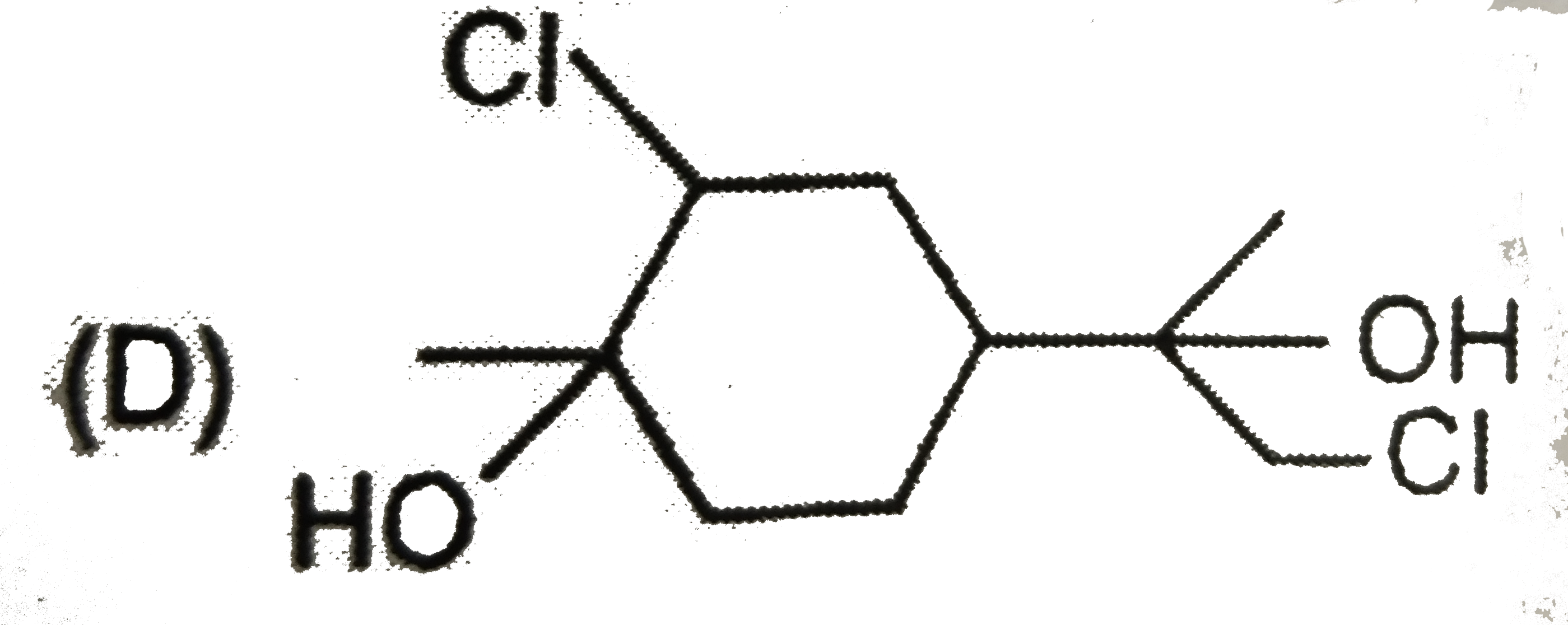
The alkyl halide, C_4H_9Br (A) reacts with alcoholic KOH and gives an alkene (B) which reacts with bromine to form a dibromide (C ). (C ) is transformed with sodamide to a gas (D) which forms precipitate when passed through an ammonical silver nitrate solution. Give the structural formula of compounds (A), (B) , (C ) and (D) and explain the reactions involved. |
Answer» Solution :According to the question , 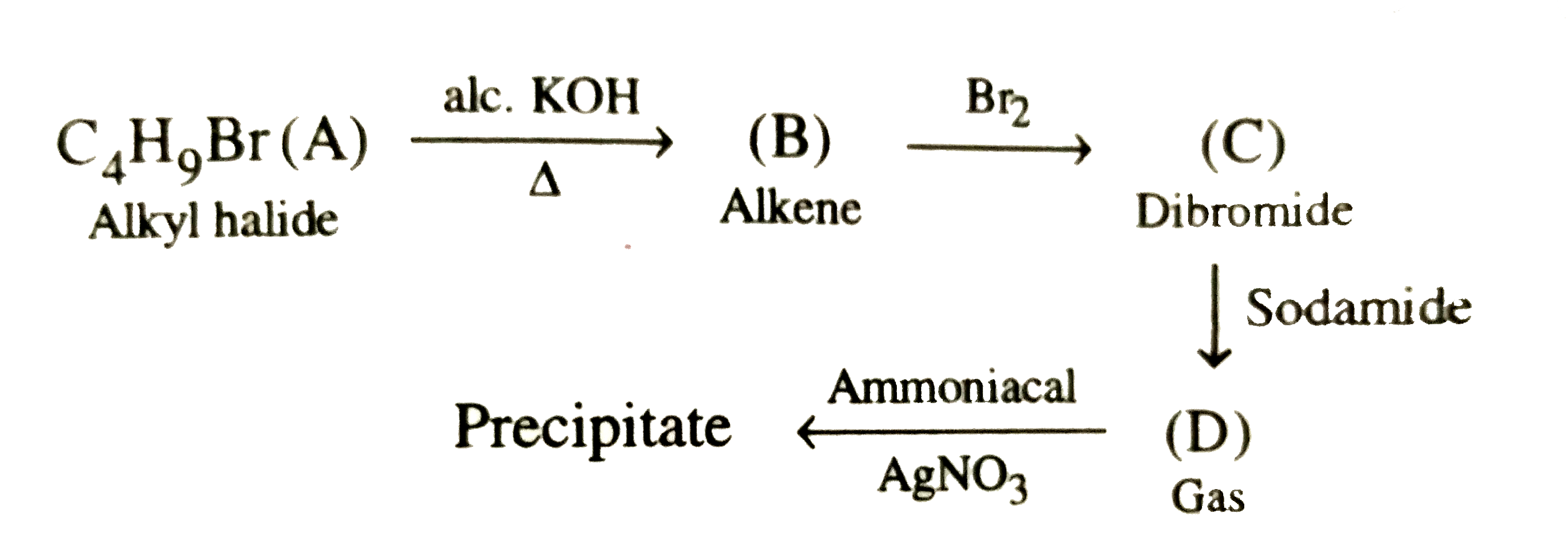 (i)Since the alkyl halide , `C_4H_9Br` (A) on treatment with alcoholic KOH gives an alkene (B) which on subsequent treatment with `Br_2` gives the dibromide (C ) , therefore , the molecular FORMULA of alkene (E) must be `C_4H_8` and that of dibromide (C ) must be `C_4H_8Br_2`. Now since the dibromide , `C_4H_8Br_2` (C ) on treatment with sodamide gives the gas (D), (D) must be an ALKYNE with the molecular formula , `C_4H_6`. Further since the alkyne `C_4H_6` (D) gives precipitate with AMMONIACAL silver nitrate solution, it must be a terminal alkyne. The only terminal alkyne possible for the molecular formula, `C_4H_6` is 1-butyne , i.e., `underset"1-Butyne (D)"(CH_3CH_2-C-=CH)` (ii)Since 1-butyne (D) is obtained by dehydrobromination of dibromide (C ) with sodamide, (C) must be 1,2-dibromobutane i.e., `underset"1,2-Dibromobutane (C)"(CH_3-CH_2-CHBr-CH_2Br)` (iii)Since the dibromide (C ) is obtained by addition of `Br_2` to alkene (B), (B) must 1-butene, i.e., `underset"1-Butene (B)"(CH_3-CH_2-CH=CH_2)` (iv)Since 1-butene is obtained by dehydrobromination of alkyl halide (A) with alcoholic KOH , (A) must be 1-bromobutane or n-butyl bromide , i.e., `underset"1-Bromobutane (A)"(CH_3-CH_2-CH_2-CH_2-Br)` (v) All the reactions cited in the question may now be explained as follows :  Thus , (A) is 1-bromobutane, (B) is 1-butene, (C) is 1,2-dibromobutane and (D) is 1-butyne. |
|
| 20. |
The alklali halide which is soluble in pyridine is |
|
Answer» NaCl |
|
| 21. |
The alkene that will give the same product with HBr in the presence as well as in the presence of peroxides is |
|
Answer» 2-butene |
|
| 22. |
The alkene limonene has the following structure. What product results from the reaction of limonene and chlorine water ? |
|
Answer»
|
|
| 23. |
The alkene that exhibits geometrical isomerism is |
|
Answer» 2-methyl propene 2-butene (CIS & TRANS) |
|
| 24. |
The alkane that yields two isomeric monobromo derivatives is |
|
Answer» Neopentane |
|
| 25. |
The alkaline hydrolysis of esters is also called |
|
Answer» esterification |
|
| 26. |
The alkaline earth metals forming ionic oxides are |
|
Answer» BEO |
|
| 27. |
The alkalineearthmetalsBa,Sr,Caand Mgmay bearrangedin theorderof theirdecreasingfirstionzationenthalpyas ____. |
|
Answer» `MG ,CA,SR ,BA` |
|
| 28. |
The alkaline earth metal whose oxide is amphoteric from the following |
|
Answer» Ba |
|
| 29. |
The alkaline earth metal which has the least density is : |
| Answer» SOLUTION :Ca has the least density . | |
| 31. |
The alkaline earth metal that does not lose its metallic lusture when exposed to air is |
|
Answer» BA |
|
| 32. |
The alkaline earth metal nitrate that does not crystallise with water molecules, is.... |
|
Answer» `Ba(NO_(3))_(2)` So, `Ba(NO_(3))_(2)` does not CRYSTALLISE with water molecules. |
|
| 33. |
The alkali metals react with most non-metals to form hydrides, oxides, sulphides, carbides, phosphides and other compounds. The binary compounds of all alkali metals are ionic with M^(+) cations. The alkali metals easily ionize and readily reduce hydrogen ions. They react with ammonia to evolve hydrogen and form blue coloured solutions. 2M(s) + 2H_2O (l) to 2MOH(aq) + H_2(g) 2M(s) + 2NH_3 (l) to 2MNH_2(am) + H_2(g)Why do alkali metals dissolve in liquid ammonia to give blue coloured solutions ? |
|
Answer» Solution :The ammoniated ELECTRONS are RESPONSIBLE for thebluecolour of the solution . `M+(x+y)NH_3 to [M(NH_3)_x]^(+)+ UNDERSET("Ammoniated electrons")([E(NH_3)_y]^(-)` |
|
| 34. |
The alkali metals react with most non-metals to form hydrides, oxides, sulphides, carbides, phosphides and other compounds. The binary compounds of all alkali metals are ionic with M^(+) cations. The alkali metals easily ionize and readily reduce hydrogen ions. They react with ammonia to evolve hydrogen and form blue coloured solutions. 2M(s) + 2H_2O (l) to 2MOH(aq) + H_2(g) 2M(s) + 2NH_3 (l) to 2MNH_2(am) + H_2(g) Arrangethe following alkali metal ions in the decreasing order of their mobility in aqueous solutions: Li^(+), Na^(+),K^(+),Rb^(+) |
| Answer» Solution :`Rb^(+) GT K^(+) gt NA^(+) gt LI^(+)` | |
| 35. |
The alkali metals react with most non-metals to form hydrides, oxides, sulphides, carbides, phosphides and other compounds. The binary compounds of all alkali metals are ionic with M^(+) cations. The alkali metals easily ionize and readily reduce hydrogen ions. They react with ammonia to evolve hydrogen and form blue coloured solutions. 2M(s) + 2H_2O (l) to 2MOH(aq) + H_2(g) 2M(s) + 2NH_3 (l) to 2MNH_2(am) + H_2(g) Complete the reaction : KO_2 + H_2O to |
| Answer» SOLUTION :`2KO_2 + 2H_2O to 2KOH + H_2O_2 + O_2` | |
| 36. |
The alkali metals react with most non-metals to form hydrides, oxides, sulphides, carbides, phosphides and other compounds. The binary compounds of all alkali metals are ionic with M^(+) cations. The alkali metals easily ionize and readily reduce hydrogen ions. They react with ammonia to evolve hydrogen and form blue coloured solutions. 2M(s) + 2H_2O (l) to 2MOH(aq) + H_2(g) 2M(s) + 2NH_3 (l) to 2MNH_2(am) + H_2(g)Which alkali metal hydroxide is thermally unstable ? |
|
Answer» SOLUTION :LiOH. It DECOMPOSES on heating to GIVE lithium oxide . `2LiOH overset("HEAT")to Li_2O+H_2O` |
|
| 37. |
The alkali metals have low melting point. Which of the following alkali metal is expected to melt if the room temperature rises to 30^(@)C ? |
|
Answer» Na |
|
| 38. |
The alkali metals form salt-like hydrides by the direct synthesis at elevated temperature . The thermal stability of these hydrides decreases in which of the following orders ? |
|
Answer» `NAH gt LiH gt KH gt RbH gt CsH` |
|
| 39. |
The alkali metals form salt like hydrides by the direct synthesis at elevated temperature. The thermal stability of these hydrides decreases in which of the following order |
|
Answer» NaH `gt` LIH `gt` KH `gt` RBH `gt` CsH |
|
| 40. |
The alkali metals dissolve in ammonia to give a deep blue solution which is cnducting in nature. M+(x+y)NH_(3)to[M(NH_(3))_(x)]^(2+)+2[e(NH_(3))_(y)]^(-) which of the followin is not true about the solutions of alkali metals in liquid ammonia? |
|
Answer» The blue colour is due to ammoniated electron |
|
| 41. |
The alkali metals form salt like hydrides by the direct synthesis at elevated temperature. The thermal stability of these hydrides decreases in which of the following order ? |
|
Answer» `NaH GT LIH gt KH gt RbH gt CsH` |
|
| 42. |
The alkali metals font salt likelydrides by direct synthesis at elevated temperatures. The thermal stability of these hydrides decreases in which of the following onders? |
|
Answer» `NaH GT LIH gt KH gt RbH gt Cs H` |
|
| 43. |
The alkali metals dissolve in ammonia to give a deep blue solution which is conducting in nature. M+(x+y)NH_3rarr[M(NH_3)_x]^(2+)+2[e(NH_3)_y]^(-) Which of the following is not true about the solutions of alkli metals in liquid ammonia ? |
|
Answer» The blue colour is due to ammoniated electron `M _((AQ)^(-)e_((aq)^(-) + NH_(3(l)) to MNH_(2(aq)) +1/2 H_(2(g))` |
|
| 44. |
Give a reason for each of the following: Alkali metals are good reducing agents. |
|
Answer» low ionisation energy |
|
| 45. |
The alkali metals are low melting . Which of the following alkali metals is expected to melt if the room temperature rises to 30^(@)C ? |
| Answer» Solution :Among alkali metals, m.p. decreases as the strength of metallic bonding decreases with increasing size of the atom . Thus , Cs has the lowest m.p. and MELTS at `30^(@)`C. | |
| 46. |
The alkali metals are low melting. Which of the following alkali metals is expected to melt if the room temperature rises to 30^(@)C ? |
|
Answer» NA |
|
| 47. |
The alkali metals are low melting. Which of the following alkali metals is expected to melt if the room temperature rises to 30^(@)C? |
|
Answer» Na `therefore` Cs has lowest melting point `(28.44^(@)C)`and will melt at `30^(@)C` |
|
| 48. |
The alkali metals and their salts impart characteristic colour when kept in Bunsen flame . Regarding this which of the following statements is not correct . |
|
Answer» The colour is produced in oxidising flame . |
|
| 49. |
The alkali metals |
|
Answer» form salt-like hydrides |
|