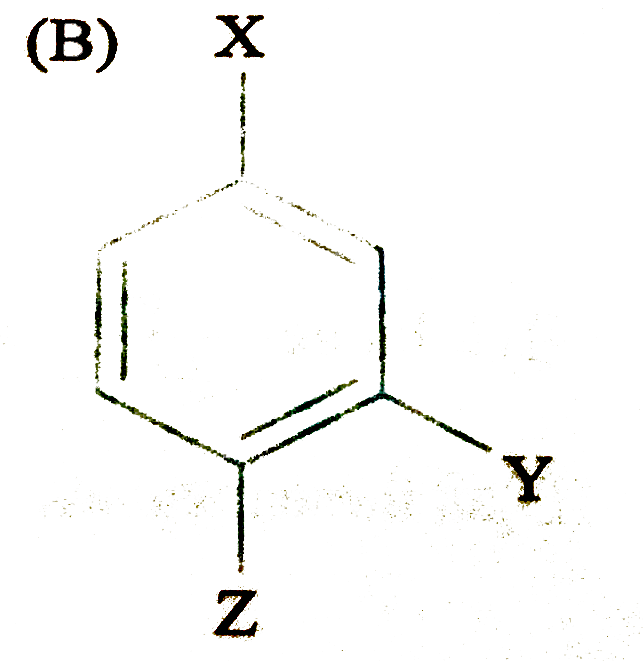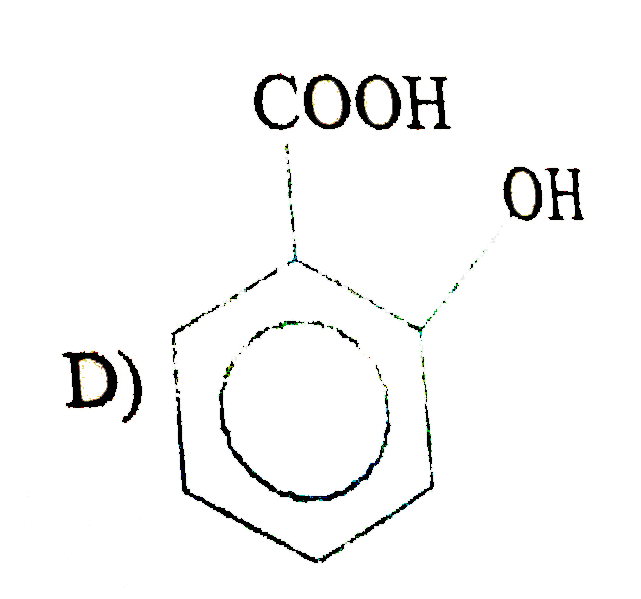Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
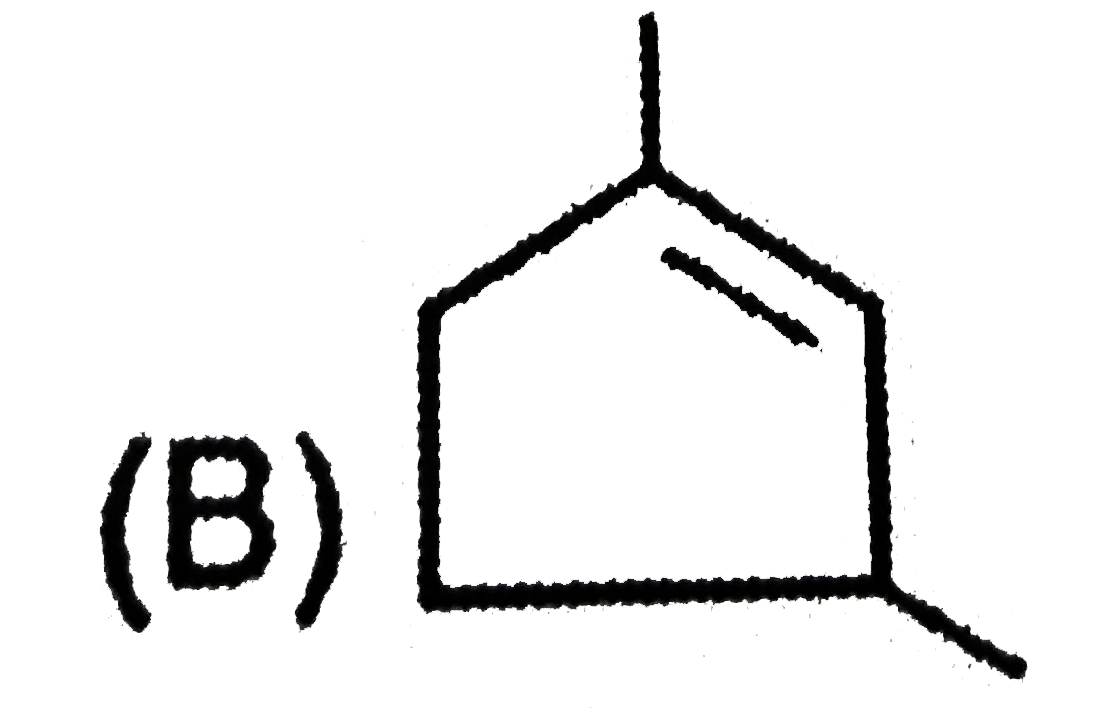
| 1. |
The compound with mol. Mass 180 is acetylated with CH_(3)COCl to get acetylated compound with mol. Mass 390. The number of amino groups present per molecule of the compound is |
|
Answer» 2 The INCREASE in mass PER acetyl group = 42 AMU Total increase in mass `= 390 - 180 = 210` `:.` The number of acetyl groups ADDED `= (210)/(42) = 5` |
|
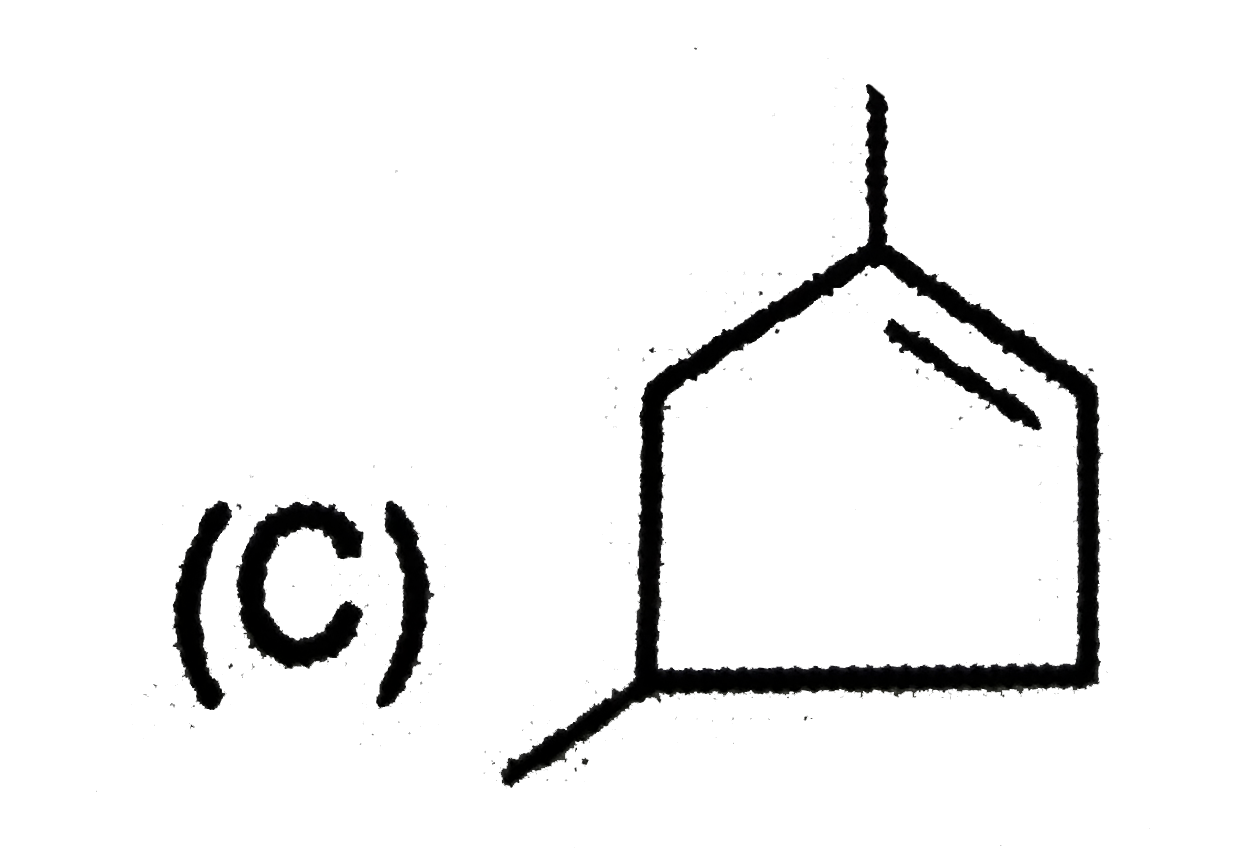
| 2. |
The compound with an isopropyl group is |
|
Answer» 2,2,3,3-Tetramethylpentane |
|
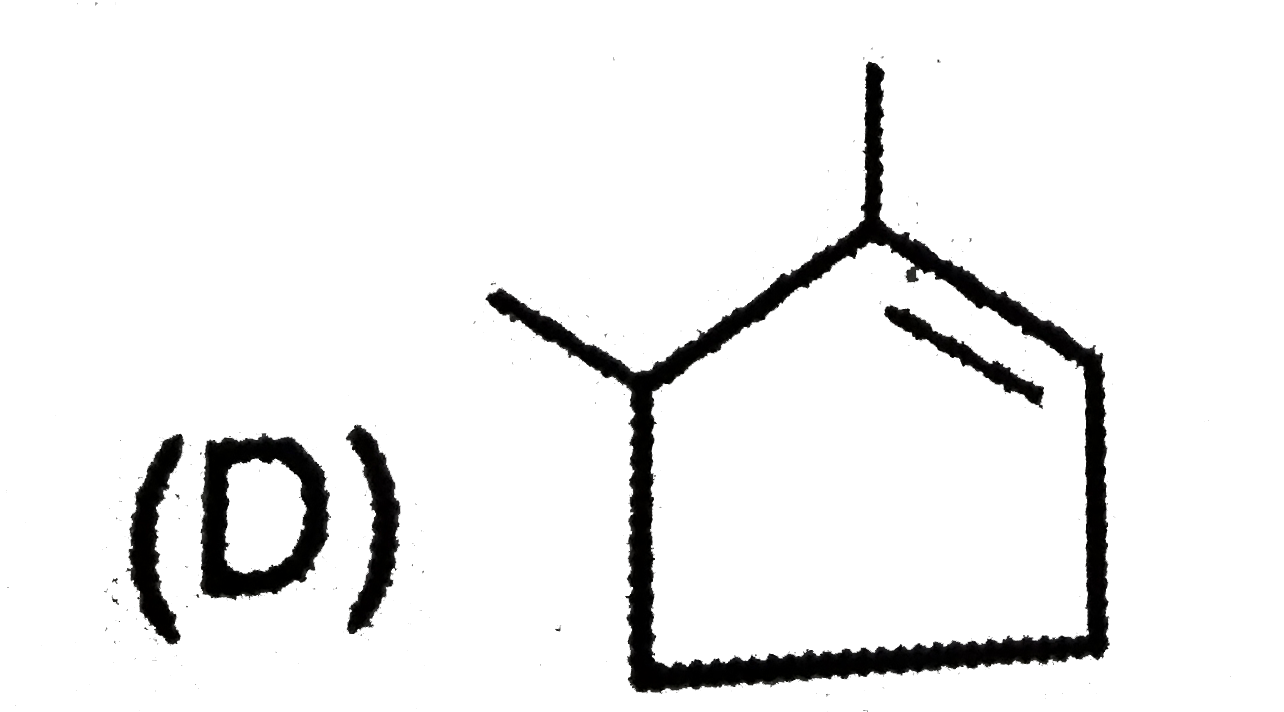
| 3. |
The compound which will give red colour in Victor Meyer's test is |
|
Answer» Propan-2-ol |
|
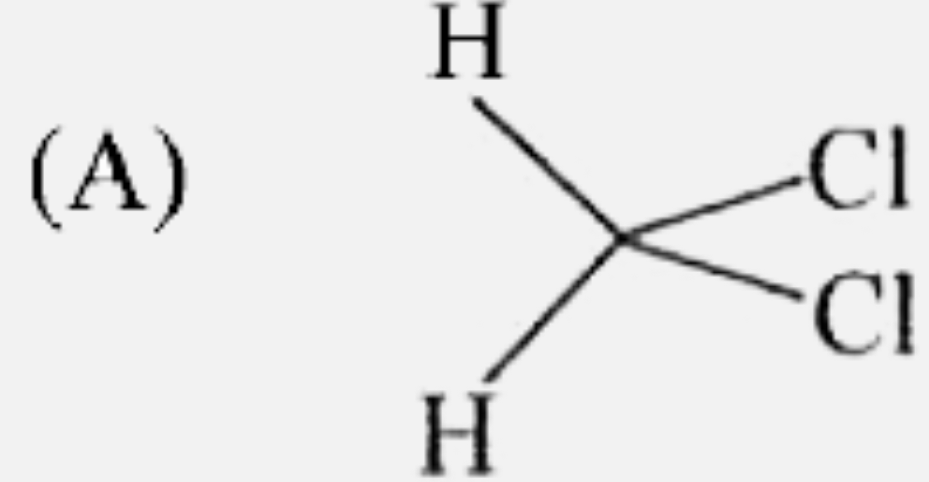
| 4. |
The compound which undergoes dehydration very easily is : |
|
Answer» 2-Methylpropan-2-ol Being TERTIARY in nature, the alcohol UNDERGOES DEHYDRATION most easily. |
|
| 5. |
The compound which produces propane on heating with HI in presence of red phosphorus is a) |
|
Answer» <P>`CH_3 CH_2CH_2I` All PRODUCE porpane on REDUCTION with HI/red P. |
|
| 6. |
The compound which possessestheantifluorite structure is |
|
Answer» `Rb_(2)S` |
|
| 7. |
Thecompoundwhichis NOTresentasimpurityin crudeNaClis ____ |
|
Answer» `CaSO_(4)` |
|
| 8. |
The compound which is not isomeric with diethyl ether is…. |
|
Answer» METHYL n-propyl ether |
|
| 9. |
The compound which is not formed during dry distillation of a mixture of calcium formate and calcium acetate is |
|
Answer» Methanal |
|
| 10. |
The compound which has molecular nature in gas phase but ionic in solid state is |
|
Answer» `PCl_(5)` |
|
| 11. |
The compound which gives the most stable carbonium ion on dehydration is : |
|
Answer» `(CH_(3))_(2)CHCH_(2)OH` |
|
| 12. |
The compound which gives the most stable carbonium ion on dehydrataion is |
|
Answer» `CH_(3) - CH_(2) - CH_(2) - CH_(2)OH` `CH_(3) - undersetunderset(CH_(3))(|)oversetoverset(CH_(3))(|)C-overset(oplus)OH_(2) overset(-H_(2)O)to(CH)overset(oplus)C` `3^(@)` CARBONIUM ION is more stable |
|
| 13. |
The compound which does not react with bromine easily at room temperature is |
|
Answer» phenol |
|
| 14. |
The compound which does not react with lithium aluminium hydride is |
|
Answer» 3-penten-2-one |
|
| 15. |
The compound which decolourises bromine water but does not give white precipitate with Tollen's reagent. |
|
Answer» `C_(2)H_(2)` |
|
| 16. |
The compound which contains both ionic and covalent bonds is |
| Answer» Answer :C | |
| 17. |
The compound which acts as an oxidising as well as a reducing agent is- |
|
Answer» `NaNO_3` |
|
| 18. |
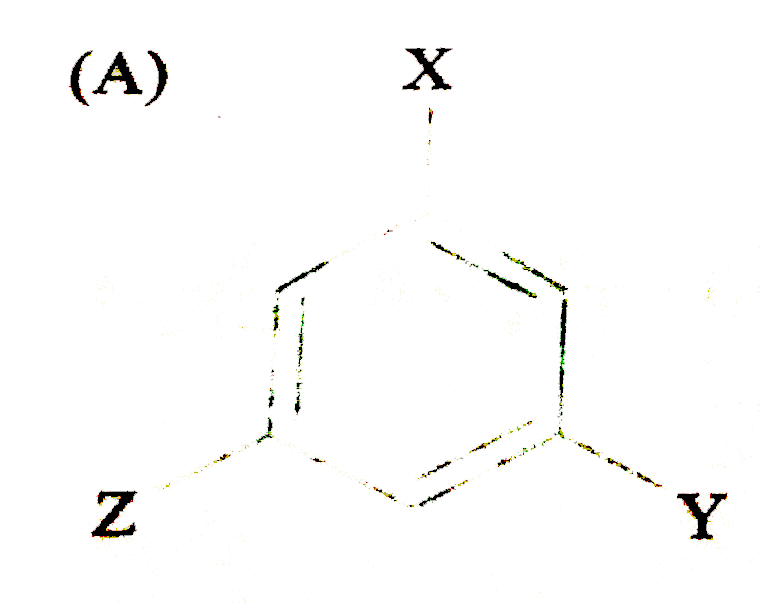
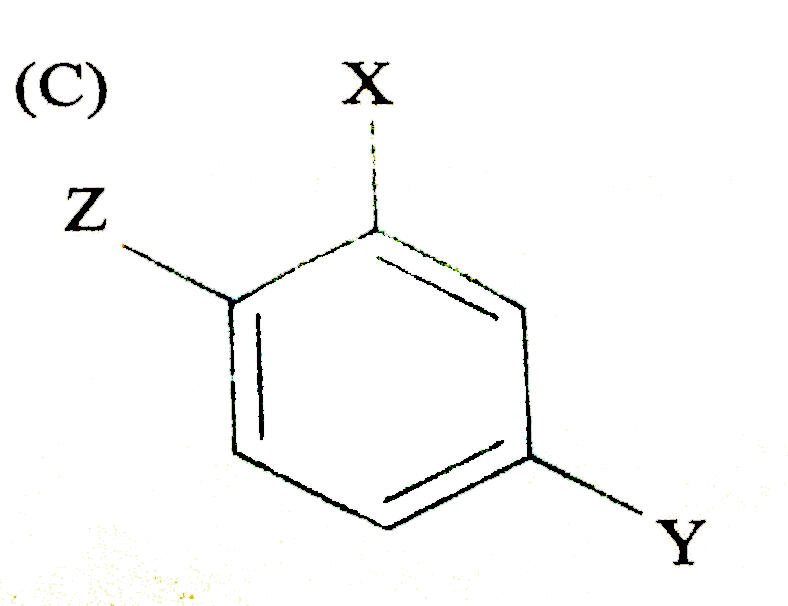
The compound (where X is an O,P-directing group and Y is m-directing group) is subjected to electrophilic substitution reaction for introduction of Z. the compound formed would be |
|
Answer»
|
|
| 19. |
The compound used to manufacture phenol is |
|
Answer» Glycerol |
|
| 20. |
The compound used as refrigerant is |
| Answer» Solution :Freon | |
| 22. |
The compound that will react most readily with gaseous bromine has the formula |
|
Answer» `C_(4)H_(10)` `CH_(3)-CH=CH_(2)overset(Br_(2))rarr underset(Br)underset(|)(CH_(2))-CH=CH_(2)` |
|
| 23. |
The compound that will have a permanent dipole moment among the following is: |
|
Answer»
|
|
| 24. |
The compound that undergoes dehydration most readily is |
|
Answer» Ethyl ALCOHOL |
|
| 25. |
The compoundthat wilreact most readily with gaseous bromine has the formula ………….. |
|
Answer» `C_(3)H_(6)` |
|
| 26. |
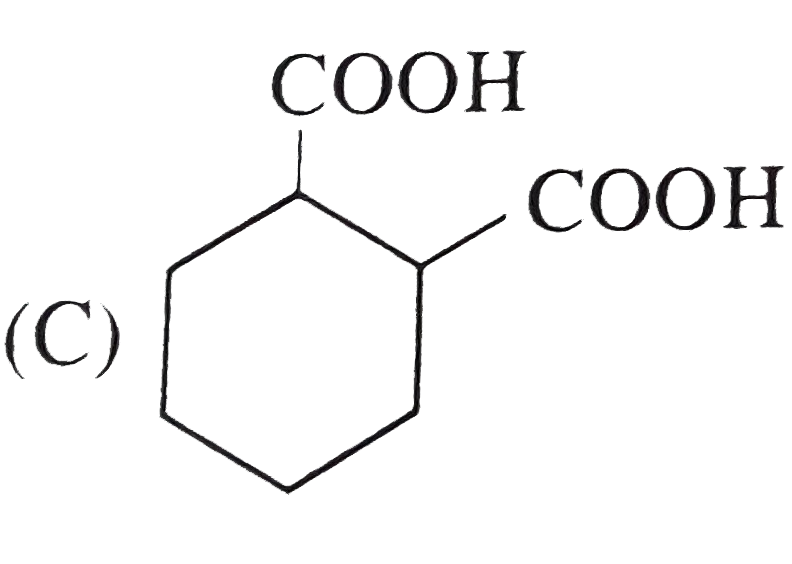
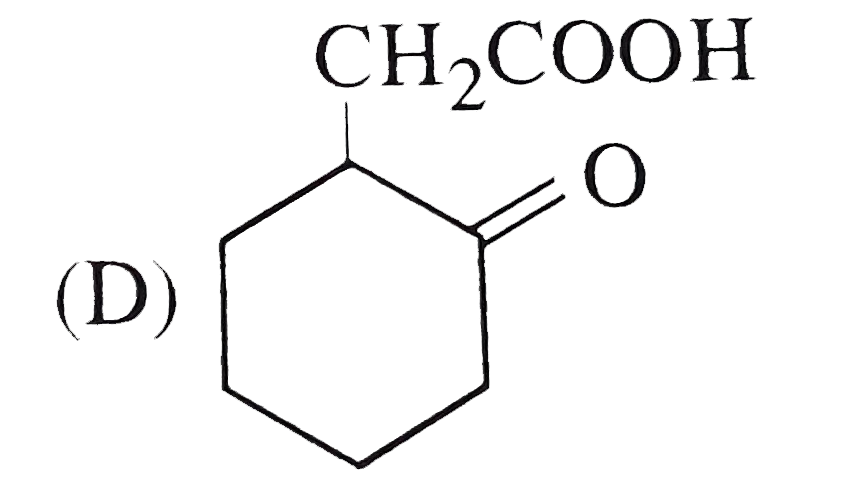
The compound that undergoes decarboxylation most readily under mild conditions are |
|
Answer»
|
|
| 27. |
The compound that is used in nuclearindustry as protective shields and control rods is |
|
Answer» METAL carbonates |
|
| 28. |
The compound that is most reactive with alcoholic KOH is |
|
Answer» `CH_2=CH-Br` |
|
| 29. |
The compound that does not undergo hydrolysis by S_(N)1 mechanism is |
|
Answer» `CH_(2)=CHCH_(2)CL` |
|
| 30. |
The compound that does not produce nitrogen gas by thermal decomposition is |
|
Answer» `BA(N_(3))_(2)` `(NH_(4))_(2)SO_(4)hArr2NH_(3)+H_(2)SO_(4)` |
|
| 31. |
The compound that does NOT liberate CO_(2) , on treatment with aqueous sodium bicarbonate solution is : |
|
Answer» BENZOIC ACID |
|
| 32. |
The compound that does not give a tribromo derivative on treatment with bromine water is |
|
Answer»
|
|
| 34. |
The compound that can work both as oxidising and reducing agent is _____ |
|
Answer» `KMnO_(4)` |
|
| 35. |
The compound that can work both as an oxidizing and reducing agent is |
|
Answer» ` KMnO_4` Element is its intermediate O.S can work both as an O.A &R.A |
|
| 36. |
The compound showing below is cyclic hemiacetal of |
|
Answer» 5-Hydroxyheptanal 
|
|
| 37. |
The compound (s) with two lone pairs of electrons on the central atom is (are) |
|
Answer» `BrF_(5)` with five F atoms . Hence, lonepair =1 In`CIF_(3)` Cl has 7 valence electrons, three are shared with three F atom, lone pairs =2. In`XeF_(4)`,Xe has 8 valence electrons, FOUR are shared with four F atoms, lone pairs =2 In`SF_(4),S` has 6 valence electrons, four are shared with four F atoms, lone pair =1 |
|
| 38. |
The compound R is : |
|
Answer»

|
|
| 39. |
The compound Q is : |
|
Answer»

|
|
| 40. |
The compound [Pd(C_(x)H_(y)N_(z)](CIO_(4))_(2) contains 30.15% carbon and 5.06% hydrogen. The compound [Pb(C_(x)H_(y)N_(z)] (SCN)_(2) contains 40.46% carbon and 5.94% hydrogen. Calculate x, y,z. |
|
Answer» |
|
| 41. |
The compound prepared by substitution reaction of benzene is |
|
Answer» ACETOPHENONE |
|
| 42. |
The compound P is : |
|
Answer»

|
|
| 43. |
The compound of 'X' and 'Y' may be: |
|
Answer» `Na_(2)CO_(3),BaCO_(3)` |
|
| 44. |
The compound of which alkali metal gives crimsonred I'm flame test? |
|
Answer» Li |
|
| 45. |
The CompoundM-O-H canactbothas anacid orbasedependinguponthe ionizationenthalpyofthe elementM. Justify by takingelements(M) of the thirdperiodof the periodic table . |
Answer» Solution :If theionizationenthalpy of theelements M islow it has astrongtendencyto getoxidisedto `M^(+)` . As aresult the M-OHbond willbreakto produce `M^(+)` and `OH^(-)` ions andhencethe compoundM-OHactsas a base  Furtherlower theionizationenthalpy of THEELEMENT (M) , stronger is the basic strength of thecompound M-O-H.Sincein generalionizationenthalpyincreases fromleftto rightin aperiodthereforebasiccharacterdecreasesin the sameorder. ThusNaOHis thestrongest basicfollowedby Mg `(OH)_(2)` while`A1(OH)_(3)`is theweakestbase. Infact`A1(OH)_(3)`is AMPHOTERICIN character. On theotherhandif theionizationenthalpyof the elementsM is high is haslittletendency ofgetconvertedinto `M^(+)` ion. Insteadthe compoundM-OH ionizationto produce `H^(+)` ionsand hencethe compoundM-O-H behavesas an acid.  Furtherhigher the IONIZATIONENTHALPY of theelement (M), stronger isthe acidicstrengthof the compound M-O-H.Sincein generalionizationenthalpyincreasesfrom lefttorightin aperiod acidiccharacterincreasesin thesameorder. forexample`Si(OH)_(4) " or" H_(2) SiO_(3)` is the weakestacid O=`P(OH)_(3)" or" H_(3) PO_(4)` is astronger acidthan `H_(2) SiO_(3), O_(2) S(OH)_(2)" or" H_(2)SO_(4)` is amoderratelystrong acidwhile`O_(3) CI(OH)` or `HC1O_(4)` is thestrongest acid . Thusfrom theabovediscussionit follows that as theionizationenthalpy of the element (M)increases from left to rightin a periodthe basiccharacterof thecompoundMOHdecreases whilethe acidic characterincreases. |
|
| 46. |
The compound insoluble in acetic acid is..... |
|
Answer» CALCIUM oxide |
|
| 47. |
The compound insoluble in acetic acid acid is |
|
Answer» `CAO` |
|
| 49. |
The compound in which Causes only SP^3hybrid orbitals for bond formation is |
|
Answer» HCOOH All CARBONS are `Sp^(3)` hybridised. |
|