Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The concentration of commercial H_2O_2is 3.125 M than its grade is ______volume. |
| Answer» SOLUTION :35 VOLUME | |
| 2. |
The concentration of Ag_2 Cr_2O_7 is 6.5xx10^(-5)M in concentrated solution of Cr_2O_7^(2-)at a temp. then calculate K_2Cr_2O_7 |
|
Answer» SOLUTION :`K_(sp)=[Ag^+]^2 [Cr_2O_7^(2-)]` `therefore K_(sp)=4S^3=4(6.5xx10^(-5))^3` `=1.098xx10^(-12)` Where S=Solubility of `Ag_2Cr_2O_7=6.5xx10^(-5)` M |
|
| 3. |
The concentration of Al^(3+)ion in aqueous solution of Al_(2)(SO_(4))_(3) is 0.28 M Then the concentration of SO_(4)^(2-) ion in this solution will be: |
|
Answer» 0.28 M `Al_(2)(SO_(4))_(3) overset(H_(2)O)rarr 2Al_((aq))^(3+) + 3SO_(4(aq))^(2-)` ACCORDING to STOCHIOMETRY of the REACTION `2AL^(3+)`IONS and `3SO_(4)^(2-)` ions are formed. `SO_(4)^(2-)` ions `=(0.28xx3)/(2) = 0.42 M` |
|
| 4. |
The concentration of acetic acid required to get 3.5 xx 10^(-4) mole/lit of H^(+) ion is [K_(a) = 1.8 xx 10^(-5)] |
|
Answer» `6.8 XX 10^(-3) mol lit^-1` |
|
| 5. |
The concentration of 500 ML NaOH solution is 0.02 M. How many grams of FeSO_4 added in this solution for precipitation of Fe(OH)_2 ? The K_(sp)of Fe(OH)_2 is 1.5 xx 10^(-15), Molecular mass of Fe(OH)_2 is 152 g "mol"^(-1)) |
| Answer» SOLUTION :More than `2.85xx10^(-10)` GM | |
| 6. |
The concentration term is used in the calculation of vapour pressure of solution is …………….. |
| Answer» SOLUTION :MOLE FRACTION | |
| 7. |
The compund Yb Ba_(2)Cu_(3)O_(7) which shows superconductivity has copper in oxidation stte…………..Assume that the reare earth element ytterbium isin the usual + 3oxidation state |
|
Answer» |
|
| 8. |
The compressibilty factor for a real gas at high pressure is: |
|
Answer» `1 + (RT)/(Pb)` `(P+ (a)/(V^(2))) (V - b) = RT` Pressure correction can be NEGLECTED at high pressure. THUS, `P (V - b) = RT` `PV = Pb + RT` `(PV)/(RT) = 1 + (Pb)/(RT)` `Z = 1 + (Pb)/(RT)` |
|
| 9. |
The compression factor (compressibility factor) for one mole of a van der Waals gas at 0^(@)C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is negligible, calculate the van der Waals constant a. |
|
Answer» Solution :`Z=(PV)/(NRT),i.e., 0.5=(100xxV)/(1xx0.082xx273)"or"V=0.1119" L"` `(P+(a)/(V^(2)))(V-B)=RT` for 1 MOL Neglecting b, `(P+(PV)/(V^(2)))V=RT "or"PV+(a)/(V)=RT"or" (PV)/(RT)+(a)/(VRT)=1` or `"" a=(1-(PV)/(RT))VRT=(1-0.5)0.1119xx0.082xx273=1.252" atm "L^(2)mol^(-2)` |
|
| 10. |
The compression factor for one mole of real gas at 0^@C and 100 atm is 0.5. Calculate the van der Waals' constant 'a', if 'b' is zero. |
|
Answer» |
|
| 11. |
The compressibility of a gas is less than unity at STP. Therefore, |
|
Answer» `V_(m) GT 22.4L` |
|
| 12. |
The compressibility of a gas is less than unity at N.T.P.Therefore. |
|
Answer» `V_(m) GT 22.4 L` For ideal gas, `V_(m) = 22.4 L` at N.T.P. At a given TEMPERATURE and pressure if `Z` is LESS than unity, then `V_(m) lt 22.4 L` |
|
| 13. |
The compressibility of a gas is less than unity at S.T.P |
|
Answer» `V_(m)gt22.7` LITRES |
|
| 14. |
The compressibility factor (Z) for one mole ofa gas is more than one under S.T.P. conditions. Therefore |
|
Answer» `V GT 11.2 L` `(PV)/(nRT) gt 1` or `V gt (nRT)/(P)` `V gt ((1MOL) XX (0.0821 L atmK^(-1) mol^(-1)) xx 273K)/((1atm)) gt 22.4 L` |
|
| 15. |
The compressibilityfactor ( z) for a real gas at its Boyle temperature is "_____________". |
| Answer» ANSWER :A | |
| 16. |
The compressibility factor Z for an ideal gas will be ......... |
|
Answer» `1.5` |
|
| 17. |
The compressibility factor of an ideal gas is |
|
Answer» 1.0 |
|
| 18. |
The compression factor (compressibility factor) for one mole of a van der Waals gas at 0°C and 100 atmospheric pressure is found to be 0.5. Assuming that the volume of a gas molecule is negligible, calculate the van der Waals constant a. |
|
Answer» 1.253 ATM LIT`""^2MOL^(-2)` |
|
| 19. |
The compressibility factor for a real gas at high pressure is : |
|
Answer» `1+ (RT)/(PB)` |
|
| 20. |
The compressibility factor for a real gas at high pressure is |
|
Answer» 1 At high pressure, `P GT gt a//V^(2)` |
|
| 21. |
The compounds which on heating produce at least one polymeric product, are: |
|
Answer» `Na(NH_(4))HPO. 4H_(2)O` |
|
| 22. |
The compound(s) which have -O-O- bond (s)is / are |
|
Answer» `BaO_(2)` |
|
| 23. |
The compounds which gives the most stable carboniurn ions on dehydrohalogenation is |
|
Answer» `CH_3I` |
|
| 24. |
The compound(s) which generate(s) N_(2) gas upon thermal decomposition below 300^(@)C is (are) |
|
Answer» `NH_(4)NO_(3)` |
|
| 25. |
The compound(s) which contain ionic, covalent and coordinate bonds is (are) |
|
Answer» `H_(2)SO_(4)` |
|
| 26. |
The compounds which are used for denaturation of ethyl alcohol are |
|
Answer» Paphtha |
|
| 27. |
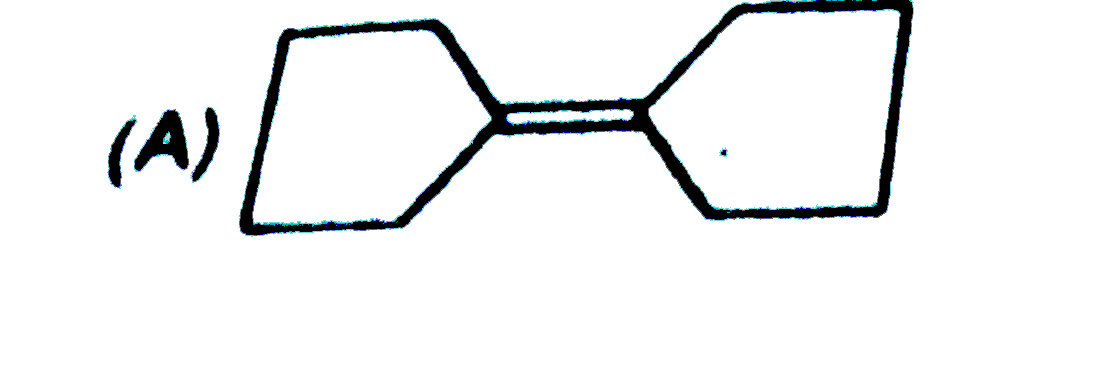
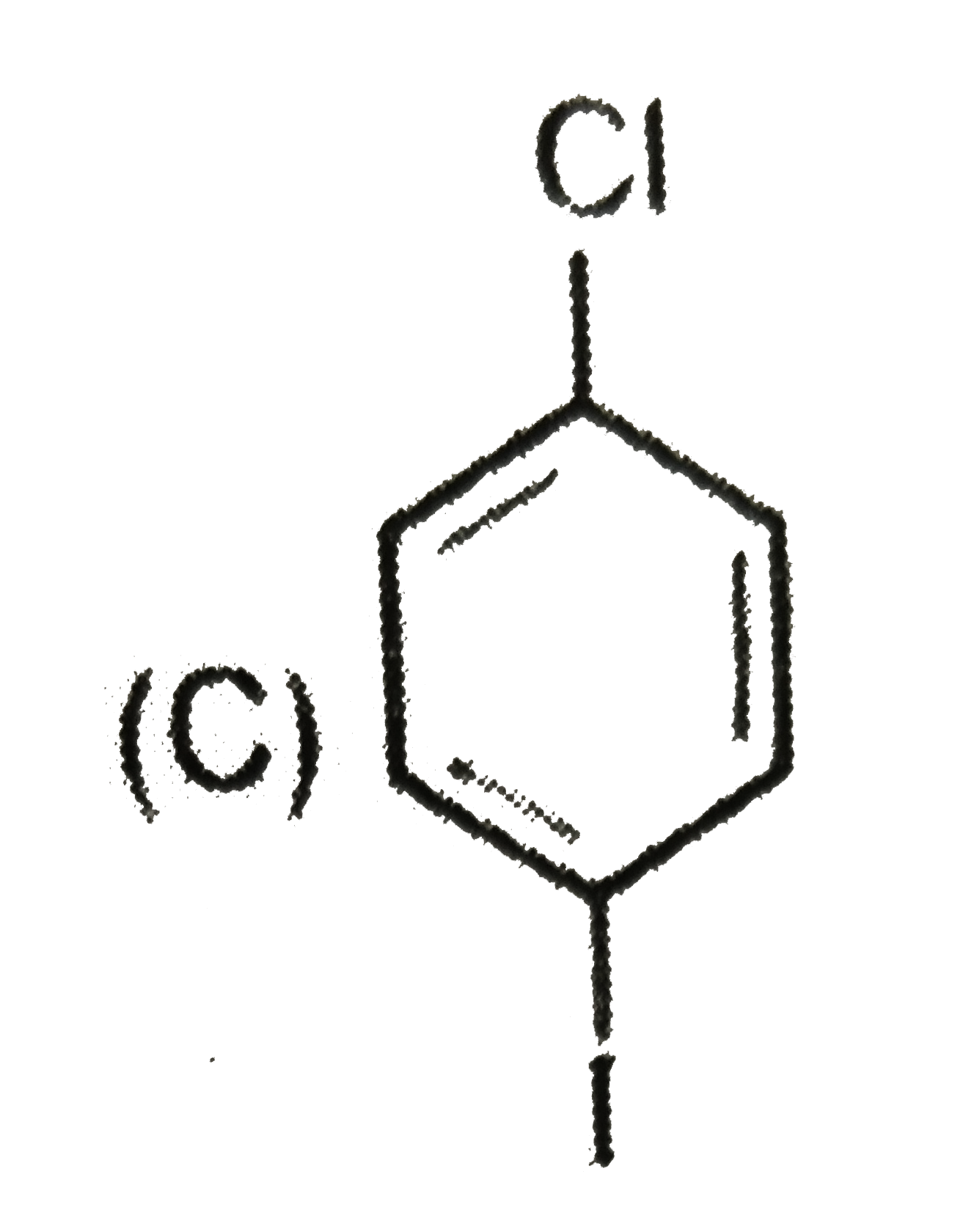
The compound(s) that undergo slower electrophilic substitution than benzene is/are |
|
Answer»
|
|
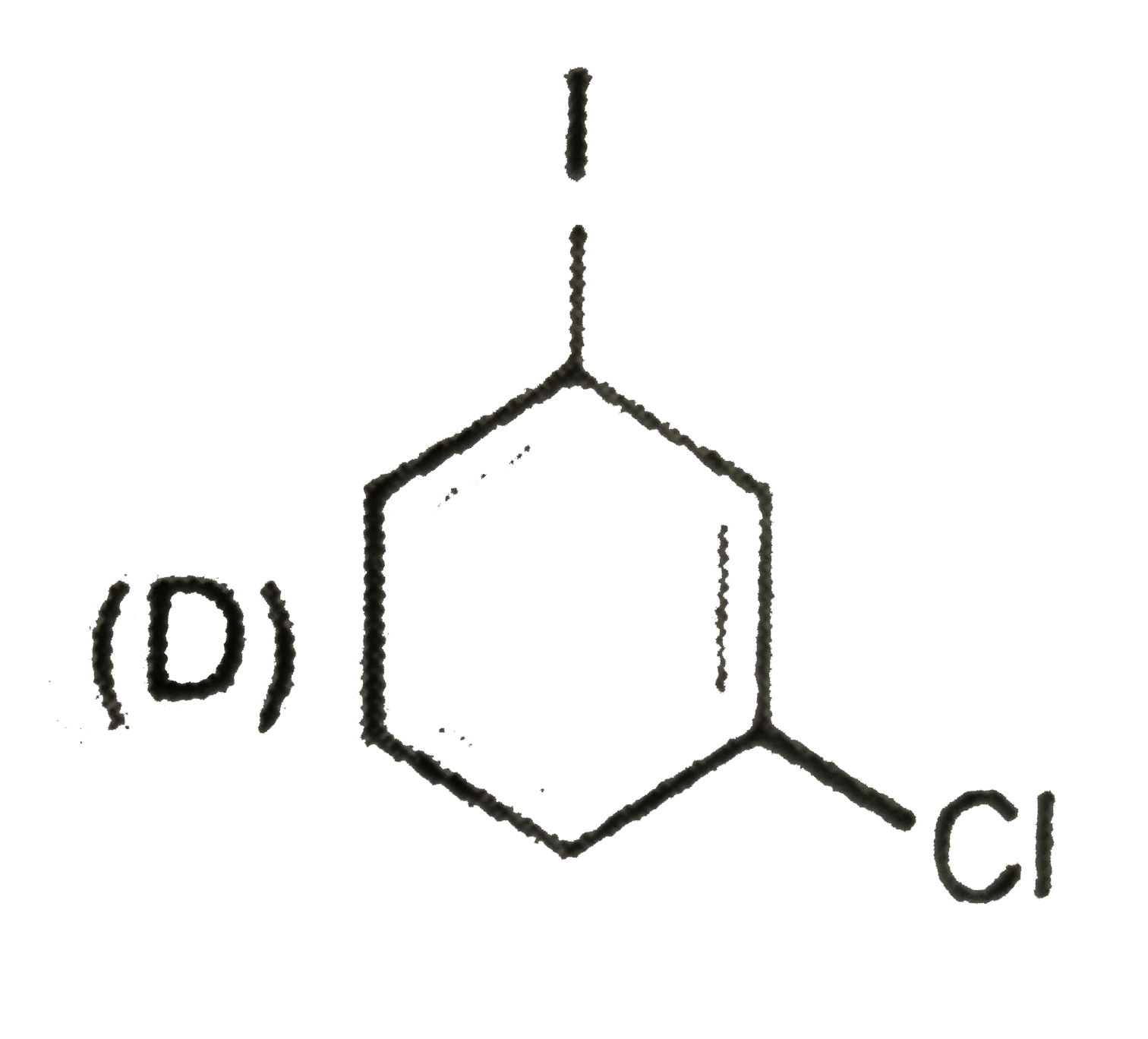
| 28. |
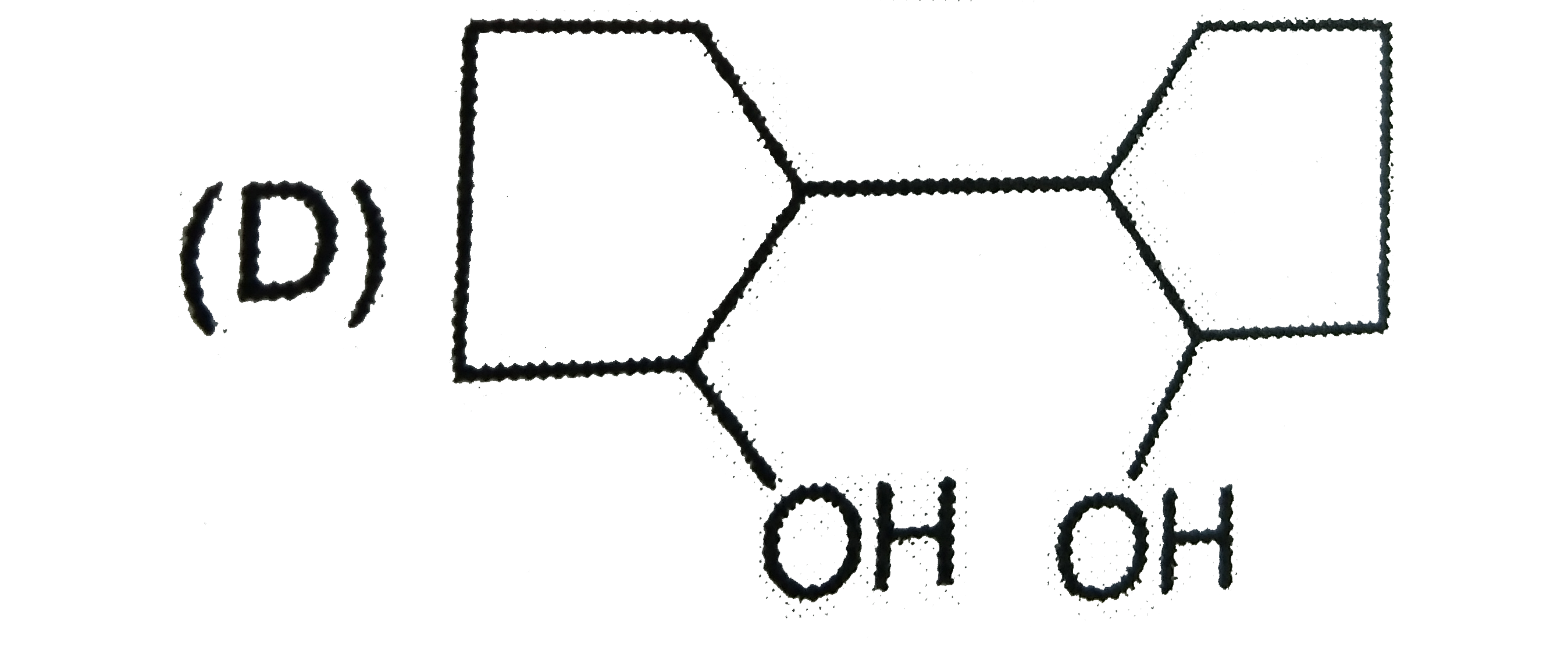
The compound(s) that direct incoming electrophile predominantly at meta position is/are |
|
Answer»
|
|
| 29. |
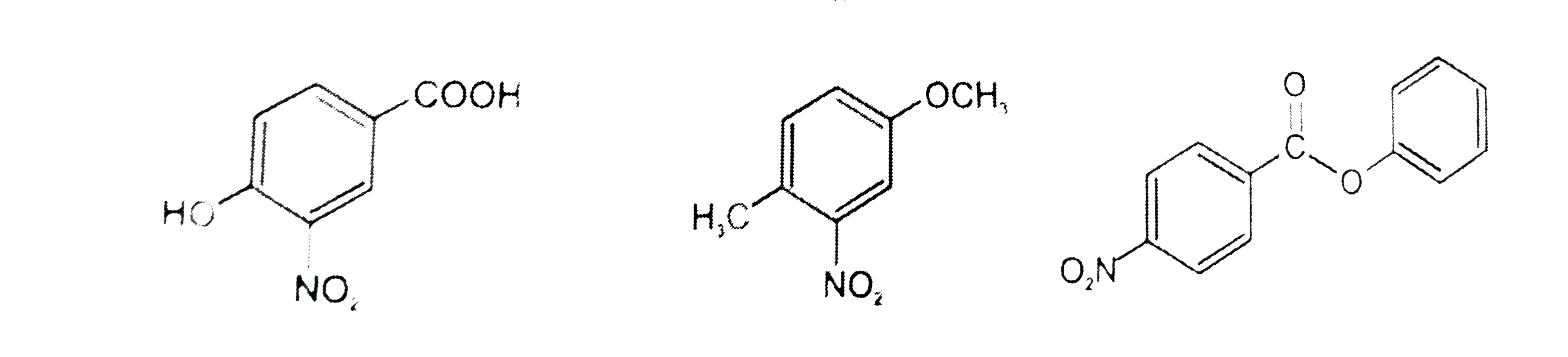
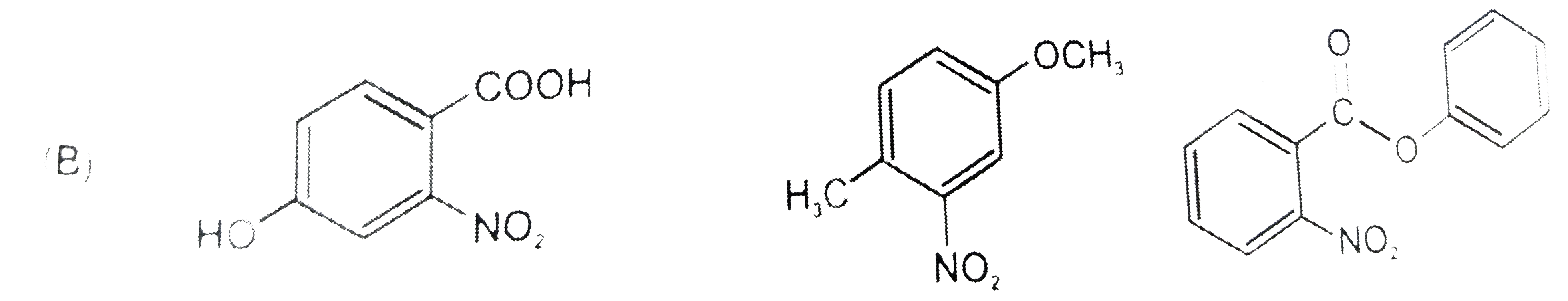
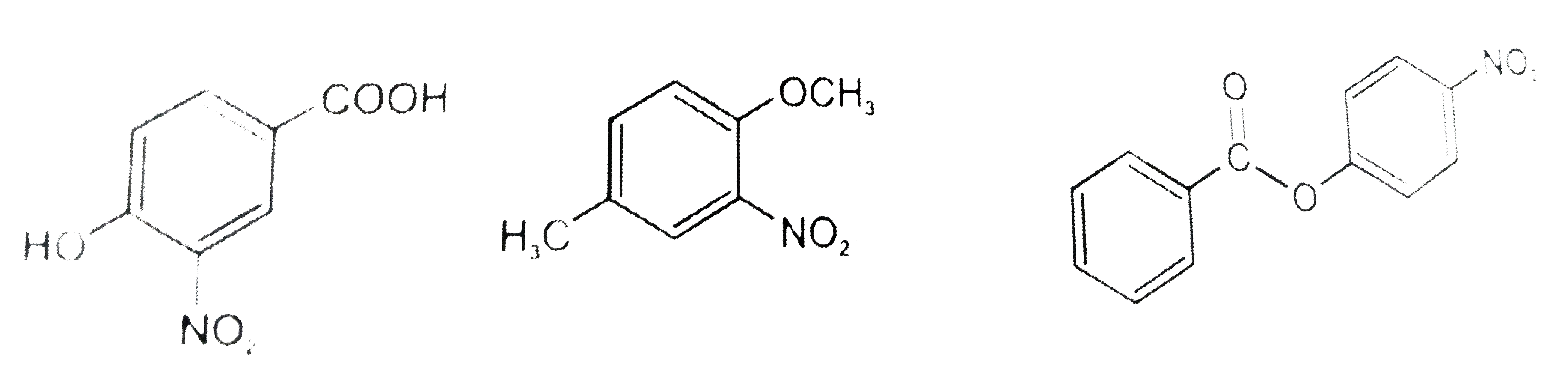
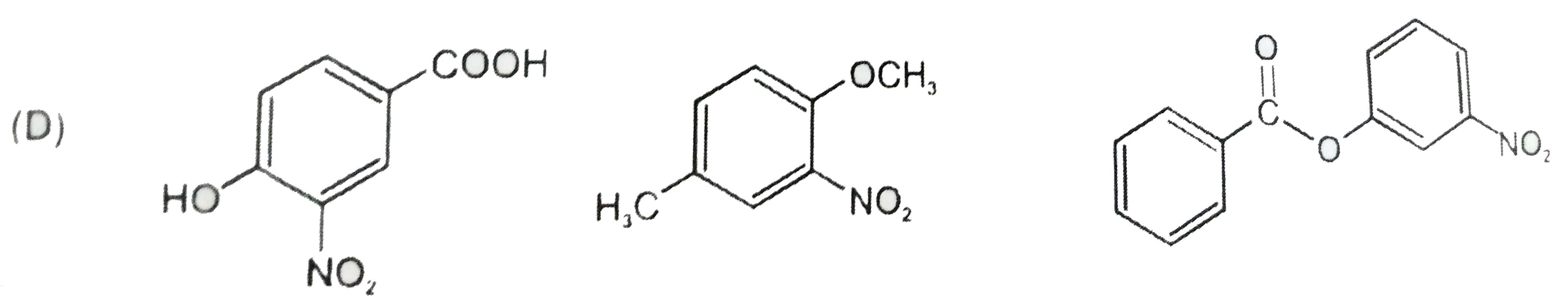
The compounds P,Q and S were separately to nitration using HNO_3//H_2SO_4 mixture.The major product formed in each case respectively is : |
|
Answer»
|
|
| 30. |
The compounds(s) of alkaline earth metals, which are amphoteric in nature is / are |
|
Answer» BeO |
|
| 31. |
The compounds of alkaline earth metals have the following magnetic nature. |
|
Answer» Diamagnetic |
|
| 32. |
Fill in the blanks The compounds of Al are predominantly ............ due to the ............ size and ............ charge of A1^(3+) ions. |
| Answer» SOLUTION :COVALENT , SMALL , HIGH | |
| 33. |
The compounds formed at anode in the electrolysisof an aqueous solution of potassium acetate are……………. |
|
Answer» `CH_(4)` NAD `H_(2)` |
|
| 34. |
The compounds CH_3 CH = CH CH_3 and CH_3 CH_2 CH = CH_2 (a) are tautomers (b) are position isomers (c) contain same number of sp^3 sp^3, sp^3 - sp^2 and sp^2 - sp^2 carbon-carbon bonds (d) exist together in dynamic equilibrium (e) are optical isomers |
|
Answer» |
|
| 35. |
The compounds C_(2)H_(5)OC_(2)H_(5) and CH_(3)OCH_(2)CH_(2)CH_(3) are |
|
Answer» CHAIN isomers |
|
| 36. |
The compounds A and B are mixed in equimolar proportion to from the products, A+B harr C+D. At equilibrium, one third of A and B are consumed. The equilibrium constant for the reaction is |
|
Answer» 0.5  `K_(C)=((a)/(3) XX (a)/(3))/((2a)/(3) xx (2a)/(3))=(1)/(4)` |
|
| 37. |
The compounds A, B, C have R or S configurations respectively |
|
Answer» R,R,S |
|
| 38. |
The compound Y is |
|
Answer» `BCl_(3)` |
|
| 39. |
The compound Y in the above sequence is |
|
Answer» 2-Methyl-2-phenyl-1-propanol 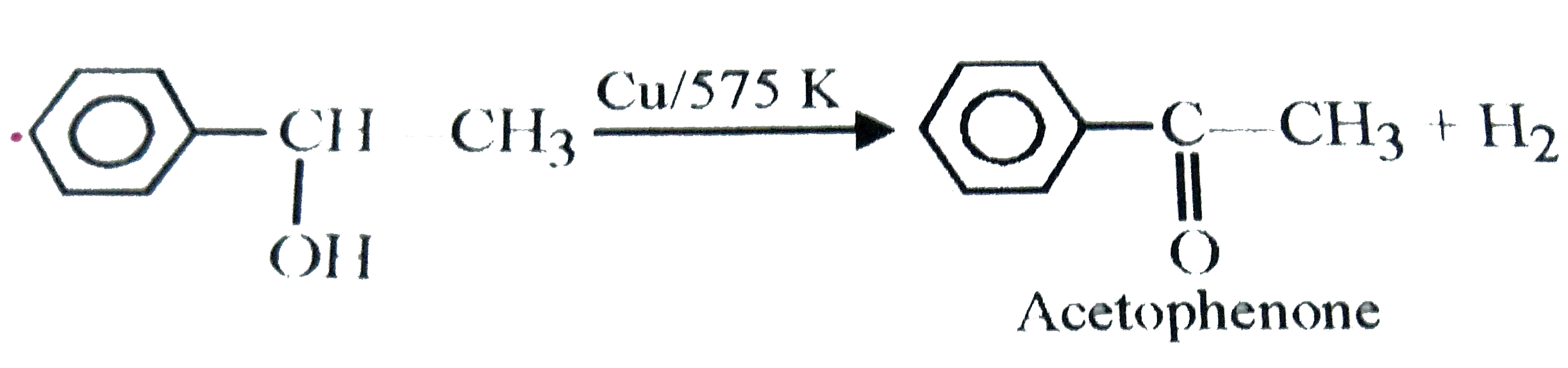
|
|
| 40. |
The compound (X) on heating gives a colourless gas and a residue that is dissolved in water to obtain (B). Excess of CO_(2) is bubbled through aqueous solution of B, C is formed. Solid (C) or heating gives back X. (B) is …...... |
|
Answer» `CaCO_(3)` `CaO+H_2OtoCa(OH)_2 , Ca(OH)_2+CO_2to CaCO_3+H_2O` |
|
| 41. |
The compound (X) on heating gives a colourless gas a nd a residue that is dissolved in water to obtain (B). Excess of CO_2 is bubbled through aqueous solution of B, C is formed. Solid (C) on heating gives back X. (B) is |
|
Answer» `CaCO_(3)` `CaO+H_(2)OtoCa(OH)_(2)` `Ca(OH)_(2)+CO_(2)toCaCO_(3)+H_(2)O` |
|
| 42. |
The compound "X" is used as |
| Answer» Answer :A | |
| 43. |
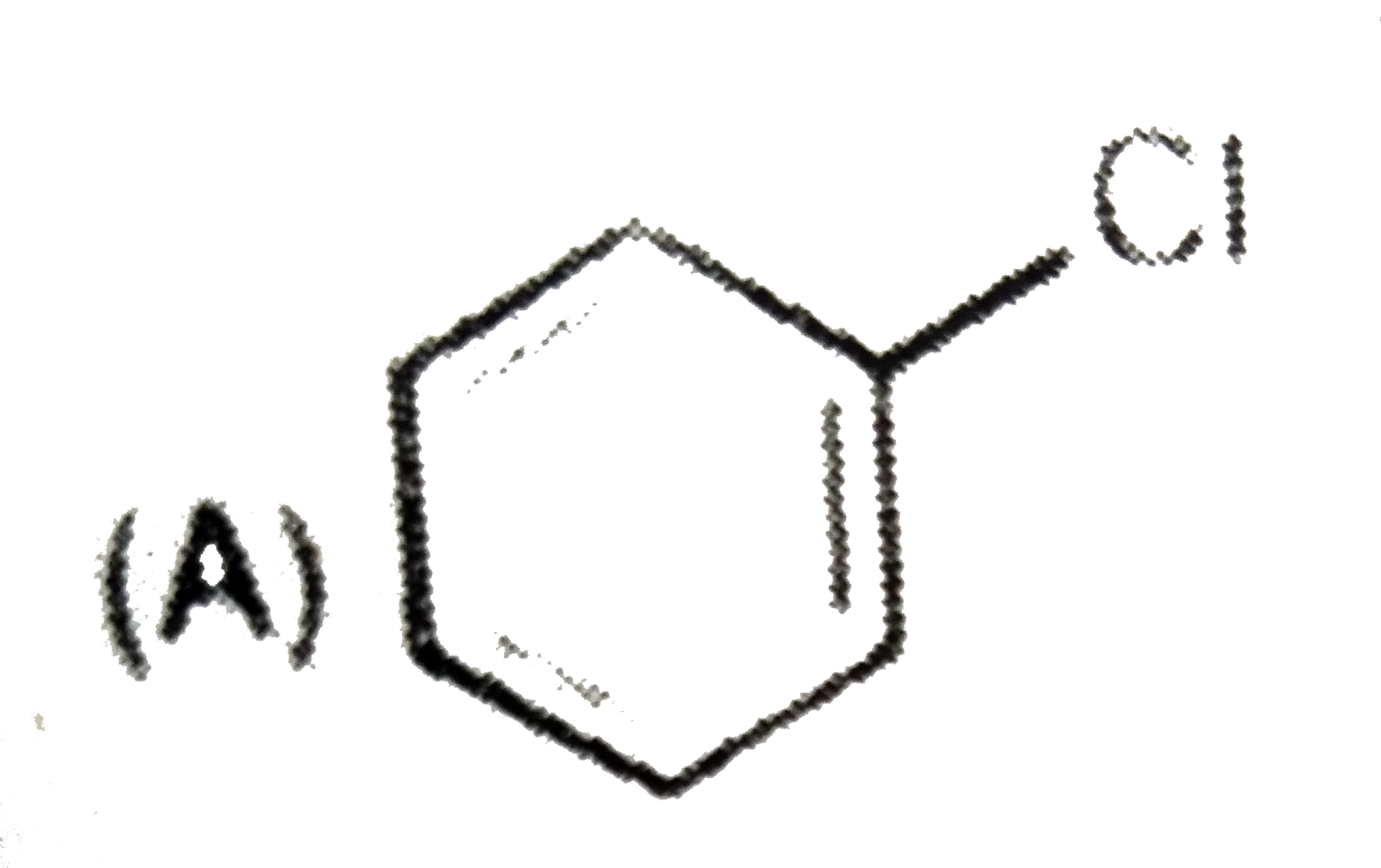
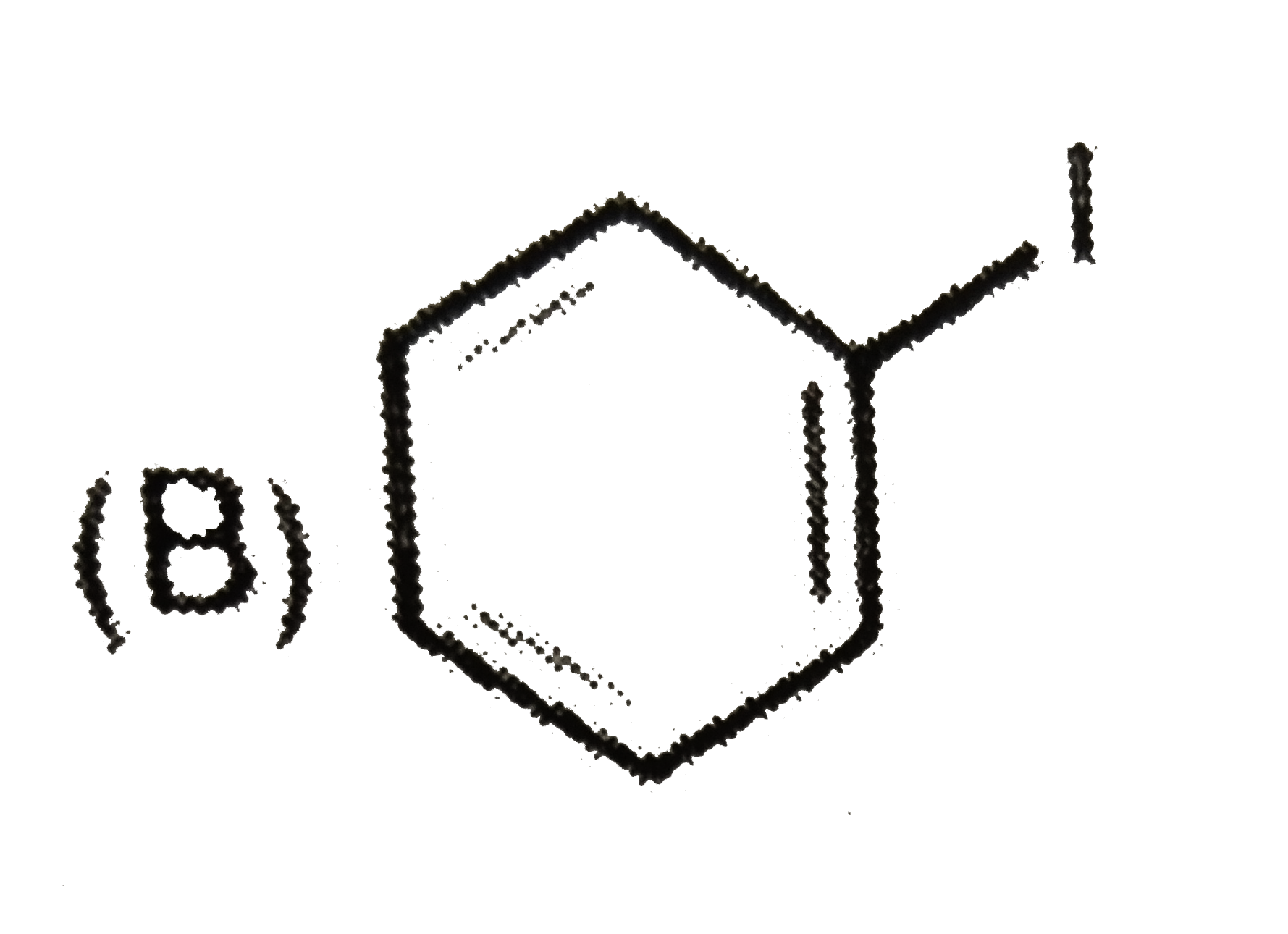
The compound 'X' is |
|
Answer»
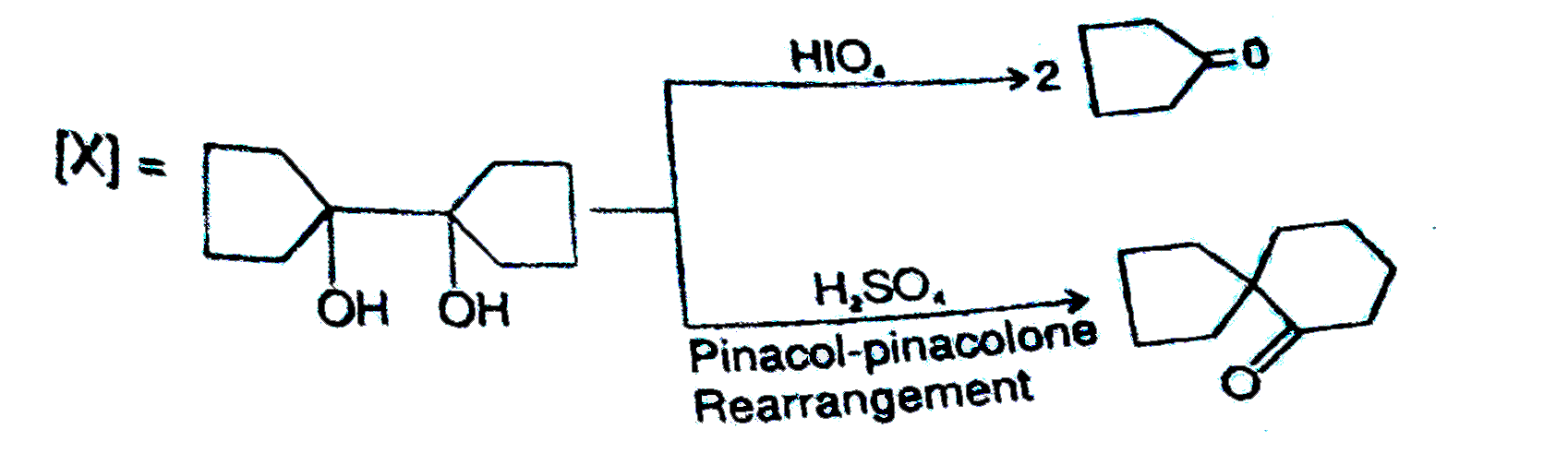
|
|
| 47. |
The compound with the lowest oxidation state of iron is: |
| Answer» Answer :A | |
| 48. |
The compound with the highest boiling point is |
|
Answer» n-hexane |
|
| 49. |
The compound with molecular formula C_8H_10which will give only two isomers on electrophilic substitution with Cl_2//FeCl_3or with HNO_3//H_2SO_4is |
|
Answer» p-dimethylbenzene 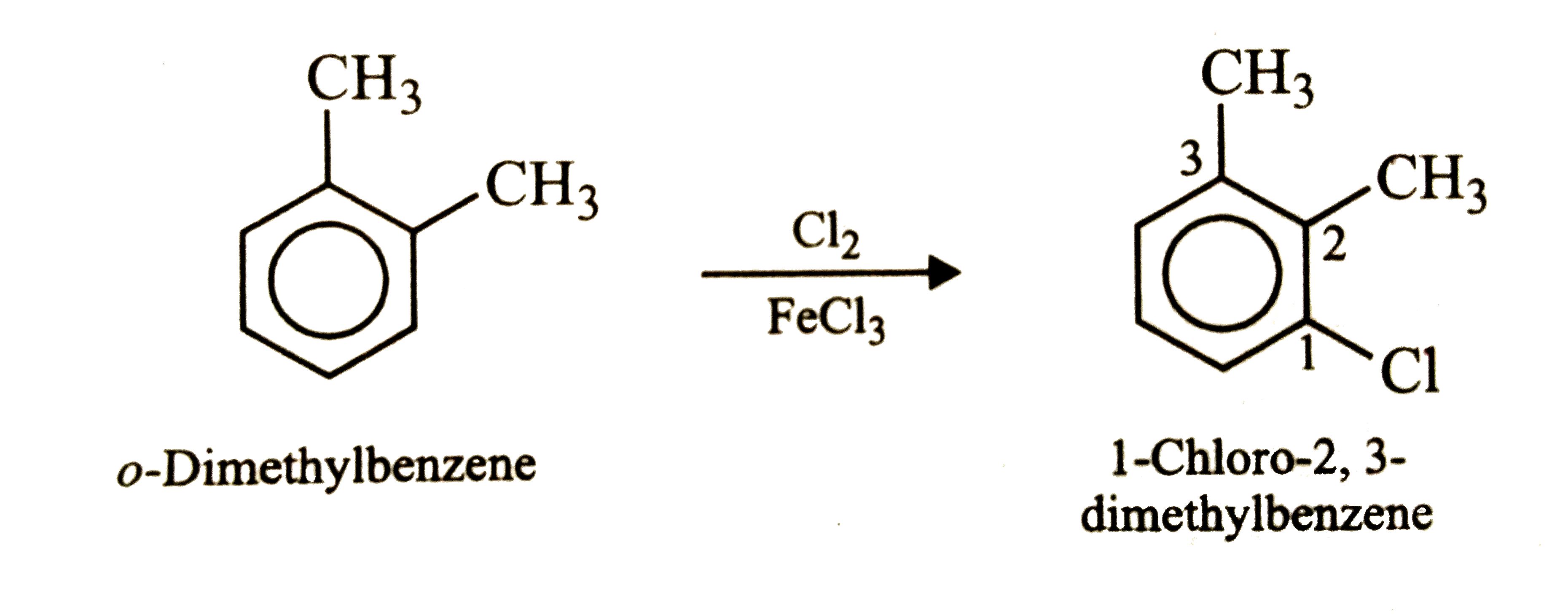 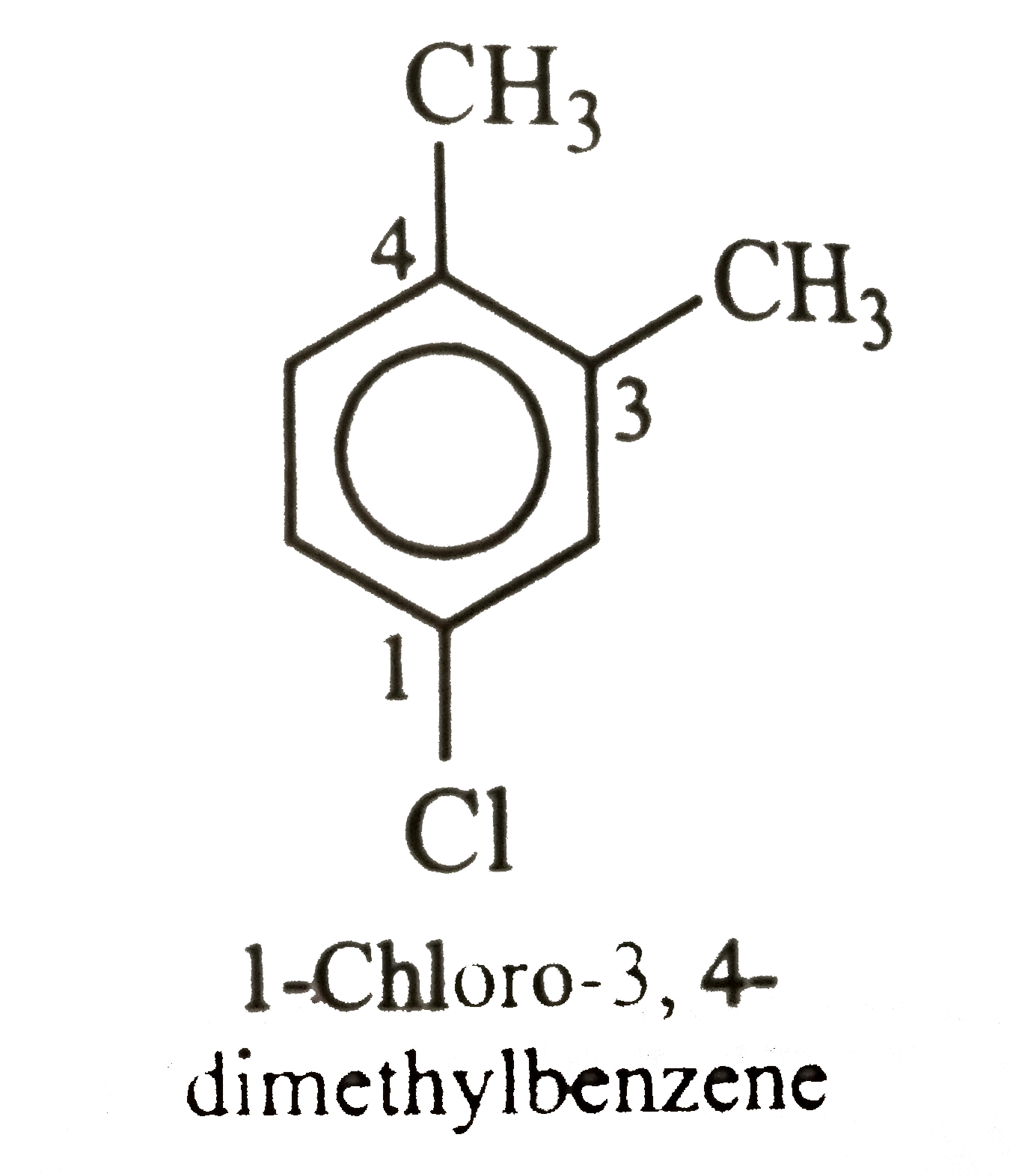
|
|