Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The cylindrical shape of alkyne is due to |
|
Answer» Two sigma `C-C` and ONE `PI C-C` bonds |
|
| 2. |
The cyclopentadienyl cation is _________ while cyclopentadienyl anion is ________ |
|
Answer» |
|
| 3. |
The cubic unit cell of Al (molar mass 27 g mol^(-1)has an edge length of 405 pm. Its density is 2.7 g cm^(-3)). The cubic unit cell is |
|
Answer» face centred `"or "Z=(d TIMESA^(3)timesN_(0)times10^(-30))/M` =`(2.7times(405)^(3)TIMES(6.02times10^(23))10^(-30))/27=4` Hence it is face centred CUBIC unit cell. |
|
| 4. |
The cubic unit cell of a metal (molar mass=63.55 g mol^(-1))has an edge length of 362pm. Its density is 8.92 g cm^(3). The typeof cell is |
|
Answer» FCC `=((8.92 g CM^(-3))times(3.62times10^(-8)cm)^(3)times(6.02times10^(23)MOL^(-1)))/(63.55 g mol^(-1))` `=(8.92times47.44times10^(-24)times6.02times10^(23))/(63.55)` =`4.001` |
|
| 5. |
The crystalline salts of alkaline earth metals contain more water of crystallisation than the corresponding alkali metal salts . Why ? |
| Answer» Solution :Due to SMALLER size and higher nuclear charge , alkaline earth metals have a higher tendency than alkali metals to attract `H_(2)O` molecules and thus contain more water of crystallization than alkali metals . For EXAMPLE, `MgCl_(2) .6H_(2)O` . However , among alkali metals , only lithium because of its small size , forms hydrated SALTS , i.e., `LiCl.2 H_(2)O` | |
| 6. |
The crystalline salt Na_(2)SO_(4) on heating loses 55.9 % of its weight. The formula of the crystalline salt is : |
|
Answer» `Na_(2)SO_(4).5H_(2)O` `((142+18x)u)/(142u)=(100)/(44.1)` `(142 + 18 x)u = (100)/(44.1) xx(142 u) = 321.99u` `18 x = 321.99 - 142 = 179.99` `x = (179.99)/(18)=10` `:.` Formula of crystalline salt `= Na_(2)SO_(4).10H_(2)O`. |
|
| 7. |
The crystaline salts of alkaline earth metals contain more water of crystallisation than the corresponding alkali metal salts. Why? |
|
Answer» SOLUTION :The charge density of alkaline earth metal ions is HIGHER than that of alkali metal ions. The higher the charger density of the CATION, the higher the htydration energy. So, the SALTS of alkaline earth METALS have more water molecules of crytallisation than those of alkali metals. |
|
| 8. |
The crystalline form of borax has |
|
Answer» tetrabnuclear `[B_(4)O_(5)(OH)_(4)]^(2-)` UNIT |
|
| 9. |
The crystal with metal deficiency is |
|
Answer» NaCl |
|
| 10. |
The crystal with metal deficiency defect is |
| Answer» Solution :In FeO, as some Fe may be PRESENT as ` Fe^(3+)`and for charge neutality ` 3 Fe^(2+) = 2 Fe^(3+)`. Hence, FeOshould have metal DEFICIENCY defect. | |
| 11. |
The crystal system of a compound with unit cell dimensions a=0.387, b=0.387 and c=0.504 nm and alpha=beta=90^(@) and gamma=120^(@) is |
|
Answer» cubic |
|
| 12. |
The crystal structure of epsom denotes many types of bonding .Substantiate. |
|
Answer» Solution :Epsom is magnesium sulphate heptahydrate .It is denoted as . `MgSO_(4).7H_(2)O=[MG(H_(2)O)_(6)]SO_(4).H_(2)O` It has six water molecule coordinate with metal ion . One water molecule associated with sulphate , through intermolecular hydrogen bond. It has ionic ,covalent ,coordinate covalent and hydrogen BONDS . |
|
| 13. |
The crystal lattice of NaCl is |
|
Answer» face-centred CUBIC lattice |
|
| 14. |
The critically density of the gas CO_(2) is 0.44 g cm^(-3) at a certain temperature, if r is the radius of the molecule, r^(3) in cm^(3) is approximately: (N is Avogadro number) |
| Answer» Answer :d | |
| 15. |
The critical temperature (T_(C )) and critical pressure (P_(C )) of CO_(2) are 30.98^(@)C and 73 atm respectivelt. Can CO_(2)(g) be liquefied at 32^(@)C and 80 atm pressure ? |
| Answer» Solution :Critical temperature and pressure of `CO_(2)` gas are `30.98^(@)C` and 73 ATM. This suggests that `CO_(2)` gas cannot be liquefied above `30.98^(@)C` and 73 atm howsoever high temperature and pressure may be applied on `CO_(2)` gas. Thus, `CO_(2)` gas can.t be liquefied at `32.^(@)C` and 80 atm. | |
| 16. |
The critical temperature (T_(c )) and critical pressure (p_(c )) of CO_(2)are 30.98^(@)C and 73 atm respectively. Can CO_(2) (g) be liquefied at 32^(@)C and 80 atm pressure ? |
| Answer» Solution :No, `CO_(2)` cannot be LIQUEFIED at `32^(@)C` because this temperature is above its critical temperature. | |
| 17. |
The critical temperature of water is higher than that of O_2 because the H_2O molecule has..... |
|
Answer» FEWER ELECTRONS than oxygen. |
|
| 18. |
The critical temperature of hydrogen gas is 33.2^(@)C and its critical pressure is 12.4 atm. Find out the values of a' and b' for the gas. |
|
Answer» Solution :We know `T_(c) = (8a)/(27 Rb) …. (i) , P_(c) = (a)/(27b^(2)) … (ii)` Dividing (i) by (ii) we get `(T_(c))/(P_(c)) = (8a)/(27Rb) xx (27 b^(2))/(a) = (8b)/(R) …. (iii)` GIVEN `T_(c) = 33.2 ^(@) C = 33.2 + 273 = 306.2 K ` and `P_c = 12.4` atm , R = 0.082 atm litre `K^(-1) mol^(-1)` . SUBSTITUTING the values in EQUATION (iii) , we get `(306.2)/(12.4) = (8 xx b)/(0.082)` `b =(306.2 xx 0.082)/(12.4 xx 8)` `= 0.253` litre `mol^(-1)` Now substituting the value of .b. in equation (i) we have `T_(c) = (8a)/(27 Rb) (or) 306.2 = (8 xx a)/(27 xx 0.082 xx 0.253)` (or) `a = 21.439 atm "litre"^(2) mol^(-1)` |
|
| 19. |
The critical temperature of a substance is defined as : |
|
Answer» The temperature above which the substance decomposses |
|
| 20. |
The critical temperature and the critical pressure of a gas are T_(c) and P_(c), respectively. If the gas exists at a temperature of T and a pressure P, then under which of the following conditions will the gas not be liquefied? (i) T gt T_(c) , P gt P_(c) (ii) T=T_(c),PgtP_(c) (iii) T=T_(c),PltP_(c) (iv) T lt T_(c),P=P_(c) |
|
Answer» Solution :(i) Under the conditions of `T GT T_(c) and P gt P_(c)`, the GAS cannot be liquefied because its temperature is above its critical temperature. (ii) Under the conditions of `T=T_(c) and P gt P_(c)`, it is possible to liquefy the gas. Because the gas is at its critical temperature and its pressure is above critical pressure. (iii) Under the conditions of `T=T_(c) and P lt P_(c)`, it is not possible to liquefy the gas as the MINIMUM pressure needed to liquefy a gas at its critical temperature most be equal to `P_(c)` to greater than `P_(c)`. (iv) Under the conditions of `T lt T_(c) and P=P_(c)`, the gas can be liquefied. because the gas is below its critical temperature and the pressure of the gas is equal to its critical pressure, the minimum pressure REQUIRED to liquefy a gas at its critical temperature. |
|
| 21. |
The critical temperature of a gas is that temperature: |
|
Answer» above which it can no longer REMAIN in the gaseous state |
|
| 22. |
The critical temperature and pressure of CO_(2) gas are 304.2K and 72.9atm respectively Calculate the radius of CO_(2) molecule assumingit to behave van der Waals gas . |
|
Answer» |
|
| 23. |
The critical micellization concentration (CMC) is |
|
Answer» The concentration at which micellization begins |
|
| 24. |
The critical constants for water are 374^(@)C, 218 atm and 0.0566 litre mol^(-1). Calculate a' and b' of water |
|
Answer» Solution :`a = 2.095 lit^(2)` atm `mol^(-1)` `B = 0.0189 lit mol^(-1)` |
|
| 25. |
The criteria for the spontaneityof a process are |
|
Answer» `(dG)_(T,P) lt 0` |
|
| 26. |
The cricital temperature of water is higher than that of oxyget because the water molecule has |
|
Answer» FEWER electrons than oxygen 
|
|
| 27. |
The creosote oil fraction of coal tar distillation contains mainly cresols and naphthalene. |
|
Answer» |
|
| 28. |
The coversion of n-hexane to benzene involves ............ |
| Answer» SOLUTION :DEHYDROGENATION, CYCLISATION | |
| 29. |
The covalent bond length is the shortest in which one of the following bonds ? |
|
Answer» <P>C-O (o-H has SHORTEST bond length due to SMELL size of H atom) |
|
| 30. |
The covalent and van der Waals radii of chlorine respectively are |
|
Answer» `1.80 Å & 0.99 Å` |
|
| 31. |
Which alkaline earth metal chloride is most covalent? |
|
Answer» `BeX_2` |
|
| 32. |
The covalency of nitrogen in HNO_(2) is |
|
Answer» 0 |
|
| 34. |
The covalency of nitrogen in HNO_2is |
|
Answer» 0 |
|
| 35. |
The correction for 'a' to the ideal gas equation corresponds to |
|
Answer» DENSITY of the gas molecules |
|
| 36. |
The correctly reported answer of the addition of 4.523, 2.3 and 6.24 will have significant figures |
|
Answer» Two |
|
| 37. |
The correction factor 'a' to the ideal gas equation corresponds to |
|
Answer» density of the GAS MOLECULES |
|
| 38. |
The corrected term for pressure in the vanderwaal's equation of state is ________. |
|
Answer» <P>(V-b) |
|
| 39. |
The correct valence electronic configuration for Cu(z = 29) is |
|
Answer» `3d^94s^2` |
|
| 40. |
The correct use of lime stone is.... |
|
Answer» to mecrize cotton cloth. |
|
| 41. |
The correct trend of acidic nature of the following alkynes is |
|
Answer» `CH-=CH GT CH_(3) - C-=CH gt CH_(3) C -=C CH_(3)` |
|
| 42. |
The correct thermodynamics conditions for the spontaneous reaction at all temperature is |
|
Answer» `Delta H lt 0` and `Delta SGT 0` |
|
| 43. |
The correct thermodynamic conditions for the spontaneous reaction at all temperatures is |
|
Answer» `Delta H LT 0 and Delta S lt 0` |
|
| 44. |
The correct thermodynamic conditions for the spontaneous reaction at all temperatures is : |
|
Answer» `Delta H GT O and Delta S lt O` If `Delta G = -ve` then, the REACTION will became itself. |
|
| 45. |
The correct thermodynamic conditions for the spontaneous reaction at all temperature is ____ |
|
Answer» `DELTAH LT 0` and `DELTAS GT 0` |
|
| 46. |
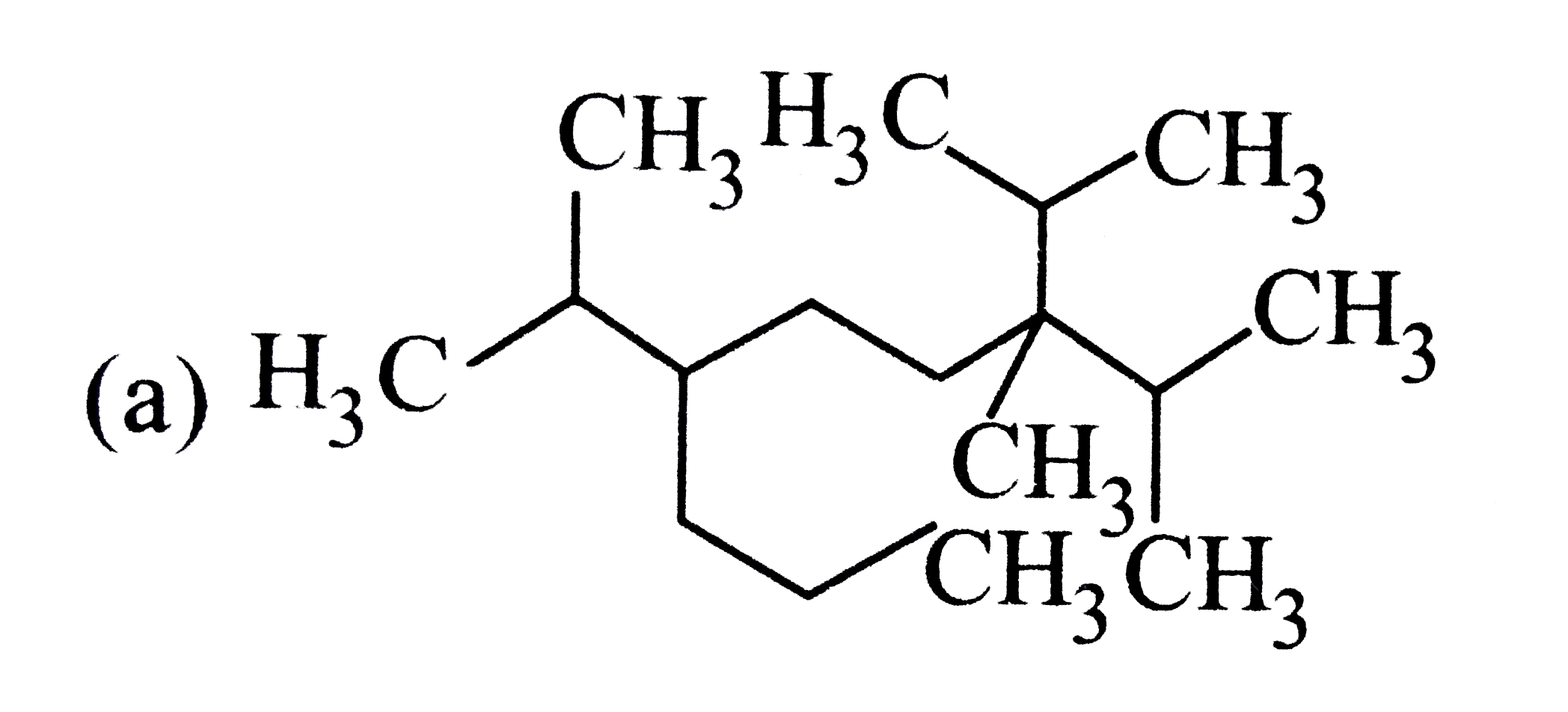
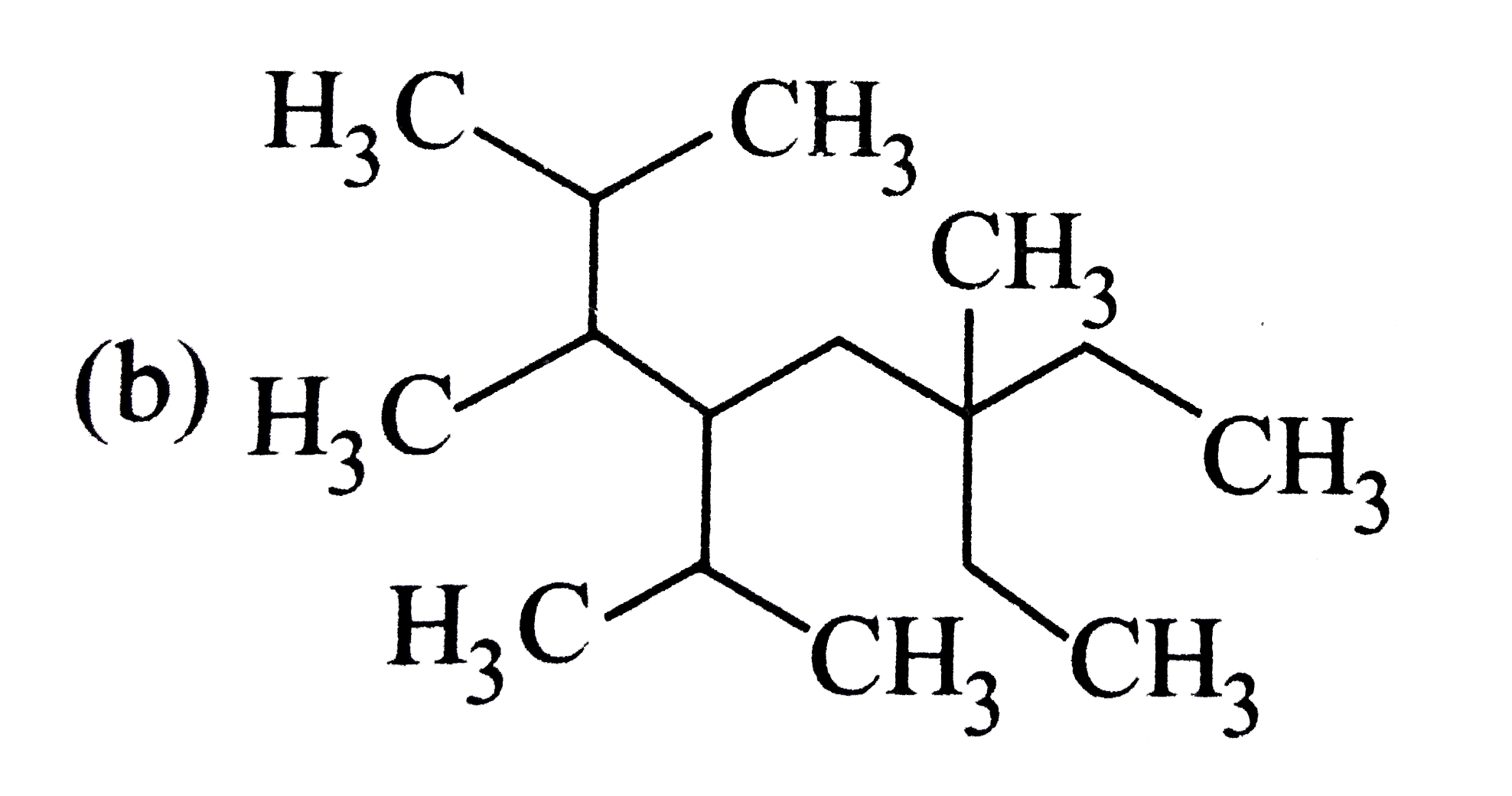
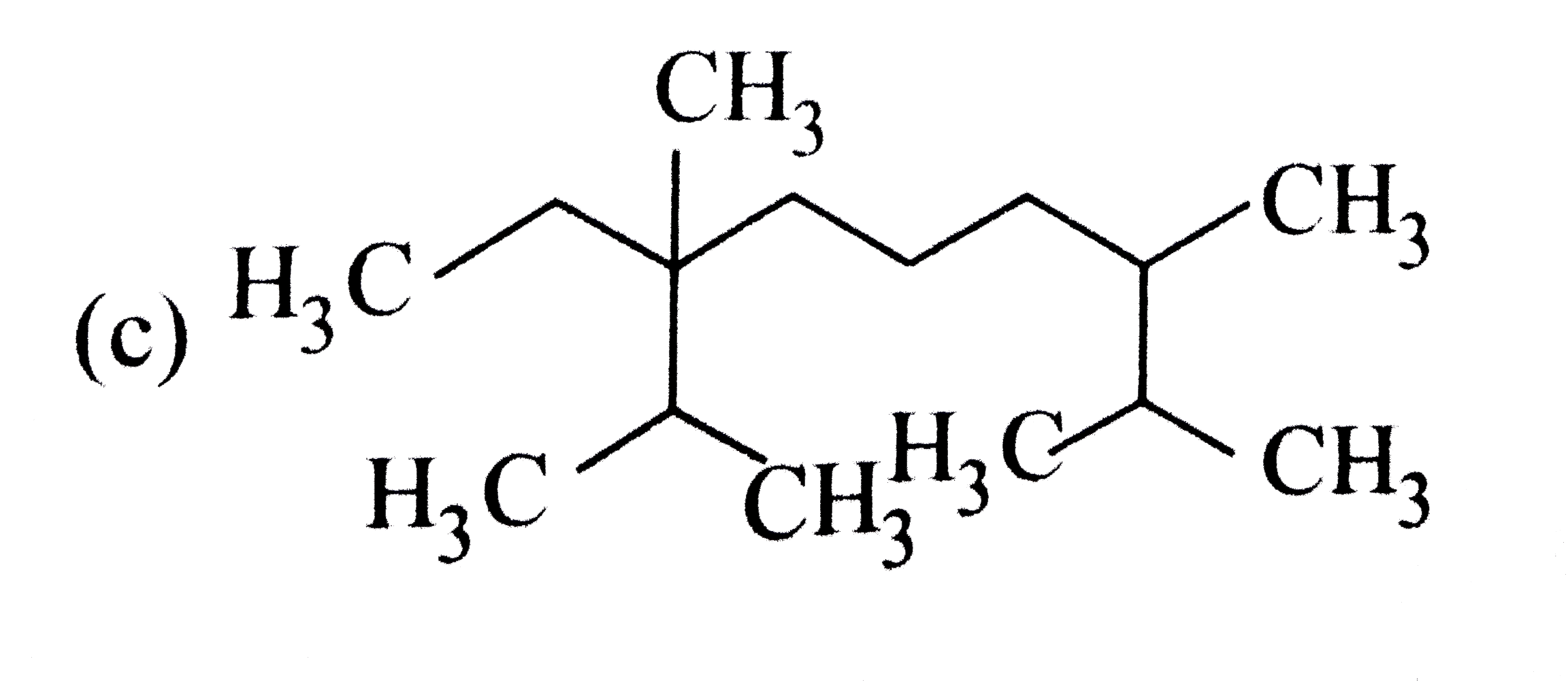
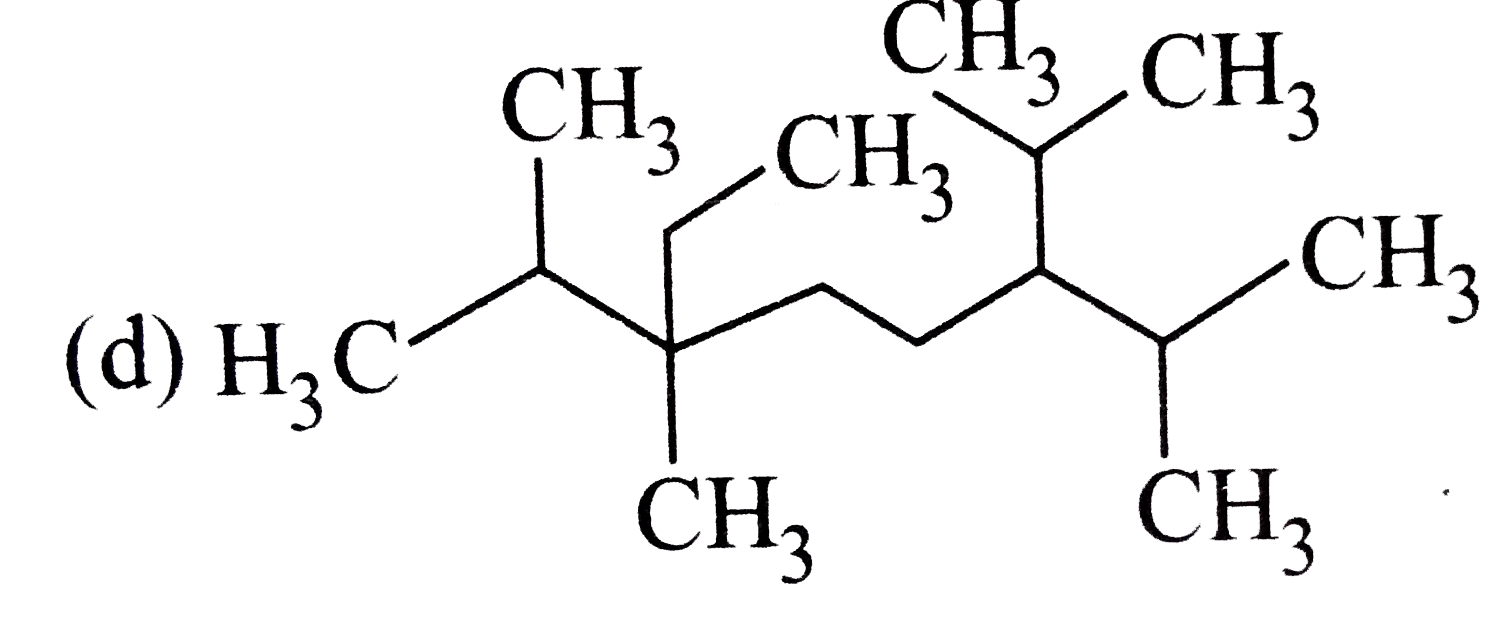
The correct structure of compound 3,6-diisopropyl -2,6- dimethylnonance is |
|
Answer»
 `3-6-`DIISOPROPYL`-2,6-`DIMETHYL NONANE |
|
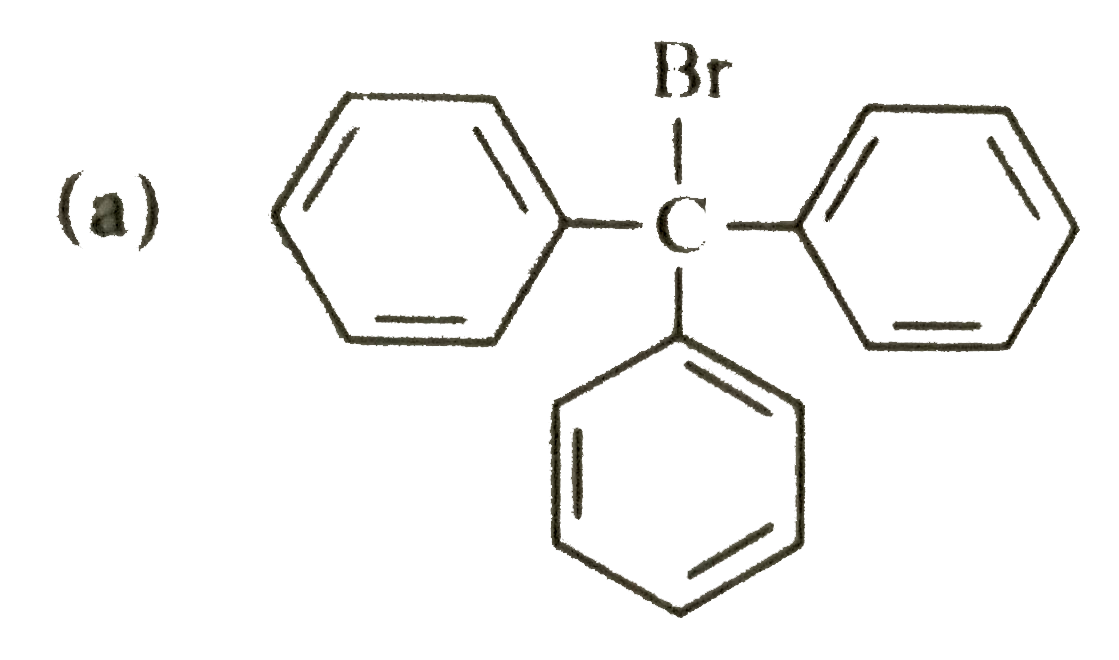
| 47. |
The correct structure for triphenylbromomethane is |
|
Answer»
|
|
| 48. |
The correct structure for (E) 1- bromo-1-chloro-2 fluoro propene is |
|
Answer»
|
|
| 49. |
The correct statement(s) regarding the above reaction is/are |
|
Answer» more than catalytic amounts of `AlCl_(3)` is required for the REACTION |
|
| 50. |
The correct statements regarding kinetic molecular theory are a) The distance between the molecules is high compared to size of the gaseous molecules b) The motion of the gaseous molecules are affected by gravitational force c) The attractive forces between the gaseous molecules are very high. d) The total K.E of a sample of gaseous molecules remains constant at a given temperature |
|
Answer» B, d |
|