Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The degree of dissociation of N_(2) O_(2) into NO_(2) at one atmosphere and 40^(@)C is 0.310. For this : |
|
Answer» `K_(P)` = 0.42 atm Total moles = 0.7 +0.6=1.3 `Kp=(P^(2)NO_(2))/(PN_(2)O_(4))=(((0.6)/(1.3))^(2))/((0.7)/(1.3))=(0.6 xx 0.6)/(1.3 xx 0.7)` `=0.4 atm implies Kp=Kc(RT)^(Delta n_((g)))` |
|
| 2. |
The degree of dissociation of phosphorous pentachloride at certain temperature at 1atm is 0.2. calculate the pressure at which phosphorous pentachloride will be half dissociated at the same temperature. |
| Answer» SOLUTION :0.125 ATM | |
| 3. |
The degree of dissociation of HI at a particular temperature is 0*8. Calculate the volume of2 MNa_(2)S_(2)O_(3) solution required to neutralize the iodine present in the equilibrium mixture of a reaction when 2 mole each of H_(2) and I_(2) are heated in a closed vessel of 2 litre capacity. |
|
Answer» Solution :Degreeof disociation of `HI (alpha) = 0*8` `{:(,2 HI,hArr,H_(2),+,I_(2)),("Before dissociation",1,,0,,0),("Moles after disso",1-alpha,,alpha//2,,alpha//2):} ` `K_(c) = ((alpha//2)(alpha//2))/(1 -alpha)^(2) = alpha^(2)/(4(1 -alpha)^(2) )=(0*8)/(4(1-0*8)^(2))= 4` Step 2. Calculation of `I_(2)`in equilibrium MIXTURE. ` {:(,H_(2),+,I_(2),hArr,2 HI),("Intial moles",2,,2,,0), ("Moles after reaction ",(2-x),,(2-x),,2x//2), ("Molar conc.",(2-x)//2,,(2-x)//2,,2x//2):}` `K'_(c)= 1/K_(c)-(2x//2)^(2)/(((2-x)/2)((2-x)/2))=(4x^(2))/(2-x)^(2)` ` :. (4x^(2))/(2-x)^(2) = 1/4 or (2x)/(2-x) =1/2 or x=2/5 ` Thus, `I_(2) "left" = 2 - 2/5 = 8/5"mole. "` Step 3. Calculation of VOLUME of hypo solution used . `2 Na _(2) S_(2) O_(3) + I_(2) to Na_(2) S_(4) O_(6) + 2 Nal ` ` :. "Moles of " N_(2)S_(2)O_(3)" reacted " = 2 xx 8/5 "mole" = 16/5 "mole " ` Volume of 2 M ` Na_(2)S_(2) O_(3) " reacted " = 1000/2 xx 16/5 = 1600 "mL " = 1*6 L` |
|
| 4. |
The degree of dissociation of an acid HA is 9 times the degree of dissociation of acidHA_(2) of the same concentration, then the ratio of the strengths of acid HA_(1) to HA_(2) will be |
|
Answer» |
|
| 5. |
The degree of dissociation of a weak electrolyte is ...........one whereas that of a strong electrolyte is ............one. |
|
Answer» |
|
| 6. |
The degreeof dissociation is 0*4 at 400 K and 1*0 atm for the gaseous reaction PCl_(5) hArr PCl_(3) + Cl_(2). ,brgt Assming ideal behaviour of all the gases , calculate the density of the equilibrium mixture at 400 K and 1*0 atmosphere . ( Atomic mass of P = 31*0 and Cl = 35*5) |
|
Answer» SOLUTION :` {:(,PCl_(5),hArr,PCl_(3),+,Cl_(2)),("At equilibrium",0*6,,0*4,,0*4 "moles"):} ` Average molecular MASS of the mixture` ( 0*6(31 + 5 xx 35*5 ) + 0*4 (31 + 3 xx 35*5)+ 0*4 (2 xx 35 *5 ))/( 0*6 + 0 *4 + 0*4 )` ` =( 125*1+ 55 + 42*6)/ (1*4)=148* 92` For ideal gases ,`PV = nRT = w/M RT or PM = w/V RT= d RT ` `:.d= (PM)/(RT)= (1xx 148* 92 )/(0* 08205 xx 400 )= 4* 5374 g L^(-1) ` |
|
| 7. |
The degree of dissociation alpha of the reaction" N_(2)O_(4(g))hArr 2NO_(2(g)) can be related to K_(p) as: |
|
Answer» `ALPHA=((K_(p))/(P))/(4+(K_(p))/(P))` |
|
| 8. |
The degree of dissociation of an acid HA in 0.1 M solution is 0.1%. Its dissociation constant is |
|
Answer» Solution :`k_a= Calpha^2` Since dissociation constant of acid, `K_a` is constant, `alpha ` depends UPON concentration. ` alpha^2 PROP (1)/(c )oralphaprop (1)/( sqrt(C )) or (alpha_2)/(alpha_1)= sqrt((c_1)/(c_2))` SUBSTITUTING the VALUES,`(alpha_2)/(0.006) = sqrt((0.1 )/( 0.05 ) )= sqrt(20)` solving thevalues`alpha_2= 0.006 xx sqrt(20 ) ` thedgreeof dissociationin 0.05Msolutionis `0.027` |
|
| 9. |
The degree of dissociation alpha is equal to …………… |
| Answer» SOLUTION :`(i-1)/(n-1)` | |
| 11. |
The decreasing size of K^(+),Ca^(2+),Cl^(-) & S^(2-) follows the order: |
|
Answer» `K^(+)gtCa^(+2)gtS^(-2)GTCL^(-)` |
|
| 12. |
The decreasing order of the first ionization energy in kJmol^(-1) of He, Mg and Na is He > Mg > Na. The increasing order of second ionization energy in of these elements will be |
|
Answer» NA LT MG lt He |
|
| 13. |
The decreasing order of stability of the carbonium ions C_(6)H_(5)CH_(2)^(+) (I), p-(CH_(3)O)C_(6)H_(4)CH_(2)^(+) (II), p-(NO_(2))C_(6)H_(4)CH_(2)^(+) (III), p-(CH_(3))C_(6)H_(4)CH_(2)^(+) (IV) is |
|
Answer» `IV gt II gt I gt III` |
|
| 14. |
The decreasing order of power of boron halides to act as Lewis acids is |
|
Answer» `BF_(3) GT BCI_(3) gt BBr_(3)` |
|
| 15. |
The decreasing order of rates of diffusion of H_2 , N_2 , O_2, and CO_2,is |
|
Answer» `H_2gtN_2gt CO_2gt O_2` |
|
| 16. |
The decreasing order of nucleophilicity among the nucleophiles is (a) CH_(3) underset(O)underset(||)(C) - O^(-) (b) CH_(3)O^(-) (c) CN^(-) (d) |
|
Answer» a, b, c, d 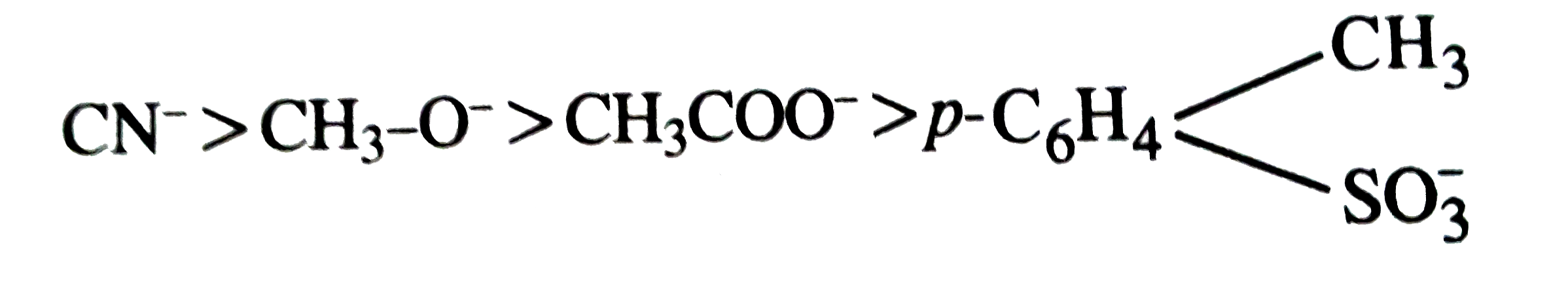
|
|
| 17. |
The decreasing order of electron density on the ring is : |
|
Answer» `IIIgtIIgtI` |
|
| 18. |
The decreasing order of C-C bond length in overset(4)(C )H_(3)- overset(3)(C )H_(2)- overset(2)(C )H= overset(1)(C )H_(2) is…… |
|
Answer» `C_(4) - C_(3) GT C_(2) - C_(1) gt C_(3) - C_(2)` `:. (C_(4)-C_(3)) gt (C_(3)-C_(2)) gt (C_(2)-C_(1))` |
|
| 19. |
The decreasing order of bond length among alkyl halides (CH_3I, CH_3Br, CH_3F, CH_3Cl) is in the order in the order ……………………… . |
| Answer» SOLUTION :`CH_3F LT CH_3Cl lt CH_3 BR lt CH_3I` | |
| 20. |
The decreasing order of bond dissociation energies of C-C,C-H and H -H bonds is |
|
Answer» `H-Hgt-C-Hgt-C-C` |
|
| 21. |
The decreasing order of bond dissociation energies of C-C, C-H and H-H bonds is |
|
Answer» `H - H GT -C-Hgt -C-C ` |
|
| 22. |
The decreasing order of bond angles in ethane, ethylene, acetylene and benzene is |
|
Answer» `C_(2)H_(6)gtC_(2)H_(4)gtC_(2)H_(2)gtC_(6)H_(6)` |
|
| 23. |
The decreasing order of boiling points is |
|
Answer» n-pentane `GT` iso-pentane `gt` neo-pentane |
|
| 24. |
The decreasing order of boiling points among the isomeric pentanes is neo gt iso gt n. |
|
Answer» |
|
| 25. |
The decreasing order of boiling point of haloalkanes CH_3Br, CH_3Cl, CH_3F, CH_3I is ……………….. . |
| Answer» SOLUTION :`CH_3I GT CH_3Br gt CH_3Cl gt CH_3F` | |
| 26. |
The decreasing order of basic character of K_(2)O BaO , CaO and MgO is |
|
Answer» `K_(2)O gt BaO gt CaO gt MGO` Combining the two trends , basic character increases in the order : `K_(2)O gt BaO gt CaO gt MgO` , i.e., option (a) is correct . |
|
| 27. |
The decreasing order of acidity among following compounds, ethanol (I), 2, 3, 2-trifluoroethanol (II), the fluoroacetic acid (III) and acetic acid (IV) is |
|
Answer» `III gt II gt IV gt I` |
|
| 28. |
The decreasing order of acidic character among ethane (I), ethene (II), ethyne (III) and propyne (IV) is |
|
Answer» `(I) gt (II) gt (III) gt (IV)` `UNDERSET((III))(CH-=CH) gt underset((IV))(CH-=C-CH_(3))gt underset((II))(CH_(2)=CH_(2)) gt underset((I))(CH_(3)-CH_(3))` HYDROGEN atoms attached to SP-hybridised CARBON-atom are most acidic, followed by those attached to `sp^(2)`-hybridised carbon-atom, and those attached to `sp^(3)`-hybridised carbon are least acidic. |
|
| 29. |
The decrease in number of neutrons when ._(a)^(x)U changes the ._(b)^(y)Pb is represented as : |
|
Answer» (x-a)+(y+B) No. of neutrons in `._(b)^(y)Pb = (y - b)` `THEREFORE`The CHANGE in no. of neutrons = (x - a) - (y - b) |
|
| 30. |
The decomposition temperature is lowest for |
|
Answer» `BeCO_(3)` |
|
| 31. |
The decomposition of solid ammonium carbamate, (NH_(4))(NH_(2)CO_(2)), to gaseous ammonia and carbon dioxide is an endothermic reaction. 9NH_(4))(NH_(2)CO_(2)))(s)hArr2NH_(3)(g)+CO_(2)(g) (a) When solid (NH_(4))(NH_(2)CO_(2)) is introduced into and evacuated flask at 25^(@)C, the total pressure of gas at equilibrium is 0.3 atm. What is the value of K_(p) "at" 25^(@)C? (b) Given that the decomposition reaction is at equilibrium. how would the following changes affect the total quatity of NH_(3) in the flask once equilibrium is re-established? (i) Adding CO_(2) (ii) Adding (NH_(4))(NH_(2)CO_(2)) (iii) Removing CO_(2) (iv) Increasing the total volume (v) Adding neon (vi) Increasing the temperature |
|
Answer» 2P P Total pressure`=2P+P=0.3` `P=0.1`atm `K_(P)=(2P)^(2)P=4P^(3)` `=4XX10^(-3) atm^(3)` |
|
| 32. |
The decomposition of KClO_3 " to"KCl and O_2on heating is an example of |
|
Answer» Intermolecular redox change It is an EXAMPLE of intermolecular redox change . |
|
| 33. |
The decomposition of HI in closed vessel at 731 K take place. Calculate equilibrium constant . What is the result on the base of problem 7.7 and 7.8 ? |
Answer» Solution : (i) The VALUE of experiment-5, 6 is same as experiment-1, 2, 3, 4. So value of `K_c` at given temp. is constant. (ii) In experiment-5, 6 reaction is CARRIED by taking HI, So equilibrium established in any direction. (III) MOLES of `H_2` per `L^(-1)` = Moles of `I_2` per `L^(-1)` |
|
| 34. |
The decay of mass during nuclear fusion and nuclear fission are respectively: |
|
Answer» `0.1%, 0.231` |
|
| 35. |
The decay constant for an alpha- decay of Th^(232) is 1.58xx10^(-10) sec^(-1). Find out the no. of alpha- decays thatoccur form 1gsample in 365 days. |
|
Answer» |
|
| 37. |
The threshold wavelength for certain metal is lambda _(0). When a light of wavelength(lambda_(0))//(2)is incident on it , the mximum velocity of photelectrons is 10^(6) m//s. If the wavelength of the incident radiation is reduced to (lambda _(0) // (5), then the maximum velocity of the photoelectrons in m//s will be , |
|
Answer» |
|
| 38. |
The de-Brogilc wavelength of an electron in the first Bohr orbit is |
|
Answer» `LAMBDA =(1.23)/(sqrt(m))` `lambda = 1/(sqrt(2xx 1.6 xx 10^(-19) xx v xx 9.11 xx 10^(-31))) = (1.23)/(sqrtV) nm` |
|
| 39. |
The de Broglie wavelength of a tennis ball of mass 60 g moving with a velocity of 10 metres per second is approximately |
|
Answer» |
|
| 40. |
The de Broglie wavelength of a ball of mass 10g moving with a velocity of 10 ms^(-1) is (h = 6.626 xx 10^(-34) Js) |
|
Answer» `6.626 XX 10^(-33) m` |
|
| 41. |
The de Broglie wavelength associated with a moving particle of fixed mass is inversely proportional to |
|
Answer» Its KINETIC energy |
|
| 42. |
The de-Broglie wavelength associated with a matter particle is |
|
Answer» Directly proportional to the MOMENTUM of the PARTICLE |
|
| 43. |
The de Broglie wavelength associated with a ball of mass, 200g and moving at a speed of 5 metres/hour, is in the order of (h = 6.625 xx 10^(-34)Js) |
|
Answer» |
|
| 44. |
The de-Broglie wavelength associated with a ball of mass 1 kg having kinetic energy 0.5 J is |
|
Answer» `6.626 xx 10^(-34) m` `(1)/(2) xx 1KG xx v^(2) = 0.5 J or v^(2) = 1 or v = 1 ms^(-1)` `LAMDA = (h)/(mv) = (6.626 xx 10^(-34) kg m^(2) s^(-1))/(1kg xx 1MS^(-1))` `= 6.626 xx 10^(-34) m` |
|
| 45. |
The de Broglie wavelength of a particle with mass 1 g and velocity 100 m/s is |
|
Answer» `6.63xx10^(-33)m` |
|
| 46. |
The de Broglie wave length of a riffle bullet of mass 2 grams moving with a velocity of 2m/sec of |
|
Answer» `(6.6xx10^(-34))/(2xx2)m` |
|
| 47. |
The de Broglie wave length associated with a particle of mass 1 mg moving with a velocity of 1m/sec is |
|
Answer» `6.63xx10^(-29)m` |
|
| 48. |
The d-orbitals involved in sp^(3)d^(2) hybridisation are (z-axis is the internuclear axis)- |
|
Answer» `d_(X^(2)-y^(2))` |
|
| 49. |
The d-orbital involved in the hybridization in PCl_(3) molecule is |
|
Answer» <P>`3d_(x^(2) - y^(2))` orbital involved in `D_(z)^(2)` . |
|