Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The group of elements with highest second ionisation energy is |
| Answer» Answer :D | |
| 2. |
The group having isoelectronic species i s .... |
|
Answer» `O^(2-),f^(-),Na^(+).MG^(2+)`  NOTE : Option A isoelectronic in species .  `therefore` option(B,C,D) are notisoelectronic species |
|
| 3. |
The group having isoelectronic species is |
|
Answer» `O^(2-) , F^(-), Na, Mg^(2+)` `Na^(+) = 11 - 1 = 10 e^(-), Mg^(2+) = 12 - 2 = 10 e^(-)` Hence, GROUP (C) contains isoelectronic species. |
|
| 4. |
The ground state term symbol for an electronic state is governed by |
|
Answer» HEISENBERG's PRINCIPLE |
|
| 5. |
The ground-state electronic configurations listed here are incorrect. Explain what mistake have been made in each and write the correct electronic configurations Al : 1s^(2) 2s^(2) 2p^(4) 3s^(2) 3p^(3) " " B : 1s^(2) 2s^(2) 2p^(5) " " F : 1s^(2) 2s^(2) 2p^(6) |
|
Answer» Solution :In `._(13)Al, 2p` should be filled first before FILLING of 3S starts Correct ELECTRONIC CONFIGURATION `= 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(1)` In `._(5)B`, TOTAL electrons = 5. Hence, E.C. `= 1s^(2) 2s^(2) 2p^(1)` In `._(9)F`, total electrons = 9. Hence, E.C. `= 1s^(2) 2s^(2) 2p^(5)` |
|
| 6. |
The ground state electronic configuration of CO molecule |
|
Answer» `1sigma^2 2sigma^2 1pi^4 3sigma^2` 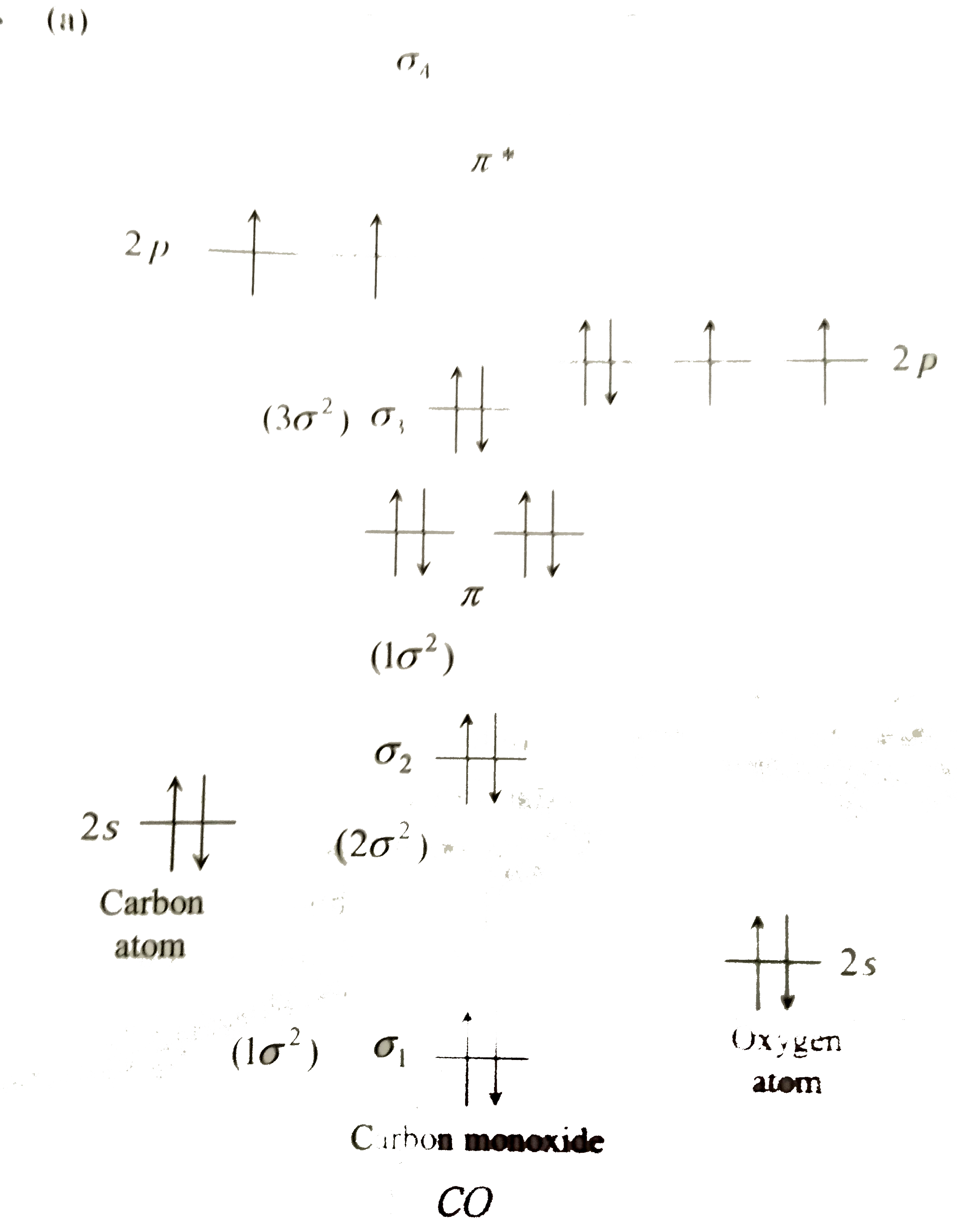 E.C for CO : `1sigma^2 2sigma^2 1pi^4 3sigma^2` |
|
| 7. |
The ground state electron configurations listed here are incorrect. Identify the mistakes that have been made in each and write the correct electronic configurations. Cl, 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) |
| Answer» Solution :Atomic number of CHLORINE is 17, so its ELECTRONIC configuration WOULD be `1s^2 2^s 2p^6 3s^2 3p^5.` The configuration MENTIONED above is that of Argon. | |
| 8. |
The ground state electron configurations listed here are incorrect. Identify the mistakes that have been made in each and write the correct electronic configurations. Al, 1s^(2) 2s^(2) 2p^(4) 3s^(2) 3p^(3) |
| Answer» Solution :3S orbital cannot be filled before complete FILLING of 2P orbital. So, this configuration is INCORRECTLY WRITTEN. | |
| 9. |
The ground state electron configurations listed here are incorrect. Identify the mistakes that have been made in each and write the correct electronic configurations. B: 1s^(2) 2s^(2) 2p^(5) |
| Answer» SOLUTION :ATOMIC number of boron is 5, so its electron configuration would be `1s^2 2^s 2p^1,` the configuration written above is that of fluorine. | |
| 10. |
Why is cement clinker obtained from rotary Kiln mixed with gypsum? |
| Answer» SOLUTION :CEMENT CLINKER | |
| 12. |
The green house effect is because of the |
|
Answer» PRESENCE of GASES, which in general are strong infrared absorbers, in the atmosphere. |
|
| 14. |
The green coloured borax bead obtained from copper salts is |
|
Answer» CUPRIC METABORATE 
|
|
| 15. |
The 'green' chemical used in household cleaners to remove stains and also a favourite dressing on salads is |
|
Answer» hydrochloric ACID |
|
| 16. |
The greatest percentage of CO isin |
|
Answer» coal GAS Coal gas = 3% CO produce gas = 33% CO |
|
| 17. |
The greatest percentage of CO is in |
|
Answer» coal GAS Coal gas = 3% CO PRODUCE gas = 33% CO |
|
| 19. |
The gravitational work done by an object is ………… |
|
Answer» Qv |
|
| 20. |
The graph relates in K_(eq) Vs 1/T for a reaction. The reaction must be : |
|
Answer» exothermic `:.` slope= `(-DeltaH^(@))/(R)` = .+. Ve `IMPLIES DeltaH^(@) LT 0` `:.` exothermic |
|
| 22. |
The graph of viscosity coefficient (T|) and absolute temperature (T) is ________ |
|
Answer» STRAIGHT line passing through origin |
|
| 23. |
The graph of atomic number (Z) Vs atomic radius (r) is shown in figure 1, 2, 3 and 4 Which graph correctly represents the element of 2^(nd) period and 1^(st) group respectively. choose your options from below. |
|
Answer» 1, 3  Atomic radius increases with atomic number in a GROUP 
|
|
| 24. |
Thegraph of P vs V is given at temperature and number of moles : The correct relationship is/are |
|
Answer» `T_(1) GT T_(2) gt T_(3)` |
|
| 25. |
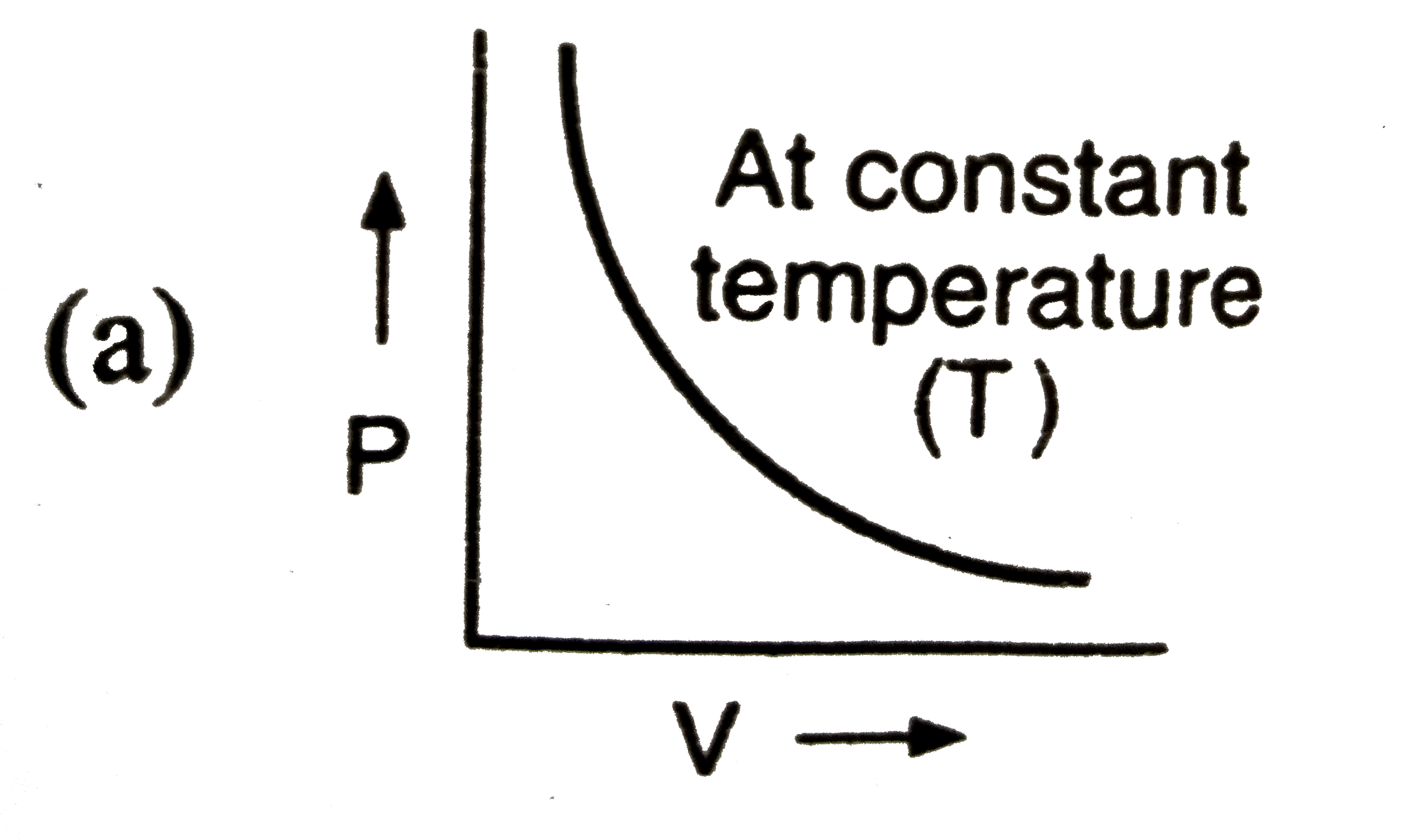
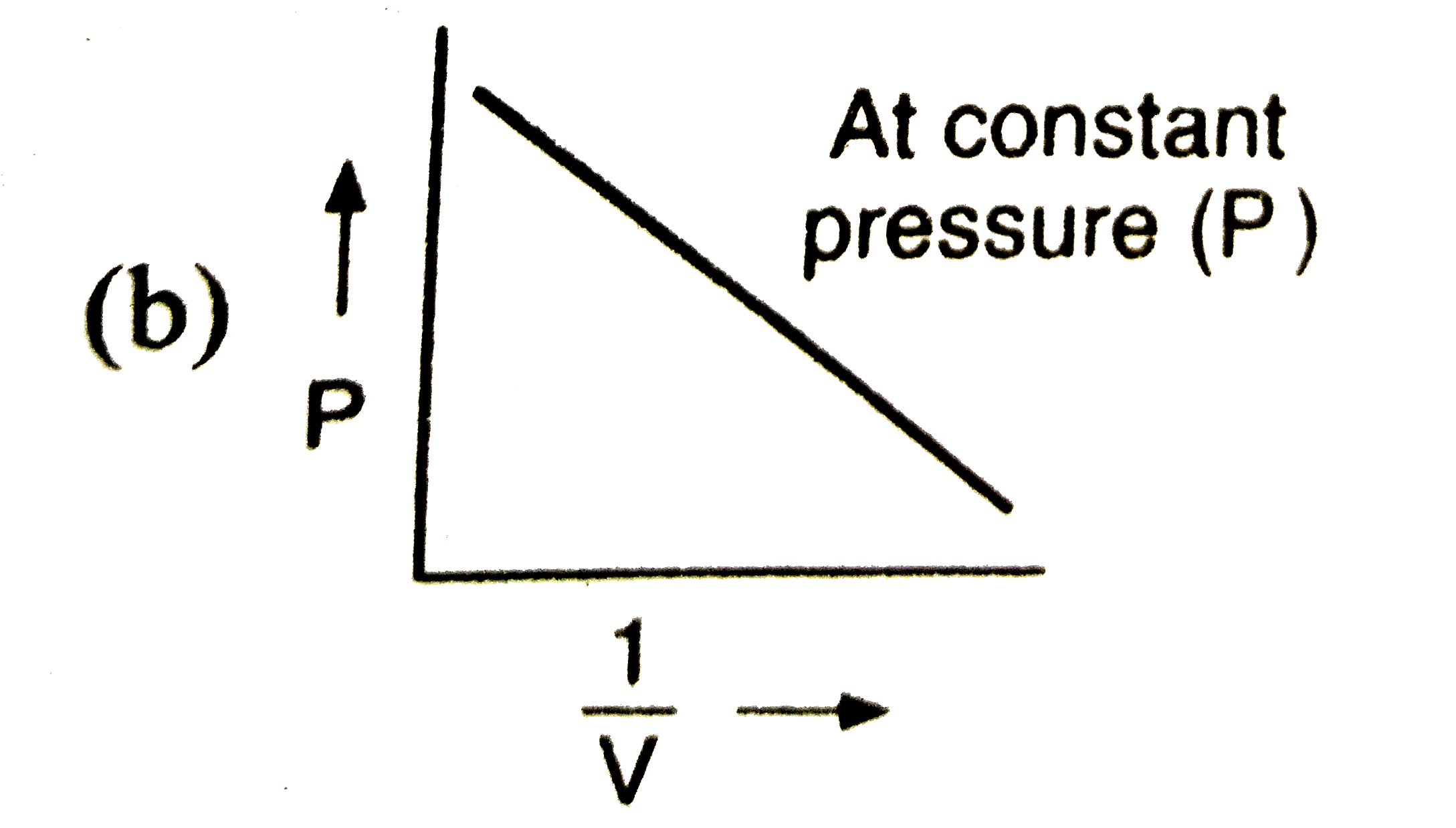
The graph ………… is known as isothermal. |
|
Answer» `p to V` |
|
| 26. |
The graph between momentum p and de-Broglie wavelength lambda of photon is |
|
Answer»
|
|
| 27. |
The grade of 2.95 M H_2O_2 in volume ofpercentage is ____ and ____ respectively. |
|
Answer» 33,10 =2.95 x 11.2 = 33.04 (VOLUME) %w/v = `(Mxx34)/10 = (2.95xx34)/10`=10.03 %w/v |
|
| 28. |
The gold number of some colloids are given below: {:("Colloid","Gold number"),(A,0.01),(B,2.5),(C,20):} The protective nature of these colloids follows the following order |
|
Answer» `C gt B gt A` |
|
| 29. |
The given following elements belongs to which block? Which of this is having the maximum metallic property ? K, Na , Li , Rb |
| Answer» Solution : Rb :(They are ALKALI METAL and top to BOTTOM METALLIC property increases.) | |
| 30. |
The given figure shows a chnge of state A to state C by rtwo paths ABC and AC for an ideal gas. Calculate. a. The path along which work done is least. b. The internal enegry at C if the internal enegry of gas at A is 10J and amount of heat supplied to change its state to C through the path AC is 200J. c. The amount of heat supplied to the gas to go from A to B, if inetrnal enegry of gas at state B is 10J. |
|
Answer» Solution :We know, Path `CA:` Isothermal compression Path `AB`: Isobaric expansion Path `BC`: Isochroic change Let `V_(i)` and `V_(f)` be initial volume and final volume at repective points. For temperature `T_(1) ("for" C): PV - nRT_(1)` `2 xx 10 = 1 xx 0.0821 xx T_(1)` `T_(1) = 243.60K` For temperature `T_(2)` (For `C` and `B): (P_(1)V_(1))/(T_(1)) = (P_(2)V_(2))/(T_(2))` `(2xx10)/(T_(1)) = (20 xx 10)/(T_(2))` `(T_(2))/(T_(1)) = 10` `T_(2) = 243.60 xx 10 = 2436.0K` Path `CA: w = + 2.303 nRT_(1) "log"(V_(i))/(V_(f))` `= 2.303 xx 1xx2xx 243.6"log" (10)/(1)` `=+1122.02 CAL` `DeltaE = -0` for isotermal compression, also `Q = W` Path `AB, w =-P (V_(f) - V_(1))` `=- 20 xx (10 - 1) =- 180 L-atm` `= (-180 xx 2)/(0.0821) =- 4384.9 cal` Pth `BC: w =- P(V_(f) - V_(1)) = 0` (`:' V_(f) - V_(1) = 0)` since volume is constant For monoatomic gas, heat chnage at constant volume `=qv = DeltaU` Thus, for path `BC, q_(v) = C_(v) xx n xx DeltaT = DeltaU` `:.q_(v) = (3)/(2)R xx1 (2436 - 243.6)` `= (3)/(2) xx2xx1xx (2192.4) = 65772.2 cal` Since process involves cooling `:.q_(v) = DeltaU =- 6577.2 cal` Also in path `AB`, the initial enegry in state `A` and state `C` is same. Thus,, during path `AB`, an INCREASE in internal enegry equivalent of change in internal enegry during path. `BC` should take place. Thus, `DeltaU` for path `AB = + 6577.2 cal` Now `q` for path `AB = DeltaU - w_(AB) = 6577.2 + 6577.2 cal` `= 10962.1 cal` Cycle: `DeltaU = 0`, `q =- w =- [w_(path CA) +w_(pathAB) +w_(pathBC)]` `=- [+1122.02 +(-4384.9+0]` `:. q =- w = + 3262.88 cal` |
|
| 31. |
The given figure shows the Maxwell distribution of molecular speeds of a gas at three different temperatures T_1, T_2 and T_3. The correct order of temperatures is: |
|
Answer» `T_1 GT T_2 gt T_3` |
|
| 32. |
The given electronic configuration of C in excited state This configuration can form |
|
Answer» TWO `PI` bond only Two `sigma` and two `pi` bonds |
|
| 33. |
The Gibb's energy for the decomposition of Al_(2)O_(3) at 500^(@)C is as follows (2)/(3)Al_(2)O_(3) rarr (4)/(3)Al+O_(2) Delta_(f) = 960 kJ mol^(-1) The potential difference needed for the electrolytic reduction of aluminium oxide (Al_(2)O_(3)) at 500^(@)C us at least |
|
Answer» `4.5 V` `{:("At anode: " 2O^(2-)+4e^(-)rarrO_(3)"]" xx 3),("At anode: " Al^(3+) rarr Al + 3e^(-)"]" xx 4),(ul(BAR("Net REACTION:" 4AL^(3+)+6O_(2-) rarr 3O_(2)+4Al))),(or 4//3 Al^(3+) + 2O^(2-) rarr O_(2)+4//3Al):}` `:. n = (12)/(3) = 4` `Delta G^(@) = -NFE^(@)` `960 xx 1000 = -4 xx 96500 xx E^(@)` [Here `Delta G^(@) = +960 kJ mol^(-1) = 960 xx 1000 J mol^(-1), n = 4, F = 96500 "coulomb mol"^(-1)]` or `E^(@) = -2.48 V` Potential difference needed `= 2.5 V` |
|
| 34. |
The geometry of H_(3)O^(+) ion is |
|
Answer» Planar |
|
| 35. |
The geometry of H_(2)S and its dipole moment are |
|
Answer» ANGULAR and non-ZERO |
|
| 36. |
The geometry of 'C' in diamond is |
|
Answer» Planar |
|
| 37. |
The geometry of a molecule depends upon the repulsions between electron pairs present in the valance shell of the central atom. LP - LP repulsions are greater than LP-BP which are greater than BP - BP. A pi bondcan not decide the geometry of a molecule because pi bond has no independent exsistance. Deviations in bond angle are due to the presence of lone pairs. In which of the following orientation of electron pairs around the central atom is tetrahedral ? |
|
Answer» `NH_3` |
|
| 38. |
The geometry of a molecule depends upon the repulsions between electron pairs present in the valance shell of the central atom. LP - LP repulsions are greater than LP-BP which are greater than BP - BP. A pi bondcan not decide the geometry of a molecule because pi bond has no independent exsistance. Deviations in bond angle are due to the presence of lone pairs. Square planar species among the following is |
|
Answer» `XeF_4` |
|
| 39. |
The geometry of a molecule depends upon the repulsions between electron pairs present in the valance shell of the central atom. LP - LP repulsions are greater than LP-BP which are greater than BP - BP. A pi bondcan not decide the geometry of a molecule because pi bond has no independent exsistance. Deviations in bond angle are due to the presence of lone pairs. Which of the following molecule is linear in shape ? |
|
Answer» `I_(3)^(-)` |
|
| 40. |
The geometry of a molecule depends upon the repulsions between electron pairs present in the valance shell of the central atom. LP - LP repulsions are greater than LP-BP which are greater than BP - BP. A pi bondcan not decide the geometry of a molecule because pi bond has no independent exsistance. Deviations in bond angle are due to the presence of lone pairs. Which of the following differs from the rest in geometry ? |
|
Answer» `BF_3` |
|
| 41. |
The geometry of a complexspecies can be understood from the knowledge of type ofhybridisation of orbitalsof central atoms. The hybridisation of orbitals of central atom. The hybridisationof orbitalsof centralatom in [Be(OH)_(4)]^(-) and the geometry of the complex are respectively |
|
Answer» `SP^(3)`, TETRAHEDRAL |
|
| 42. |
The geometry of a complex species can be understood from the knowledge of type of hybridization of orbitals of central atom. The hybridization of orbitals of central atom in [Be(OH)_4]^(-)and the geometry of the complex are respectively .......... |
|
Answer» `sp^3` , tetrahedral |
|
| 43. |
The geometrical shape of carbocation is |
|
Answer» LINEAR |
|
| 44. |
The geometrical shape of carbocation is ............. |
|
Answer» Linear |
|
| 45. |
The general trend in the properties of elements of carbon family shows that, with the rise in atomic number. |
|
Answer» The tendency TOWARDS catenation INCREASES |
|
| 46. |
The generalouter electronicconfigurationoftransitionmetals is |
|
Answer» `NS^(2)nd^(1-10)` |
|
| 47. |
The generaloutershellelectronicconfiguration of d- block element is …….. |
| Answer» SOLUTION :`(n-1) d^(1-10) NS^(0-2) ` where `n=4 -7` | |
| 48. |
The general molecular formula which represents the homologousseries of alkanols is |
|
Answer» `C_(N)H_(2N+2)O` |
|
| 49. |
The general formula of primary alcohol is: |
|
Answer» `GT CHOH` |
|