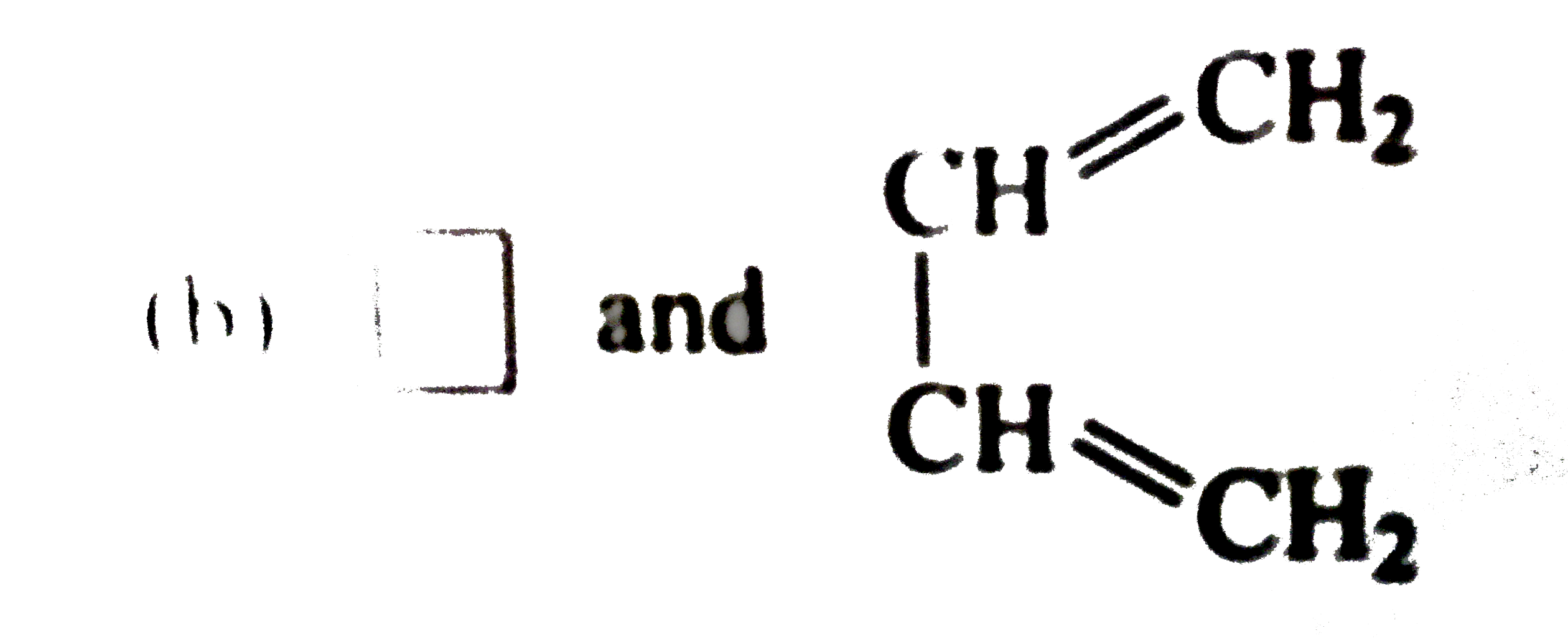Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The pair(s) of reagents that yield paramagnetic species is/are |
|
Answer» Na and excess of `NH_(3)` A `M+(x+y)NH_(3)rarr[M(NH)_(3)_(x)]^(+)+[e(NH)_(3)_(y)]^(-)` B `K+O_(2)rarrKO_(2)[k^(+),O_(2)^(-)`PARAMAGNETIC C `3Cu+8HNO_(3) rarr3 Cu(NO_(3))+_(2)+2NO+4H_(2)O` |
|
| 2. |
The pair(s) of ions where BOTH the ions are precipitated upon passing H_(2)S gas in presence of dilute HCl is (are) |
|
Answer» `BA^(2+), ZN^(2+)` |
|
| 3. |
The pairs of bases in nucleic acids are held together by |
|
Answer» HYDROGEN bonds |
|
| 4. |
The pairs of bases in DNA are held together by |
|
Answer» HYDROGEN BONDS |
|
| 5. |
The pair whose both species are used in acid medicinal preparation is: |
|
Answer» `NaHCO _(3) andMg (OH)_(2)` |
|
| 6. |
The pair (s) where both the ions are precipitated upon passing H_(2)S gas in the presence of dilute HCl is (are) : |
|
Answer» `BA^(2+) , Zn^(2+)` |
|
| 7. |
The pair of substances that has no hydrogen bonding between them is |
|
Answer» `CH_3 COOH ,H_2 O` |
|
| 8. |
The pair of substances that has hydrogen bonding between them is |
|
Answer» `C_(2)H_(4),H_(2)o` |
|
| 9. |
The pair of structure that are resonance hybrid is: |
|
Answer» `Hoverset(..)underset(..)O-OVERSET(+)(C)HCH_(3)` and `Hoverset(+)underset(..)(O)=CHCH_(3)` |
|
| 10. |
The pair of species with the same bond order is : |
|
Answer» `O_(2)^(2-), B_(2)` ` NO^(+) = 3 NO = 2.5 , CO = 3, N_(2) = 3, O_(2) = 2 ` THUS , `O_(2)^(2-) and B_(2)` have tha same bond order viz 1. |
|
| 11. |
The pair of species with the same bond order is |
|
Answer» `NO, CO` |
|
| 12. |
The pair of molecules forming strongest inter molecular hydrogen bonds are |
|
Answer» `SiH_4 and SiF_4` |
|
| 13. |
The pair of ions having same electronic configuration is..... |
|
Answer» `Cr^(3+) , Fe^(3+)` THUS, `Fe^(3+) and Mn^(2+)` have the same ELECTRONIC CONFIGURATION |
|
| 14. |
The pair of elements with the following atomic numbers are chemically similar |
| Answer» Answer :C | |
| 15. |
The pair of elements with same electronegatively and stardard oxidation potential are |
|
Answer» Mg and CA |
|
| 16. |
The pair of elements do not impart flame colour test |
|
Answer» `Be & CA` |
|
| 17. |
The pair of compunds in which both the metals are in the highest possible oxidation state is: |
|
Answer» `[FE(CN)_(6)]^(3-) and [CO(CN)_(6)]^(3-)` |
|
| 18. |
The pair of compounds which cannot exist together is |
|
Answer» `NaHCO_(3)` and NaOH |
|
| 19. |
The pair of compounds which cannot exist together in solution is |
|
Answer» `NaHCO_(3)` and NaOH |
|
| 20. |
The pair of compounds which cannot exist together in aqueous solution is |
|
Answer» `Na_(2)CO_(3)` and `NaHCO_(3)` `NaHCO_(3)+NaOH rarrNa_(2)CO_(3)+H_(2)O` `2NAOH + NaH_(2)PO_(4)rarrNa_(3)PO_(4)+2H_(2)O` |
|
| 21. |
The pair of compounds which cannot exist together in aqueous solution are |
|
Answer» `NaH_(2)PO_(4)` and `Na_(2)HPO_(4)` |
|
| 22. |
The pair of compounds that will not react with each other in an aqueous solution, at room temperature is |
|
Answer» `FeCl_(3),SnCl_(2)` |
|
| 23. |
The pair of compounds that can exist together is : |
|
Answer» `FeCl_(3),SnCl_(2)` |
|
| 24. |
The pair of compounds that can exist together is |
|
Answer» `FeCI_(3),SnCI_(2)` |
|
| 25. |
The pair of compounds having metals in their highest oxidation state is: |
|
Answer» `MnO_(2) and CrO_(2)Cl_(2)` |
|
| 26. |
The pair of compounds having metals in their highest oxidation state is |
|
Answer» `MnO_(2)FeCI` |
|
| 27. |
The pair of compounds having metals in their highest oxidation state is "________________". |
|
Answer» `MnO_(2) , FeCl_(3)` |
|
| 28. |
The pair of compound which cannot exit together in solution is |
|
Answer» `NaHCO_(3) and H_(2)O` |
|
| 29. |
The pair of boiling point and compound ar given as, {:(C_6H_6, CH_3OH, C_6H_5NO_2, C_6H_5NH_2),(80^@C, 65^@C, 212^@C, 184^@C),(I,II,III,IV):} Which will show lowest vapour pressure at room temperature |
|
Answer» `C_6H_6` |
|
| 30. |
The pair of atomic numbers which represent the p-block elements |
|
Answer» 6,12 |
|
| 31. |
The pair o f ions configuration is .... |
|
Answer» `Cr^(3+), Fe^(3+)`  So, `Fe^(3+) and Mn^(2+)`have the same ELECTRONIC configureation. |
|
| 32. |
The pair of amphoteric hydroxides is |
|
Answer» `Be(OH)_(2)`,`Al(OH)_(3)` |
|
| 33. |
The pair in which phosphorous atoms have a formal oxidation state of +3 is |
|
Answer» orthophosphorous acid and pyrophosphorous acids Pyrophosphorus acid, `H_(4)P_(2)O_(5)(+3)`, Hypophosphoric aicd, `H_(4)P_(2)O_(6)+(4)` and Pyrophosphoric acid, `H_(4)P_(2)O_(7)(+5)`. |
|
| 34. |
The pair in which phosphorusatoms have a formal oxidation state of +3is |
|
Answer» orthophosphorus and pyrophosphorus acid LET the oxidation NUMBER of P in pyrophosphorus acid be x. So, 4(+1)+2x+5(-2)=0 or ,2x=6 or, x=+3 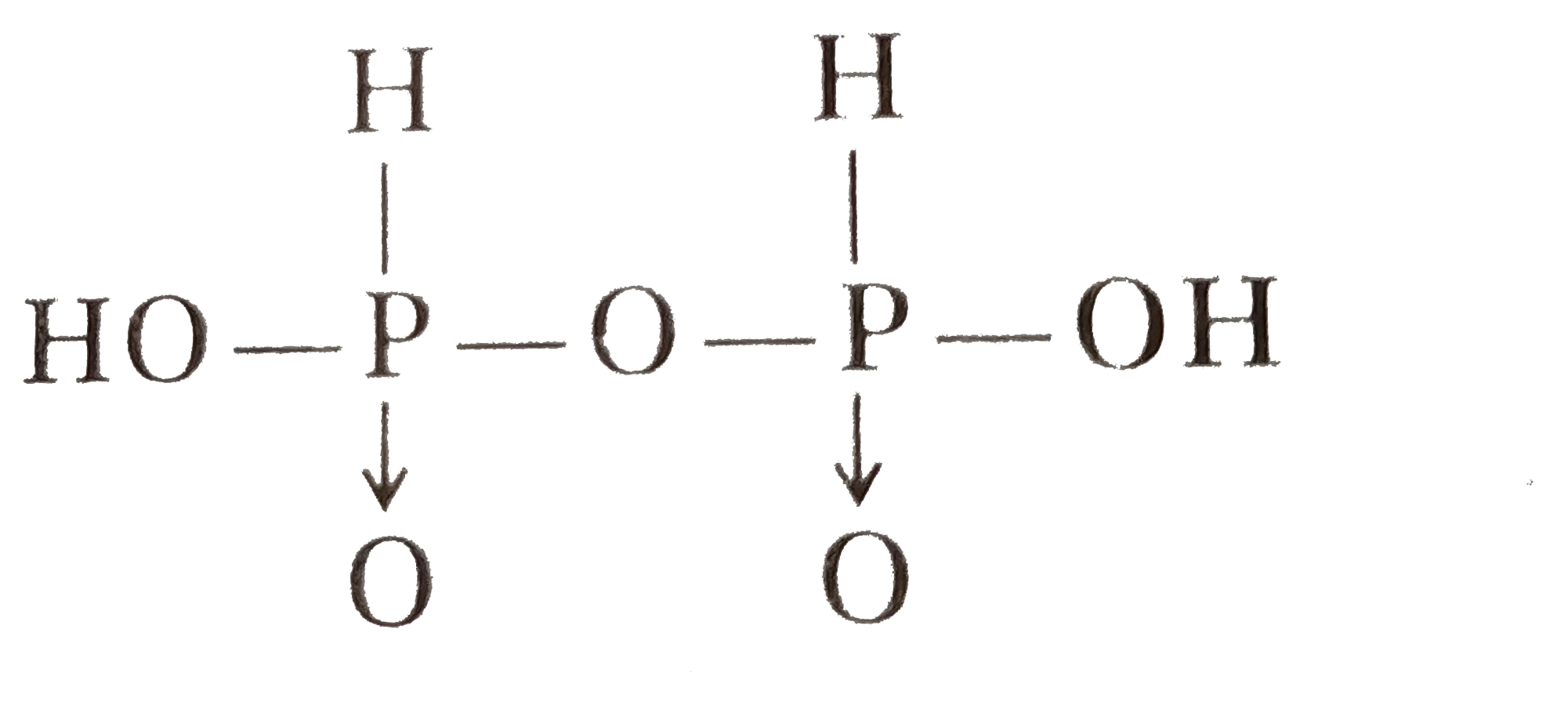
|
|
| 35. |
The pair having similar geometry is |
|
Answer» `BF_(3) , NH_(3)` |
|
| 36. |
The packing fraction of a simple cubic unit cells ________ |
|
Answer» |
|
| 37. |
The packing efficiency of the two dimensional square unit cell show in the adjoining fig ,. Is (##PR_CHE_V01_XII_C01_E20_009_Q01.png" width="80%"> |
|
Answer» 0.3927 Area occupied by circles ` (" within the square")/("Area of the square") ` ` ( 2 xx pir^(2))/a^(2) ` AC = `SQRT(AB^(2) + BC^(2)) = sqrt(a^(2) + a^(2)) = sqr2 a ` but AC = 4 r( r= radius of each sphere) ` sqrt2 a= 4R or a = 4/sqrt2 r = 2 sqrt2r` packing fraction ` ( 2XX pi r^(2))/((2 sqrt2 r)^(2))= pi/4 = ( 3.143)/4 = 0.785i.e, 78.5%` |
|
| 38. |
The packing effciencyof the two dimensional cell square unit as shownis |
|
Answer» 0.3927 Area of unit all=`a^(2)` 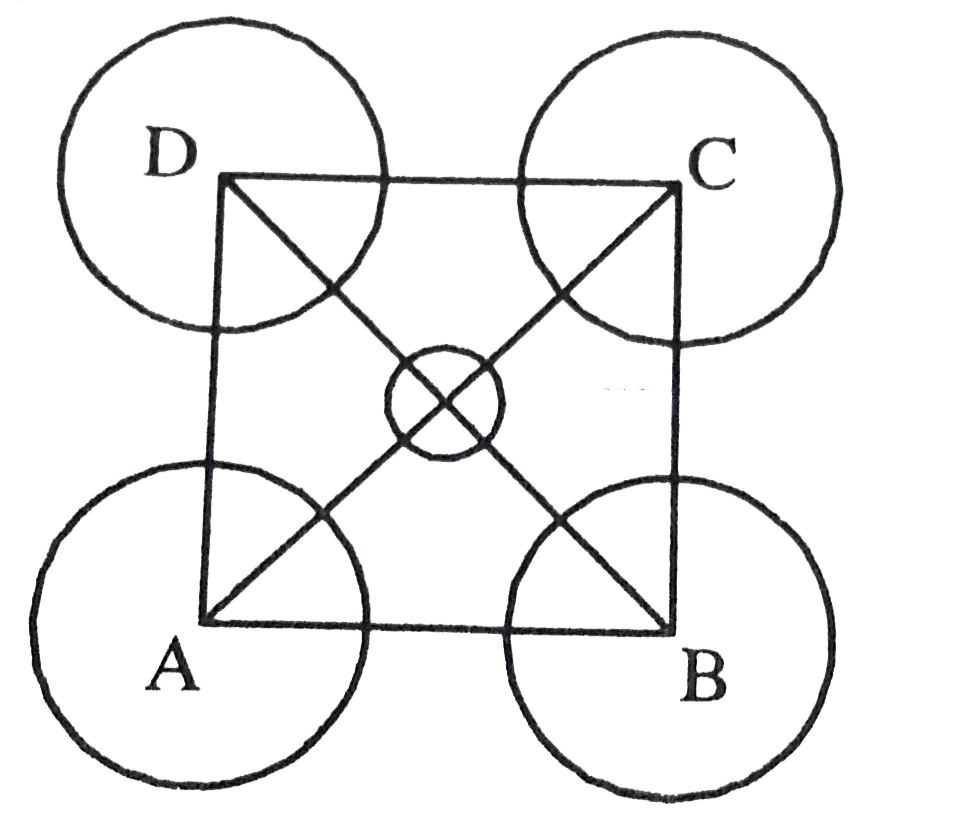 `"Face diagonal " AC=sqrt2a` `"But" "" AC = 4r` `therefore""sqrt2a =4r` `or""a = 2sqrt2r` `"No. of""""PARTICLES" = 4times1/4+1=2` `"Area occupied by particles" = 2timespir^(2)` `"Packing efficiency" = (2timespir^(2))/((2sqrt2r)^(2))=pi/4=0.7857` =`78.57%`. |
|
| 39. |
The p pi- p piback bonding occurs in the halides of boron and not in those of aluminium. Explain. |
|
Answer» <P> Solution :The tendency to show `p pi -p pi` BACK bonding depends upon the size of central ATOM. This tendency decreases as the size of the central atom in a group increases.SINCE aluminium has LARGER size than boron, the back bonding is not possible. |
|
| 40. |
The P-P-P angle in P_(4) molecule is …………..degree while S-S-S angle in S_(8) is ………..degree. |
|
Answer» |
|
| 41. |
the ozonolysis product(s) of the following reaction is(are) CH_(3)CH_(2)-C-=CH underset("(ii) "H_(2)O)overset((i)" "O_(3))toproduct(s) |
|
Answer» `CH_(3)COCH_(3)` |
|
| 42. |
The ozonolysis product of an alkyne is 2-oxopentanal. Then the alkyne is___________ |
|
Answer» Solution :2-oxopentanal is `CH_(3)-CH_(2)-CH_(2)-underset(O)underset(||)C-underset(O)underset(||)CH` HENCE the triple BOND should be present between `C_(1)` and `C_(2)`, so that the alkyne is 1-pentyne. `CH_(3)-CH_(2)-CH_(2)-C-=CH` |
|
| 43. |
The ozone layer protecting the lives is situated in the troposphere. |
| Answer» SOLUTION :FALSE (OZONE layer is SITUATED in the stratosphere.) | |
| 44. |
The ozone layer is depleted by |
|
Answer» NO |
|
| 45. |
The oxygen needed for complete combustion of 4 gm CH_(4) may be obtained from decomposition of : |
|
Answer» `(4)/(3)` moles of `KCIO_(3)` with 25% YIELD `(KCIO_(3) rarr KCI+(3)/(2)O_(2))` |
|
| 46. |
The oxoacid of phosphorus that reduces silver nitrate into metallic silver is |
|
Answer» `H_(3)PO_(2)` |
|
| 47. |
The oxisation of SO_(2) and O_(2) to SO_(3) is an exothermic reaction. The yield of SO_(3) will be maximum if |
|
Answer» Temperature and PRESSURE both are increased |
|
| 48. |
The oxidising agent in the reaction 2MnO_(4)^(-) +16H^(+) +5C_(2)O_(4)^(-2) rarr 2Mn^(+2) +8H_(2)O +10CO_(2) |
|
Answer» `MnO_(4)^(-)` |
|
| 49. |
The oxides which give H_(2)O_(2) on treatment with dilute acid are |
|
Answer» `PbO_2` |
|