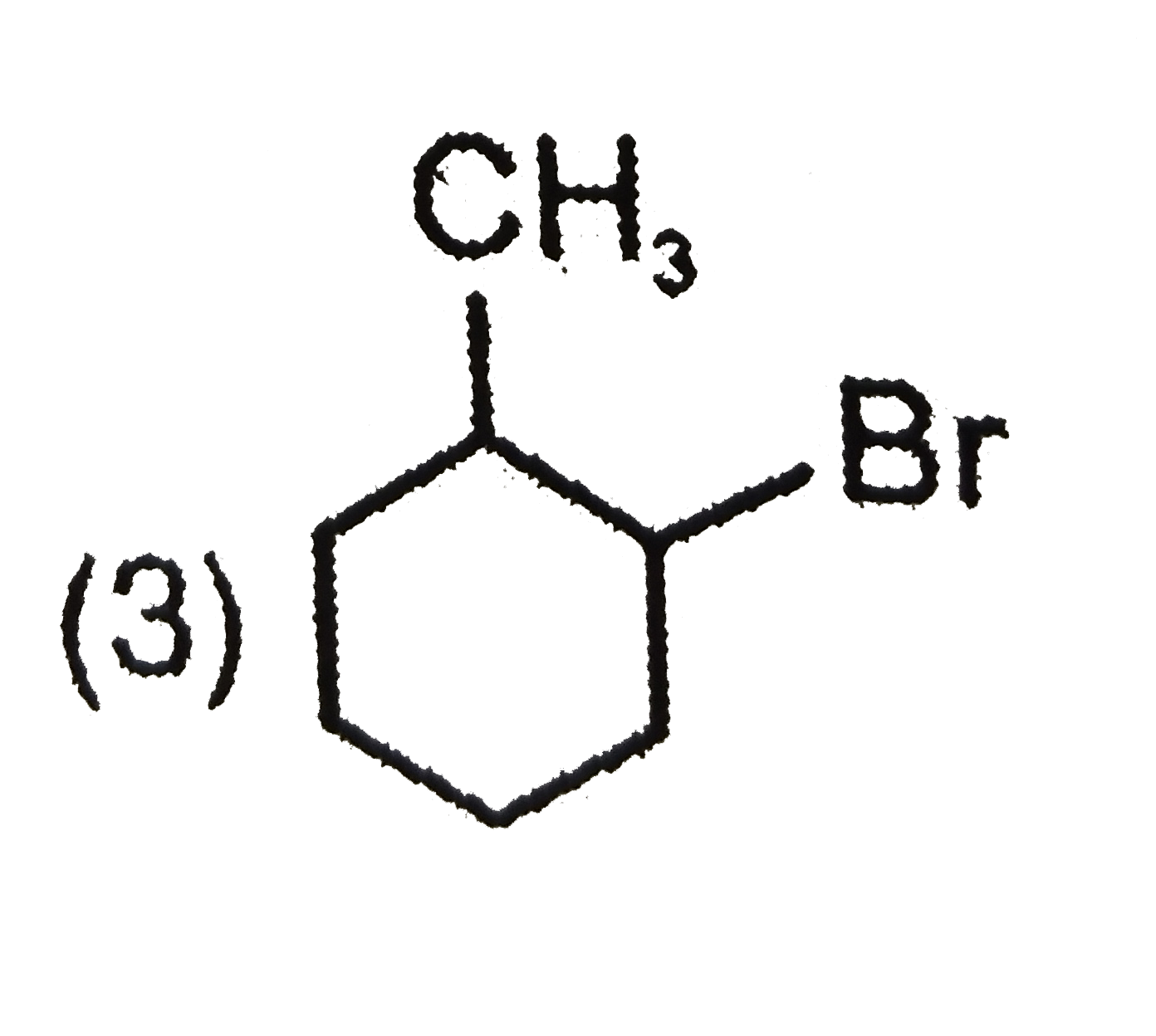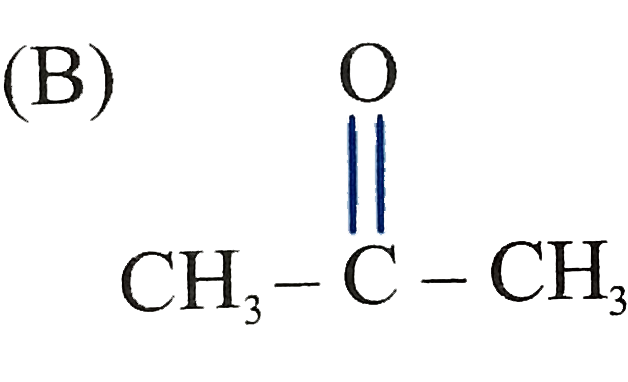Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
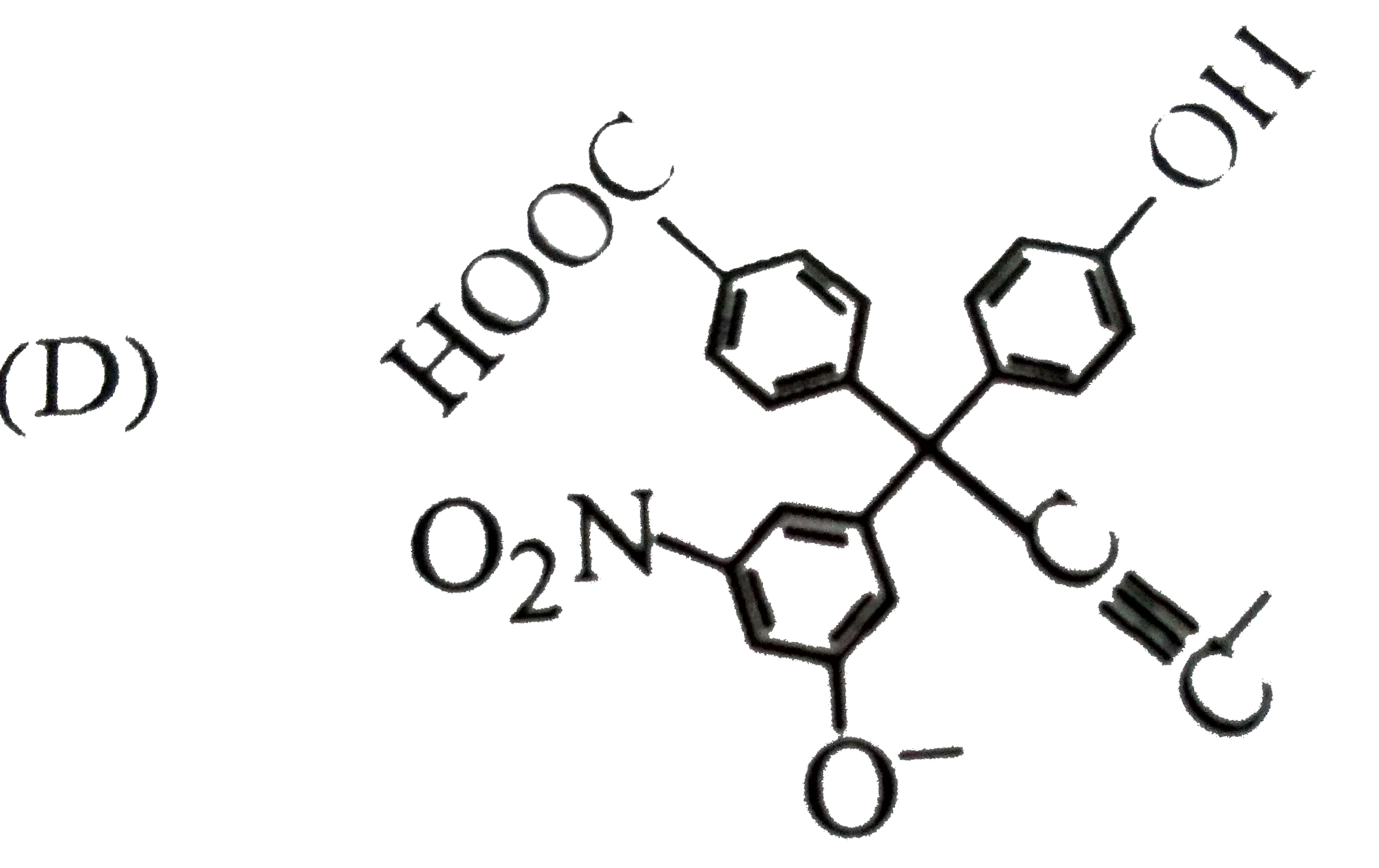
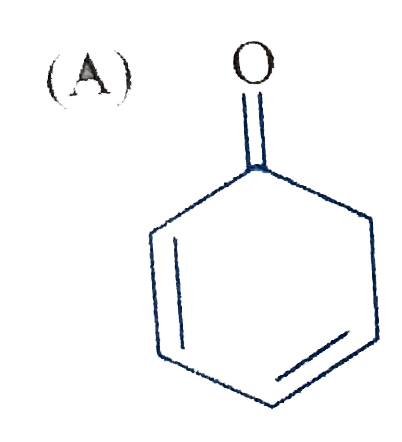
The product 'E' is : |
|
Answer» `CH_3-CH_2-CH=CH_2` |
|
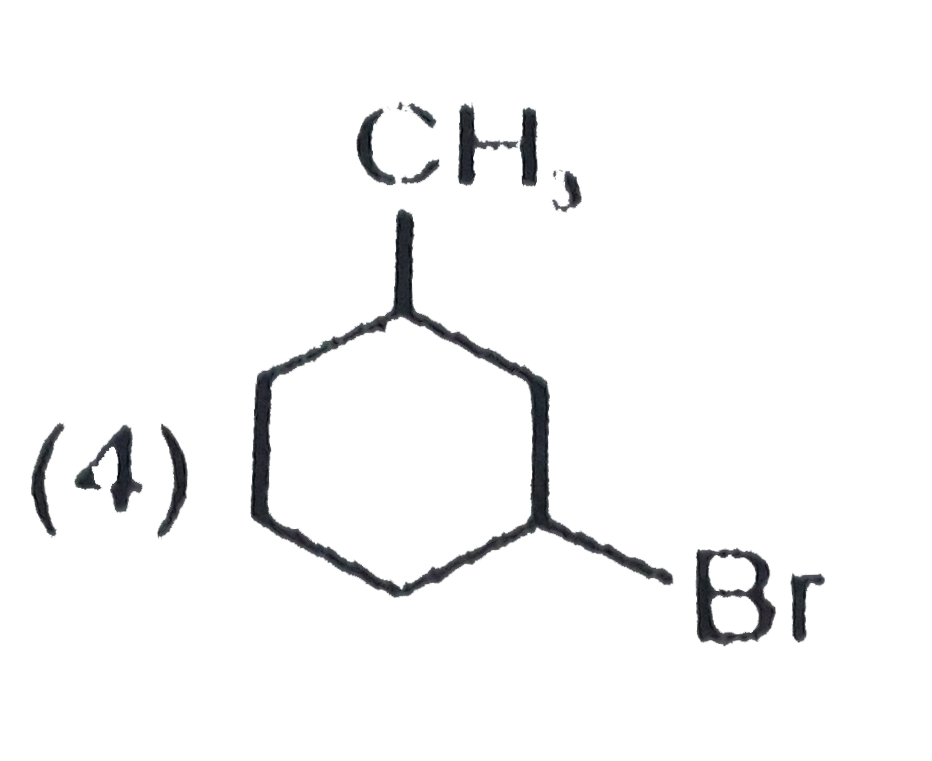
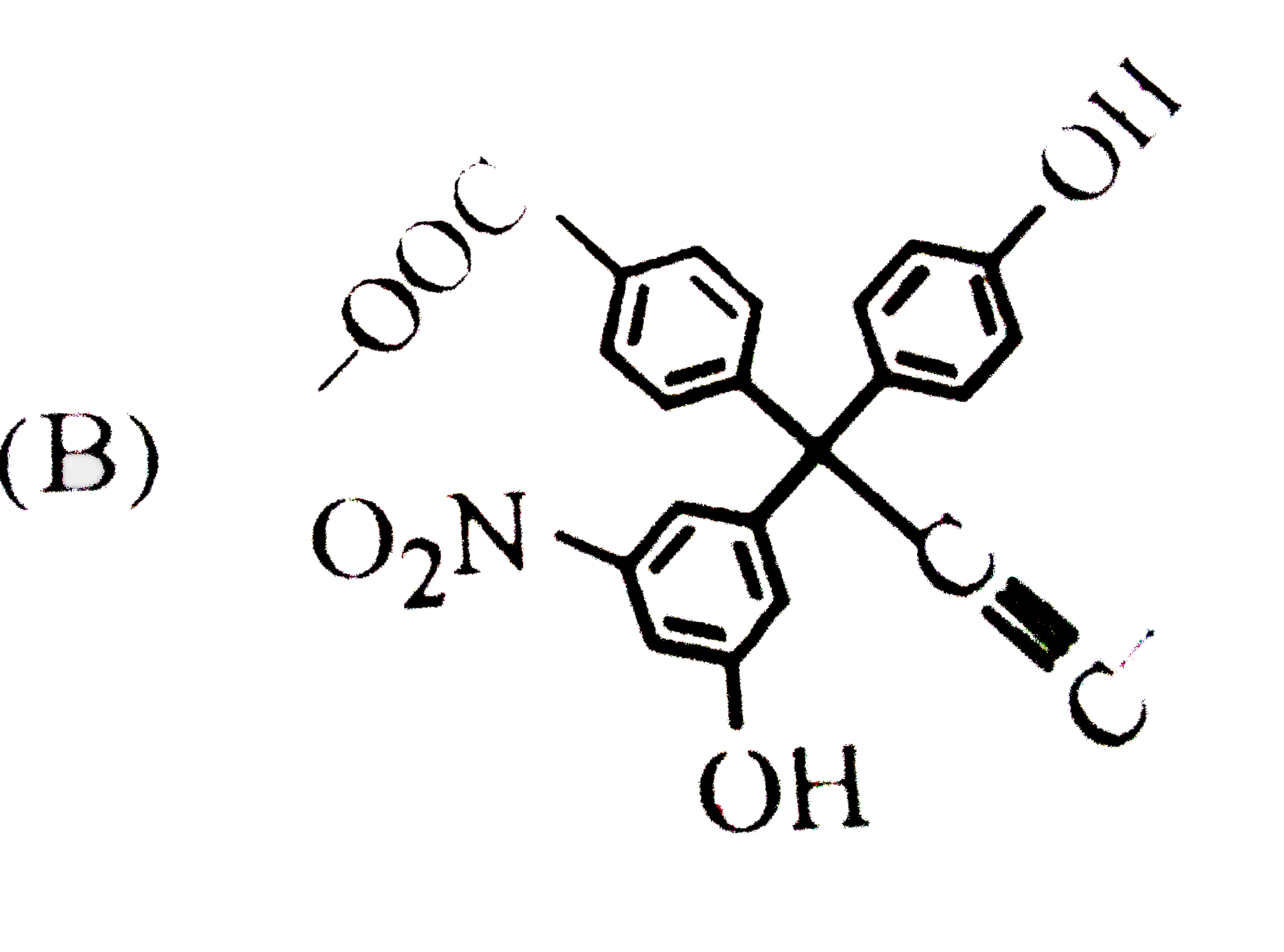
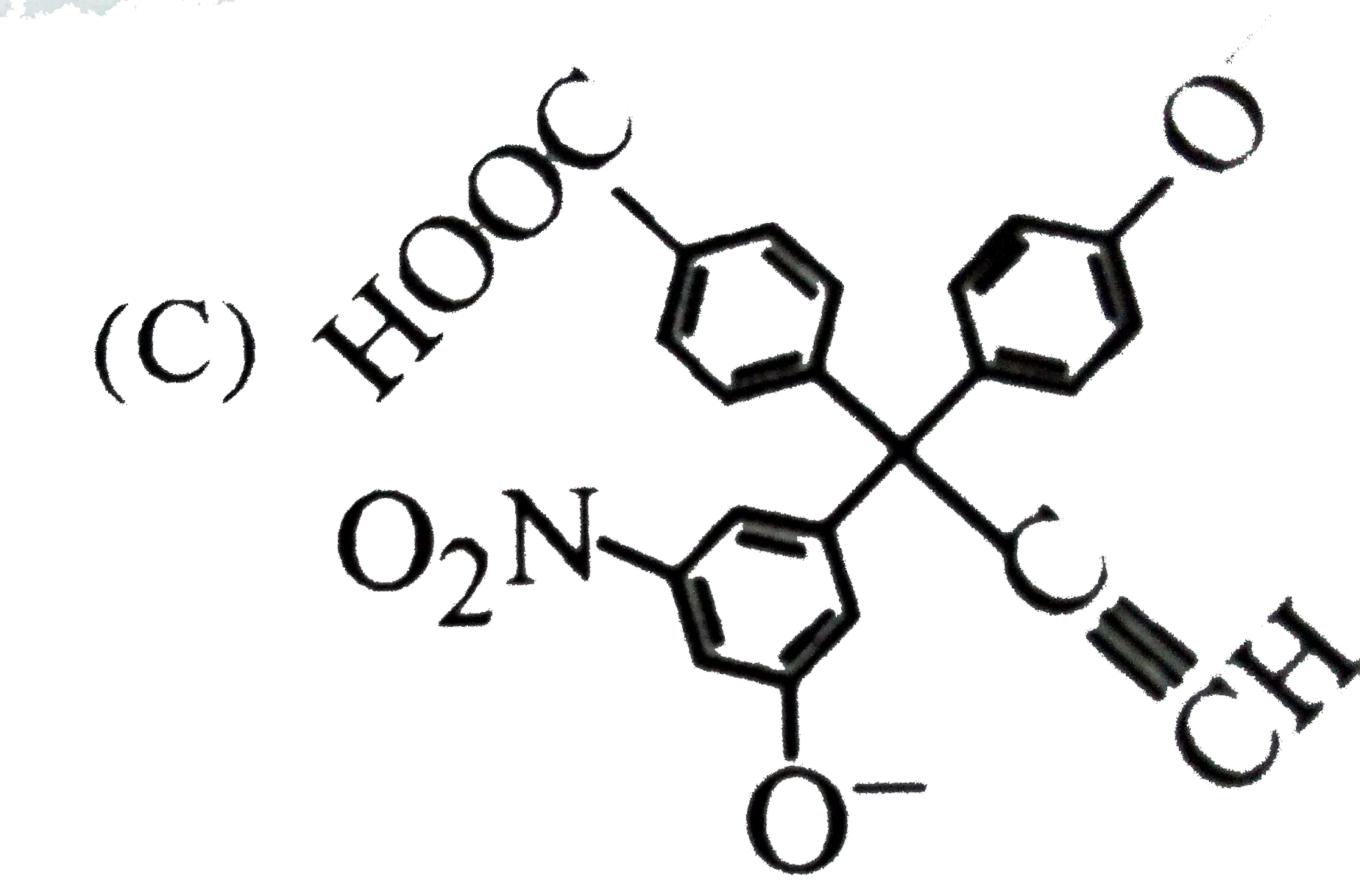
| 3. |
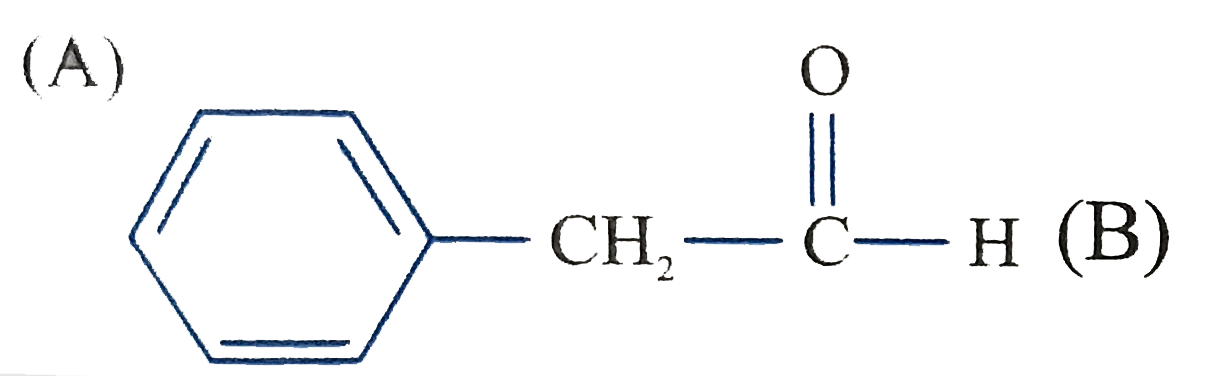
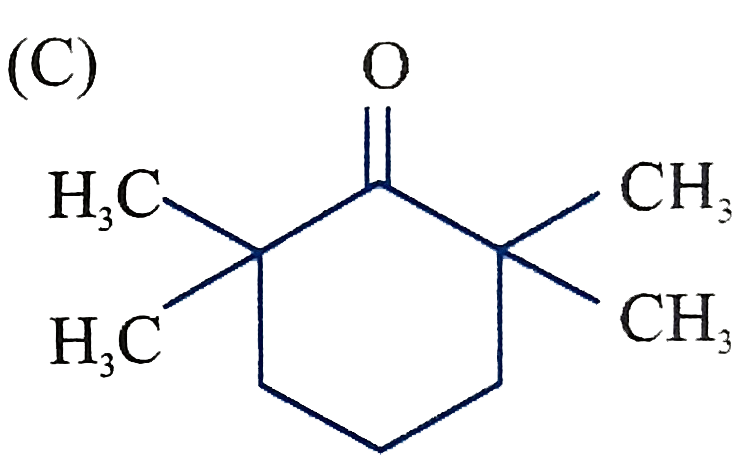
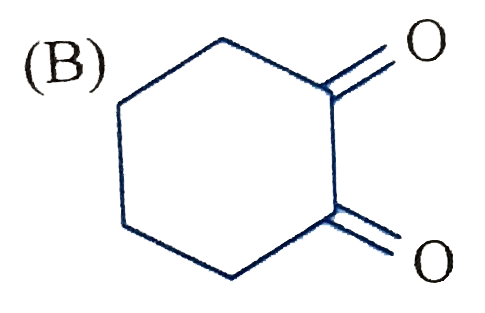
The product C of the following sequence is : |
|
Answer»

|
|
| 4. |
The product {:(CH_(2)OOC CH_(3)),("|"),(CH_(2)OOC CH_(3)):} is obtained by the reaction of |
|
Answer» Acetone and Glycol |
|
| 5. |
The product A will be |
|
Answer»
|
|
| 6. |
The prod electron is transferred towards the attacking reagent it is called ____________ |
|
Answer» `+I ` EFFECT |
|
| 7. |
The processes commonly used for the separation and purification of organic substances are crystallisation, sublimation and |
|
Answer» Stearn DISTILLATION |
|
| 8. |
The process with negative entropy change is |
|
Answer» Dissolution of iodine in water Decrease in number of gaseous molecules. |
|
| 9. |
The process whereby a hydrogen atom attached to the alpha-carbon carbonyl compound moves to the cabonyl oxygen atom is nown as enolization or keto-enol tautomerism. Isomeric carbonyll and enol structures are tautomers. Normally, the carbonyll form is favoured but structural factor market affect K_(T). Which of following compounds will not exhibit enolization?. |
|
Answer»
|
|
| 10. |
The process whereby a hydrogen atom attached to the alpha-carbon carbonyl compound moves to the cabonyl oxygen atom is nown as enolization or keto-enol tautomerism. Isomeric carbonyll and enol structures are tautomers. Normally, the carbonyll form is favoured but structural factor market affect K_(T). Which of the following does not exits in enol forms? |
|
Answer»
|
|
| 11. |
The process whereby a hydrogen atom attached to the alpha-carbon carbonyl compound moves to the cabonyl oxygen atom is nown as enolization or keto-enol tautomerism. Isomeric carbonyll and enol structures are tautomers. Normally, the carbonyll form is favoured but structural factor market affect K_(T). Arrange the following compounds in decreasing order of k_(T) (Tautomeric constant) |
|
Answer» `PgtQgtR GTS` |
|
| 12. |
The process used for the removal of hardness of water is |
|
Answer» Baeyer |
|
| 13. |
The process requiring the absorption of energy is |
|
Answer» `F RARR F^(-)` |
|
| 14. |
The process requiring absorption of energy is (1) F to F^(-) (2) Cl to Cl^(-) O to O^(2-) (4) H to H^(-) |
|
Answer» |
|
| 15. |
The process of zone refining is used in the purification of |
|
Answer» SI |
|
| 16. |
The process of warming of the earth is known as …………… |
|
Answer» GREEN house EFFECT |
|
| 17. |
The process of setting of cement under water is essentially an oxidation process. |
|
Answer» |
|
| 18. |
The process reduction. involves |
|
Answer» addition of `O_2`or REMOVAL of `H_2`to a MOLECULE `""rarr`removal of `e^(-)` `""rarr`removal of Hydrogen `""rarr` Increase in oxidation no . |
|
| 19. |
The process of evaporation of a liquid is accompanised by (A) Increase in enthalpy (B) Increase in entropy ( C) Decrease in Gibbs energy The correct statement(s) is/are |
| Answer» Answer :D | |
| 20. |
The process of eutrophication is due to |
|
Answer» increase in CONCENTRATION of insecticide in water |
|
| 21. |
The process of 'eutrophication' is due to |
|
Answer» Increase in concentration of INSECTICIDE in water. |
|
| 22. |
The process of eutrophication is due ot |
|
Answer» increase in the concentration of INSECTISIDE in WATER |
|
| 23. |
The process of dialysis finds application in the purification of blood by artifical kidney.In this impure blood is introduced in the artificial kidney, apparatus, where the waste material (electrolyte) diffuses through the membrane. The membrane used in the dialyser is different from the membrane used in osmosis. these membrane allow the movement of ions through them. Blood is a negatively charged sol. The haemogolobin particles carry a positive charge. Blood is slightly alkaline (pH 7.36-7.42). Acidic salts like alum and FeCI_3 decrease the pH of the blood and the denaturation of globular proteins present in blood takes place. Due to denaturation, these globular proteins become fibrous which are insoluble and stop bleeding . Blood is lyophobic in nature. which of the following is/are lyophobic colloids? |
| Answer» ANSWER :A,D | |
| 24. |
The process of dialysis finds application in the purification of blood by artifical kidney.In this impure blood is introduced in the artificial kidney, apparatus, where the waste material (electrolyte) diffuses through the membrane. The membrane used in the dialyser is different from the membrane used in osmosis. these membrane allow the movement of ions through them. Blood is a negatively charged sol. The haemogolobin particles carry a positive charge. Blood is slightly alkaline (pH 7.36-7.42). Acidic salts like alum and FeCI_3 decrease the pH of the blood and the denaturation of globular proteins present in blood takes place. Due to denaturation, these globular proteins become fibrous which are insoluble and stop bleeding . Blood is lyophobic in nature. which of the following statements is/are not true ? |
|
Answer» Blood is positively charged SOL |
|
| 25. |
The process of dialysis finds application in the purification of blood by artifical kidney.In this impure blood is introduced in the artificial kidney, apparatus, where the waste material (electrolyte) diffuses through the membrane. The membrane used in the dialyser is different from the membrane used in osmosis. these membrane allow the movement of ions through them. Blood is a negatively charged sol. The haemogolobin particles carry a positive charge. Blood is slightly alkaline (pH 7.36-7.42). Acidic salts like alum and FeCI_3 decrease the pH of the blood and the denaturation of globular proteins present in blood takes place. Due to denaturation, these globular proteins become fibrous which are insoluble and stop bleeding . Blood is lyophobic in nature. The coagulating power of an electrolyte for blood decreases in the order : |
|
Answer» `NA^+`, `AL^(3+)`, `Ba^(2+)` |
|
| 26. |
The process of dialysis finds application in the purification of blood by artifical kidney.In this impure blood is introduced in the artificial kidney, apparatus, where the waste material (electrolyte) diffuses through the membrane. The membrane used in the dialyser is different from the membrane used in osmosis. these membrane allow the movement of ions through them. Blood is a negatively charged sol. The haemogolobin particles carry a positive charge. Blood is slightly alkaline (pH 7.36-7.42). Acidic salts like alum and FeCI_3 decrease the pH of the blood and the denaturation of globular proteins present in blood takes place. Due to denaturation, these globular proteins become fibrous which are insoluble and stop bleeding . Blood is lyophobic in nature. To stop bleeding , FeCI_3 is applied locally because : |
|
Answer» `FeCI_3` seals the blood vessels |
|
| 27. |
The process of dialysis finds application in the purification of blood by artifical kidney.In this impure blood is introduced in the artificial kidney, apparatus, where the waste material (electrolyte) diffuses through the membrane. The membrane used in the dialyser is different from the membrane used in osmosis. these membrane allow the movement of ions through them. Blood is a negatively charged sol. The haemogolobin particles carry a positive charge. Blood is slightly alkaline (pH 7.36-7.42). Acidic salts like alum and FeCI_3 decrease the pH of the blood and the denaturation of globular proteins present in blood takes place. Due to denaturation, these globular proteins become fibrous which are insoluble and stop bleeding . Blood is lyophobic in nature. Which of the following colloidal solutions does not contain negatively charged particles? |
| Answer» Answer :A | |
| 28. |
The process of adding impurities to a crystalline substance so as to change its properties like conductivity etc. is called __________ |
|
Answer» |
|
| 29. |
In which process there is no exchange of heat between the system and surrounding during the process ? |
| Answer» SOLUTION :ADIABATIC PROCESS | |
| 30. |
The process in which the volume of the system remains constant is called _____ |
| Answer» SOLUTION :ISOCHORIC PROCESS | |
| 31. |
The process in which liberation of energy associated in the |
|
Answer» `F to F^-` |
|
| 32. |
The process 1/2 H_(2)(g)+e^(-) to H^(-)(g) is endothermic (DeltaH=+151kJ mol^(-)), still ionic hydrides are known. How do you accound for this: |
| Answer» Solution :In the formation of solid ionic hydrides, energy is released as released as ENTHALPY `Delta_(F)H` from the corresponding ions in the GASEOUS state that are involved in the hydride. This compensates the energy which is needed for the formation of `H^(-)` ions from `H_(2)` GAS | |
| 33. |
The process 1/2 H_(2)(g) + e^(-) to H^(-)(g) is endothermic(DeltaH=+151 KJ "mol"^(-1)) , yet salt like hydrides are known. How do you account for this ? |
| Answer» Solution :It is true that formation of hydride `(H^(-))` ion is an endothermic process, yet ALKALI and alkaline earth metals form salt like hydrides. This is DUE to the REASON that high lattice ENERGY released (energy released during the formation of solid metal hydrides from their correspoinding gaesous IONS, `M^(+) (g) and H^(-)(g))` more than compensates the energy needed for the formation of `H^(-)` ions from `H_(2)` gas. | |
| 34. |
The probability of finding the electron in the orbital is |
|
Answer» zero at the NUCLEUS |
|
| 35. |
The probability of finding electron in XY plane for P_(Z) - orbital is |
|
Answer» 1 |
|
| 36. |
The probability of finding an electron in the P_x orbital is |
|
Answer» ZERO at nucleus |
|
| 37. |
The probability of finding an electron in p_(y) orbital along the x-axis is |
|
Answer» Maximum |
|
| 38. |
The probability density plots of Is and 2s orbitals are given in The density of dots in a region represents the probability density of finding electrons in the region. On the basis of above diagram which of the following statements is incorrect |
|
Answer» Is and 2s orbitals are SPHERICAL in shape. |
|
| 39. |
The principle which gives a system to fill the electrons in increasing order of energy level is |
|
Answer» HUND's RULE |
|
| 40. |
The principle involved in paper chromatography is |
|
Answer» adsorption |
|
| 41. |
The principle involved in paper chromatography is……… |
|
Answer» adsorption |
|
| 43. |
The principle involved in Ammonia-solvay process is |
|
Answer» Low solubility of `Na_(2)CO_(3)` |
|
| 44. |
The principal quantum number represents |
|
Answer» Shape of an orbital |
|
| 45. |
The principal quantum number of an atom is related to the |
|
Answer» SIZE of the ORBITAL |
|
| 46. |
The principal organic product formed in the reaction CH_(2) = CH (CH_(2))_(8) COOH + HBr overset("Peroxide")rarr is |
|
Answer» `CH_(3) CH (Br) (CH_(2))_(8) COOH` |
|
| 47. |
The principal products obtained on heating iodine with cold and concentrated caustic soda solution |
|
Answer» `NaI0_(4) + NAI` |
|
| 48. |
The principal buffer present in human blood is |
|
Answer» `NaH_(2)PO_(4)+Na_(2)HPO_(4)` |
|
| 49. |
The primary salt formed when H_2O_2 reacts with NaOH , is |
|
Answer» `Na_2O_2` |
|
| 50. |
The pressure -volume work for an ideal gas canbe calculated by using the expression w= -int_(V_(l))^(V_(f))p_(ex)dV.The workcan also be calculated from the pV - plotby using the area under the curve within the specified limit. When anideal gas is compressed(a)reversibly or (b) irreversibly from volumeV_(i) to V_(f), choose the correct option. |
|
Answer» <P>W ( reversible)=w ( irreversible) |
|