Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The pressure-volume work for an ideal gas canbe calculated by using the expression W=-int_(V_(i))^(V_(f))P_(ex)dV. The work can also be calculated from the pV - plot by using the area under the curve within the specified limits. When an ideal gas is compressed (i) reversibly or (ii)irreversibly from volume V_(i) to V_(f). Choose the correct option. |
|
Answer» `W_("reversible")=W_("IRREVERSIBLE")` 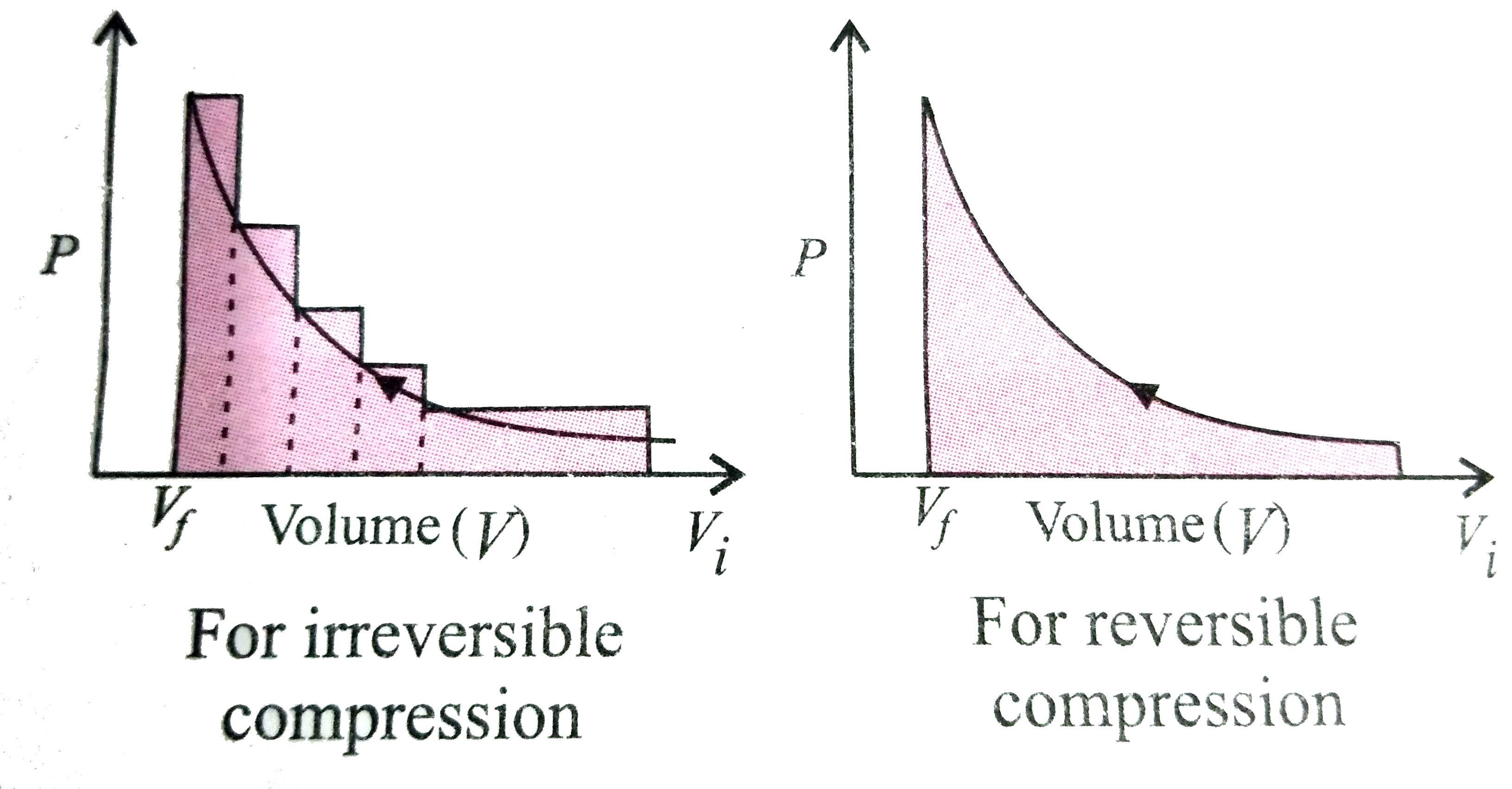 In both the curves, work DONE on the gas is REPRESENTED by the shaded area and the area under the curve is alwaysmore in irreversible compression. Hence, `W_("irreversible") gt W_("reversible")` |
|
| 2. |
The price incurred in producing one kg of magnesium is Rs. 25. What is the price of one mole of metal? |
|
Answer» |
|
| 3. |
The pressure-volume work for an ideal gas can be calculated by using the expression w = -int_(V_(i))^(V_(f)) p_(exdV).The work can be calculated form the pV- plot by using the area under the curve within the specified limits. When an ideal gas is compressed (a) reversibly or (b) irreversibly from volume V_(i) to V_(f), choose the correct option. |
|
Answer» <P>w (REVERSIBLE) = w (IRREVERSIBLE) |
|
| 4. |
The pressure-volume work for an ideal gas can be calculated by using the expression W= - int_(V_i)^( V_f) P_(ex) dV. The work can also be calculated from the pV- plot by using the area under the curve within the specifiedlimits. When an ideal gas is compressed (A) reversibly or (B) irreversibly from volume V_i to V_f. choose the correct option. |
|
Answer» <P>W (reversible) = W (irreversible)  pV-plot when pressure is not constant and CHANGES in finite steps during compression from INITIAL volume, `V_i` to find volume, `V_f`. WORK done on the gas is represented by the shaded area.  pV-plot when pressure is not constant and changes in infinite steps (reversible conditions) during compression from initial volume, `V_i` to final volume, `V_f`. Work done on the gas is represented by the shaded area. |
|
| 5. |
The pressure of the atmosphere is 2xx 10^(-6) mm at about 100 mile from the earth and temperature is -180^(@)C. How many moles are three in 1 mL gas at this attitude? |
| Answer» SOLUTION :`3.45 XX 10^(-13) MOL` | |
| 6. |
The pressure of an equilibrium mixture of the three gases NO, Cl_(2) and "NOCl" 2NO(g) + Cl_(2) (g ) hArr 2NOCl(g)is suddely decreased by doubling by doubling the volume of the container at constant temperature. When the systemreturns to equilibrium |
|
Answer» the concentration of `NOCI` will be increased |
|
| 7. |
The pressure of a gas is due to ........... exerted by its molecules per ........... of the walls of the container. |
| Answer» SOLUTION :FORCE, UNIT AREA | |
| 8. |
The pressure of a 1 : 4 mixture of dihydrogen and dioxygen enclosed in a vessel is one atmoshpere. What would be the partial pressure of dioxygen ? |
|
Answer» `0.8xx10^(5)` ATM `THEREFORE` Partial pressure of `O_(2)=` Mole fraction of `O_(2)xx` total pressure of mixture `= (4)/(1+4)xx1=(4)/(5)xx1` atm `= 0.8 atm = 0.8xx105 Nm^(-2)=8xx10^(4)Nm^(-2)` |
|
| 9. |
The pressure of a 1:4 mixtureof dihydrogen and dioxygen enclosed in a vessel is one atmosphere. What would be the partial pressure of dioxygen ? |
|
Answer» `0.8xx10^(5)` atm `=(4)/(5)xx1 "atm"=0.8xx10^(5)" N "m^(-2)=8xx10^(4)" N "m^(-2)`. |
|
| 10. |
The pressure of 1 atmosphere is equal to |
|
Answer» 760 cm |
|
| 11. |
The pressure exerted by the gas is directly proportional to |
|
Answer» ROOT mean square SPEED |
|
| 12. |
The pressure exerted by a mass of 'x' mg resting on the area of 1.00 cm^2 is 1.00 Pa, then 'x' is |
|
Answer» 10.300 |
|
| 13. |
The pressure exerted by a mass of 'x' mg resting on the area of 1.00 cm^2is 1.00 Pa, then 'x' is |
|
Answer» 10.3 |
|
| 14. |
The pressure exerted by 6.0 g of methane gas in a 0.03 m^(3) vessel at 129^(@)C is (Atomic masses : C=12.01, H=1.01 and R=8.314 JK^(-1)mol^(-1)) |
|
Answer» 215216 Pa `=16.05" G "mol^(-1)` `:. 6" g of "CH_(4)=(6)/(16.05)"mole"`, `T= 129^(@)C=129+273" K"=402" K"` PV=nRT or `P=(nRT)/(V)=(6)/(16.05)xx(8.314xx402)/(0.03)=41648" Pa"` |
|
| 15. |
The pressure exerted by 6.0 g of methane gas in a 0.03 m^(3) vessel at 129^(@)C is (Atomic masses : C = 12.01, H = 1.01 and R = 8.314 JK^(-1)mol^(-1)) |
|
Answer» 31684 PA `= (6)/(16.02)=(8.314xx402)/(0.03)=41648` Pa |
|
| 16. |
The pressure exerted by 6.0 g fo methane gas in 0.03m^3 vessel at 129^@ C is (Atomic masses : C = 12.01, H = 1.01 and R = 8.314JK^(-1) mol^(-1)) |
|
Answer» 215216 Pa |
|
| 17. |
The pressure exerted by 12 g of an ideal gas at temperature t^(@)C in a vessel of volume V litre is one atm. When the temperature is increased by 10 degree at the same voume, the pressure increases by 10%. Calculate the temperature t and volume V (Molecular weight of the gas=-120). |
|
Answer» Solution :Applying PV=nRT In the 1 st CASE,`1xxV=(12)/(120)xxRxx(273+t)""`……….(i) In the 2nd case,`1.1xxV=(12)/(120)xxRxx(283+t)""`………..(ii) DIVIDING (i) by (ii), `(1)/(1.1)=(273+t)/(283+t)` On SOLVING it gives `t=-173^(@)C`. Putting in (i), `"" V=0.1xx0.082xx100=0.82" litre"` |
|
| 18. |
The presence of which one of the following groups on benzene nucleus activates it towardselectrophilic substitution? |
| Answer» SOLUTION :`-O-COR` group on benzene NUCLEUS activates it towards electrophilic substitution | |
| 19. |
The presence of -OH groups on adjacent carbon atoms can be detected by the reaction of the compound. |
|
Answer» CONC. `H_(2)SO_(4)` |
|
| 20. |
The presence ofNH_(4)Cl " in " NH_(4)OH solution will ............. the degree of dissociation of NH_(4)OH. |
|
Answer» |
|
| 21. |
The presence of ionic salts in a liquid |
|
Answer» Decreases the viscosity of the liquid |
|
| 22. |
The presence of Hg^(2+) ions is detected as its sulphide in group IIA of qualitative analysis. The black HgS is one of the least soluble precipitate known (K_(sp) = 4 xx 10^(-54)). Aqua regia and sodium sulphide (2M) dissolve the black precipitate. potassium iodide when added slowly to the solution contaning Hg^(2+) ions, a red precipitate is formed which dissolves in excess KI to form a colourless soluble complex. when NH_(4)CI solution is added to the above colourless soluble complex containing some NaOH, a brown precipitate is formed. when to a warm alkaline aqueous solution of Hg(NO_(3))_(2) a solution a ammonium chloride is added, a white precipitate is obtained. Identify the incorrect statement with respect to Hg(II) salts. |
|
Answer» `Hg^(2+)` ions in the presence of DILUTE (M) hydrochloric acid gives initially a white PRECIPITATE of `Hg(II)` chloro sulphide with `H_(2)S` which then reacts with further quantity of `H_(2)S` to form black precipitate of `HgS`. `K_(2)HgI_(4)+NH_(4)CI +KOH rarr HgO.Hg(NH_(2))I` `Hg^(2+) +CO^(2+)+4SCN^(-) rarr underset(("Deep blue crystalline ppt"))(Co[Hg(SCN)_(4)]darr)` `2HG^(2+) +NO_(3)^(-) +4NH_(3)+H_(2)O rarr HgO.underset(("White ppt"))(Hg(NH_(2))NO_(3)darr +3NH_(4)^(+)` |
|
| 23. |
The presence of Hg^(2+) ions is detected as its sulphide in group IIA of qualitative analysis. The black HgS is one of the least soluble precipitate known (K_(sp) = 4 xx 10^(-54)). Aqua regia and sodium sulphide (2M) dissolve the black precipitate. potassium iodide when added slowly to the solution contaning Hg^(2+) ions, a red precipitate is formed which dissolves in excess KI to form a colourless soluble complex. when NH_(4)CI solution is added to the above colourless soluble complex containing some NaOH, a brown precipitate is formed. when to a warm alkaline aqueous solution of Hg(NO_(3))_(2) a solution a ammonium chloride is added, a white precipitate is obtained. The white precipitate formed is due to the formation of: |
|
Answer» mercury(II) oxide `K_(2)HgI_(4)+NH_(4)CI +KOH rarr HgO.Hg(NH_(2))I` `Hg^(2+) +Co^(2+)+4SCN^(-) rarr underset(("Deep blue crystalline ppt"))(Co[Hg(SCN)_(4)]DARR)` `2HG^(2+) +NO_(3)^(-) +4NH_(3)+H_(2)O rarr HgO.underset(("White ppt"))(Hg(NH_(2))NO_(3)darr +3NH_(4)^(+)` |
|
| 24. |
The presence of Hg^(2+) ions is detected as its sulphide in group IIA of qualitative analysis. The black HgS is one of the least soluble precipitate known (K_(sp) = 4 xx 10^(-54)). Aqua regia and sodium sulphide (2M) dissolve the black precipitate. potassium iodide when added slowly to the solution contaning Hg^(2+) ions, a red precipitate is formed which dissolves in excess KI to form a colourless soluble complex. when NH_(4)CI solution is added to the above colourless soluble complex containing some NaOH, a brown precipitate is formed. when to a warm alkaline aqueous solution of Hg(NO_(3))_(2) a solution a ammonium chloride is added, a white precipitate is obtained. What product is formed when black precipitate dissolves in sodium sulphide (2M) solution? |
|
Answer» `NA[HgS_(2)]` `K_(2)HgI_(4)+NH_(4)CI +KOH rarr HgO.Hg(NH_(2))I` `Hg^(2+) +Co^(2+)+4SCN^(-) rarr underset(("Deep blue crystalline ppt"))(Co[Hg(SCN)_(4)]darr)` `2HG^(2+) +NO_(3)^(-) +4NH_(3)+H_(2)O rarr HgO.underset(("White ppt"))(Hg(NH_(2))NO_(3)darr +3NH_(4)^(+)` |
|
| 25. |
The presence of Hg^(2+) ions is detected as its sulphide in group IIA of qualitative analysis. The black HgS is one of the least soluble precipitate known (K_(sp) = 4 xx 10^(-54)). Aqua regia and sodium sulphide (2M) dissolve the black precipitate. potassium iodide when added slowly to the solution contaning Hg^(2+) ions, a red precipitate is formed which dissolves in excess KI to form a colourless soluble complex. when NH_(4)CI solution is added to the above colourless soluble complex containing some NaOH, a brown precipitate is formed. when to a warm alkaline aqueous solution of Hg(NO_(3))_(2) a solution a ammonium chloride is added, a white precipitate is obtained. The brown precipitate formed is due to the formation of: |
|
Answer» oxy-dimercuric ammonium iodide `K_(2)HgI_(4)+NH_(4)CI +KOH rarr HgO.HG(NH_(2))I` `Hg^(2+) +Co^(2+)+4SCN^(-) rarr UNDERSET(("Deep BLUE crystalline ppt"))(Co[Hg(SCN)_(4)]darr)` `2HG^(2+) +NO_(3)^(-) +4NH_(3)+H_(2)O rarr HgO.underset(("White ppt"))(Hg(NH_(2))NO_(3)darr +3NH_(4)^(+)` |
|
| 26. |
The presence of Hg^(2+) ions is detected as its sulphide in group IIA of qualitative analysis. The black HgS is one of the least soluble precipitate known (K_(sp) = 4 xx 10^(-54)). Aqua regia and sodium sulphide (2M) dissolve the black precipitate. potassium iodide when added slowly to the solution contaning Hg^(2+) ions, a red precipitate is formed which dissolves in excess KI to form a colourless soluble complex. when NH_(4)CI solution is added to the above colourless soluble complex containing some NaOH, a brown precipitate is formed. when to a warm alkaline aqueous solution of Hg(NO_(3))_(2) a solution a ammonium chloride is added, a white precipitate is obtained. Hg^(2+) ions give deep blue crystalline precipitate with: |
|
Answer» `KI` `K_(2)HgI_(4)+NH_(4)CI +KOH rarr HgO.Hg(NH_(2))I` `Hg^(2+) +Co^(2+)+4SCN^(-) rarr underset(("Deep blue crystalline ppt"))(Co[Hg(SCN)_(4)]darr)` `2Hg^(2+) +NO_(3)^(-) +4NH_(3)+H_(2)O rarr HgO.underset(("White ppt"))(Hg(NH_(2))NO_(3)darr +3NH_(4)^(+)` |
|
| 27. |
The presence of colloidal particles of dust in air imparts blue colour to the sky. This is due to |
|
Answer» ABSORPTION of light |
|
| 28. |
The presence of delocalized pi - electrons in benzene indicates that it is |
|
Answer» less stable than cyclohexatriene |
|
| 29. |
The presence of carbon in an organic compound can be shown by |
|
Answer» heating the compound wih sodium `C+2CuO to 2Cu+CO_(2)` `CO_(2)+CA(OH)_(2)to underset(("Milky"))(CaCO_(3))+H_(2)O` |
|
| 30. |
The prepare a pure sample of normal hexane using sodium metal as one reactant, the other reactant or reactants will be |
|
Answer» Isobutyl CHLORIDE and ethyl chloride |
|
| 32. |
The prefix and the suffix for CHO group are..............and.............respectively when the carbon atom of the CHO group is not included in the parent chain. |
|
Answer» |
|
| 33. |
The preferred sites of protonation in the following compounds are (I) Ph-CH_(2)-overset(1)overset(NH)overset(||)C-overset(2)overset(NH_(2))""(II)Ph-CH_(2)-overset(3)overset(O)overset(||)C-overset(4)(N)H_(2) |
|
Answer» 1 and 3 |
|
| 34. |
The preferred sites of protonation in the following compounds are |
|
Answer» 1 and 3 |
|
| 35. |
The predominant yellow line in the spectrum of a sodium vapour lamp has a wavelength of 590 nm. What minimum accelerating potential is needed to excite this line in an electron tube having sodium vapours? |
|
Answer» |
|
| 36. |
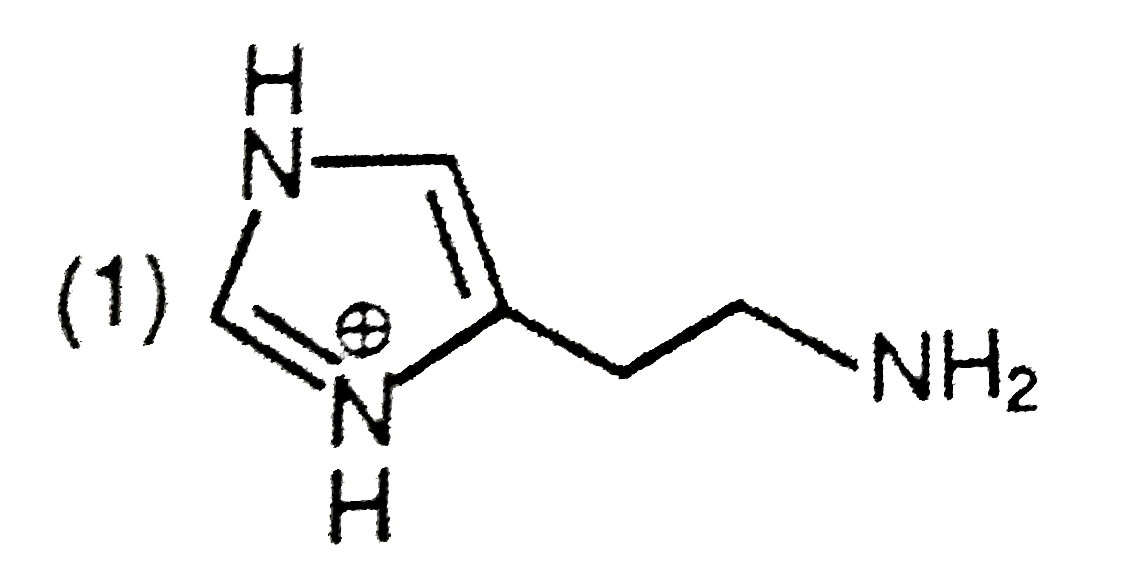
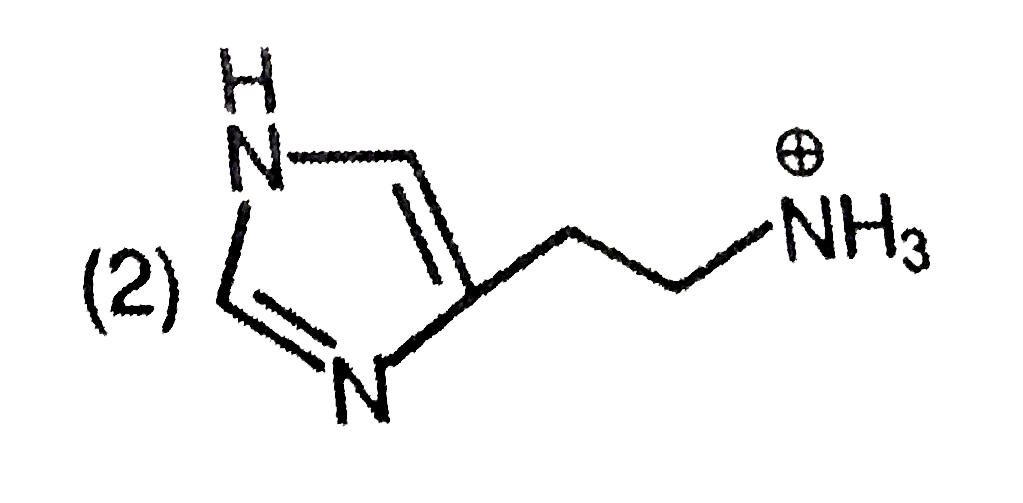
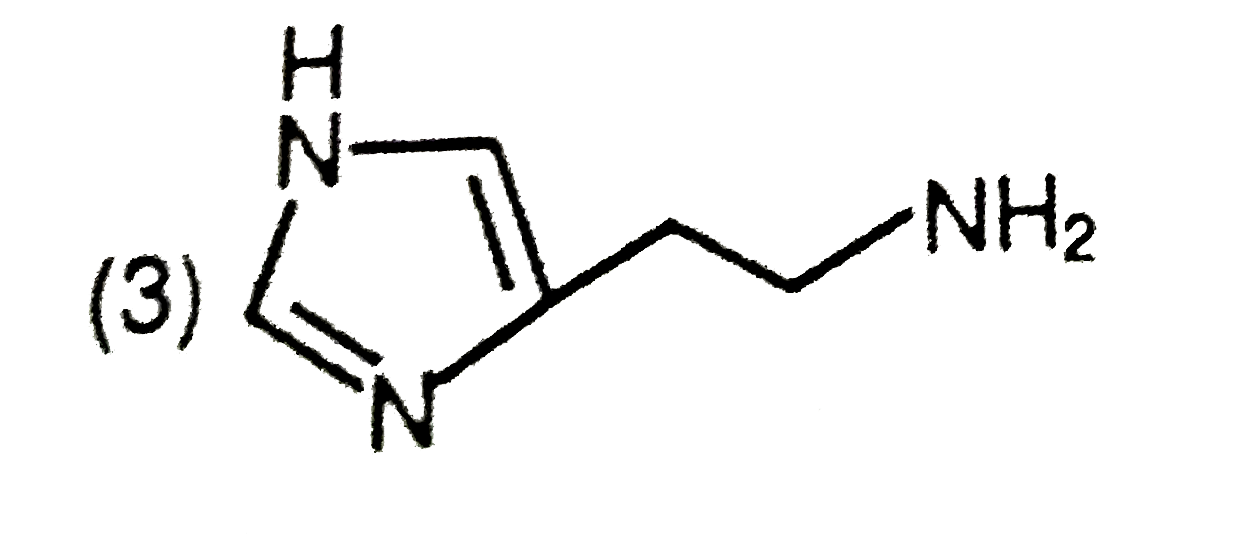
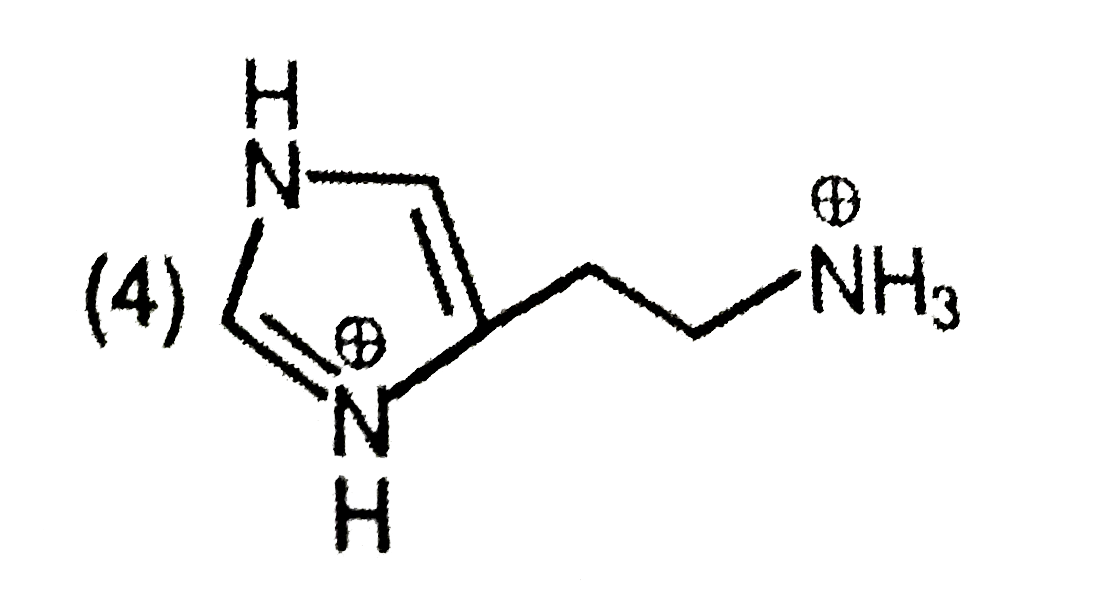
The predominant form of histamine present in human blood is (pK_(a) , Histidine=6.0) |
|
Answer»
|
|
| 37. |
The precipitate of CaF_(2) (K_(sp) = 1.7 xx 10^(-10)) is obtained when equal volumes of the following are mixed ? |
|
Answer» `10^(-4) M Ca^(2+) + 10^(-4) M F^(-)` |
|
| 38. |
The precent composition of an organic compound A is carbon 85.71% and hydrogen 14.29. its vapour density is 14. Consider the following reactions sequence A overset(Cl_(2) //H_(2)O)rarr B underset((2) H_(3)O^(+))overset((1) KCN, EtOH)rarr C Identify C |
|
Answer» `CH_(3)- underset(OH)underset(|)(CH) - CO_(2)H` |
|
| 39. |
The precentage of empty space in a body centred cubic arragngement is ……. |
|
Answer» 74 |
|
| 40. |
The potential of an electrode is represented as Pt. (0.25 atm)//H^(+) [0.5] |
|
Answer» 0 `=0+(0.059)/(2)"log"((0.5)^(2))/((0.25)^(2))=`Zero |
|
| 41. |
The potential energy of an electron in the hydrogen atom is -6.8 eV. Indicate in which excited state, the electron is present ? |
|
Answer» first |
|
| 42. |
The potentialenergy difference between the staggered and eclipsed conformation of ethaneis ……………… |
| Answer» SOLUTION :12.5 KJ`//`MOL | |
| 43. |
The postulate of Bohr theory that electrons jump from one orbit to the other, rather than flow is according to |
|
Answer» The QUANTISATION CONCEPT |
|
| 44. |
The possition number of 'Cl' in the major product of the reactio (locant) |
|
Answer» |
|
| 45. |
The positive value of DeltaS indicates that |
|
Answer» the SYSTEM becomes LESS disordered |
|
| 46. |
The position of the element with electronicconfiguration1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) 3d^(3) 4s^(2) is placed in the group ….. . |
|
Answer» 3 |
|
| 47. |
The position of both, an electron and a helium atom, is known within 1.0 nm. Further, the momentum of the electron is known within 5.0 xx 10^(-26) kg ms^(-1). The minimum uncertainty in the measurement of momentum of helium atom is |
|
Answer» `50 KG ms^(-1)` |
|
| 48. |
The polymerisation of ethylene to linear polyethylene is represented by the reaction, nCH_(2)= CH_(2) rarr (- CH_(2) - CH_(2) -) where n has a largeintegral value. Given that the average enthalpies of bond dissociation forC=C andC-C at298 K are+590 and+331kJ mol^(-1) respectively, calculate the enthalpy of polymerisation per mole of ethyleneat298 K. |
|
Answer» Solution :In the given polymerisation reaction,ONE molecule of ETHYLENE involves breaking of C`=` C double bond and formation of threeC-C single bonds.However,in the complete polymer chain,the NUMBER of single C-C bondsformedin two per C=C double bond broken Energy required in breaking of one moleofC=C doublebonds `= 590 kJ` Energy released in the formation of two moles of`C-C` single bonds `= 2 xx 331 = 662kJ` `:.`Net energy released per moleof ethylene`= 662 - 590 = 72 kJ , i.e.,DELTAH =- 72kJmol^(-10` |
|
| 49. |
The pollutants which come directly in the air from sources are called primary pollutants Primary pollutants are sometimes converted into secondary pollutants. Which of the following belongs to secondary air pollutants? |
|
Answer» CO |
|
| 50. |
The pollutants which come directly in the air from sources are called primary pollutants. Primary pollutants are sometimes converted into secondary pollutants. Which of the following belongs to secondary air pollutants ? |
|
Answer» CO Hydrocarbon + O `to RCO^@` (Free radicals) `RCO^* + O_2 to RCO_3^*` `RCO_3^* + NO_2 to RCO_3 NO_2` |
|