Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
There are certain substances which behave as normal, strong electrolytes at low concentration but at higher concentration they behave as colloidal solutions due to the formation of aggregated particles. Such colloids are called associated colloids and the aggregated particles are called micelles. Soaps and detergents are the example of associated colloids. the formation of micelles takes place above certain concentration called critical micellization concentration (CMC) and a characteristic temperature. Micelles are : |
|
Answer» EMULSIONS CUM gels |
|
| 2. |
What is atomic radius ? |
| Answer» Solution :DISTANCE between ELECTRON and NUCLEUS is known as ATOMIC RADIUS. | |
| 3. |
Which of the following reaction has K_p=K_c ? |
|
Answer» `2HI_((g)) hArr H_(2(g)) + I_(2(g))` |
|
| 4. |
Whichof thefollowingstatementisINCORRECT? |
|
Answer» When pointdetermination , mechanicalstrringof paraffinoilis required. |
|
| 5. |
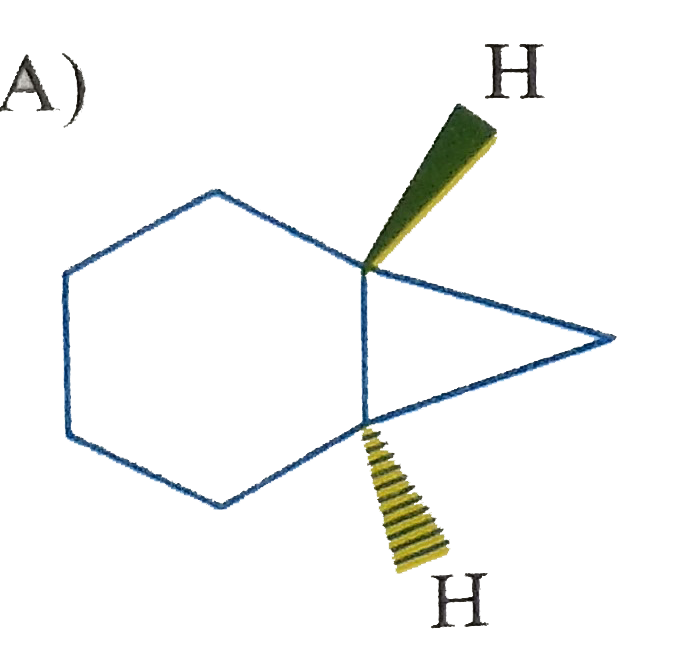
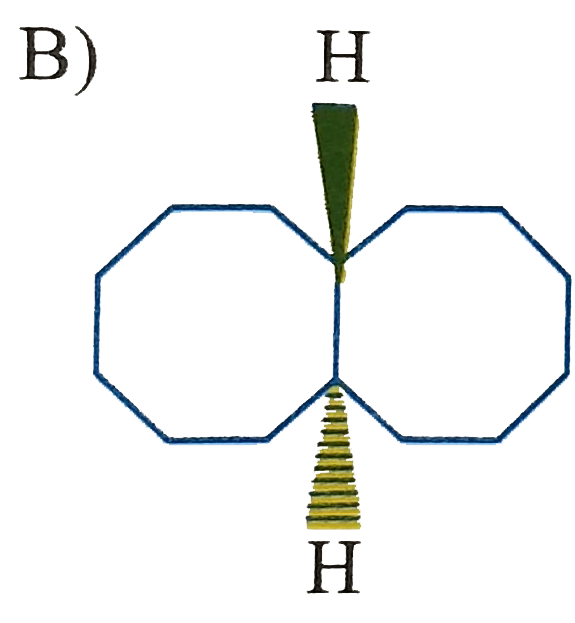
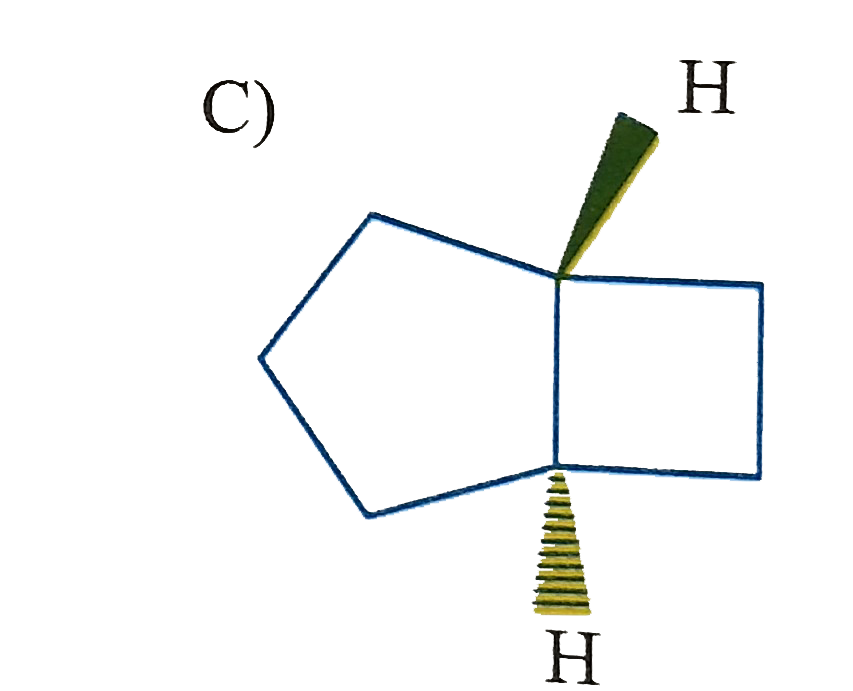
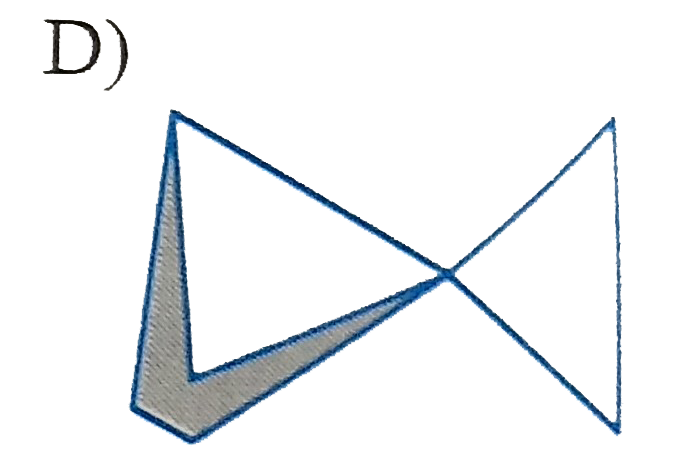
Which of the following is (are) isolable? |
|
Answer»
B) yes, large can be fused trans C)No. cannot be fused trans D) yes. Sprinanes are kinown for any size ring if they are ABLE to be at right angle. |
|
| 6. |
The wave number of a particular spectral line in the atomic spectrum of a hydrogen like species increases 9/4 times when deuterium neuleus is introduced into its nucleus, then which of the following willbe the initial hydrogen like species? |
|
Answer» `LI^(2+)` |
|
| 7. |
When an electron is transisted from 2E to E energy level, the wavelength of resultant photon produce is lambda. If electronic transition involves (4)/(3) E to e level, the wavelength of resultant photon is lambda_(2). The correct relation between lambda_(1) & lambda_(2) is lambda_(2) = x lambda_(1). Then find the value of x ? |
|
Answer» |
|
| 8. |
The solubility product of MgF_(2) is 7.4xx10^(-11). Calculate the solubility of MgF_(2) in 0.1M NaF solution |
|
Answer» `7.4xx10^(-9)` |
|
| 9. |
Which of the following compounds can be used as anifreeze in automobile radiators? |
|
Answer» METHYL alcohol |
|
| 10. |
Which of the following features are not shown by quartz glass ? |
|
Answer» This is a CRYSTALLINE solid |
|
| 11. |
When organic substances undergo anaerobic degradation the product formed mainly is |
|
Answer» `CO_2` |
|
| 12. |
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions : (i) K and S (ii) Ca and O (iii) Al and N |
Answer» SOLUTION :
|
|
| 13. |
Which of the following show electromeric effect? |
|
Answer» Alkanes |
|
| 14. |
Which of the following is contaminant responsible for Bhopal gas tragedy? |
|
Answer» `CO_2` |
|
| 15. |
Which of the following product is formed when B is treated with hot and conc. NaOH? |
|
Answer» `NACI` and `NaCIO_(3)` (C) `rarr SO_(2)CI_(2),` (D) `rarr H_(2)SO_(4)`, (E) `rarr HCI`, (G) `rarr BaSO_(4)` `underset(("Hot and conc"))(3CI_(2)+6NaOH rarr) 5NaCI +NaCIO_(3)+3H_(2)O` |
|
| 16. |
Which of the following statement (s) is /are not true about the following decomposition reaction. 2KClO_3 rarr 2KCl+3O_2 |
|
Answer» Potassium is undergoing oxidation (a) Since O.N. of potassium does not change , it neither undergoes oxidation nor reduction. (B) O.N. of Cl decreases from +5 `(KClO_3)` to -1 (in KCl), THEREFORE , it is undergoing reduction. (c) O.N. of O increases from -2 (in `KClO_3` ) to zero (in `O_2`) therefore , O is oxidised . (d) This statement is not correct because Cl is undergoing reduction and O is undergoing oxidation. |
|
| 17. |
The true peroxide of the following is |
|
Answer» `MnO_(2)` |
|
| 18. |
When alpha-rays hit a thin foil of gold, very few alpha-particles are deflected back. What does it prove ? |
| Answer» Solution :There is a very SMALL HEAVY body PRESENT within the atom, called NUCLEUS | |
| 19. |
What is meant by setting of cement? |
| Answer» Solution :When gypsum is added to CEMENT by mixing with an adequate quantity of water, it for PLASTIC mass that gets into a HARD solid in 5 to 10 MINUTES. | |
| 20. |
Which of the following organic compounds has same hybridization as its combustion product -(CO_(2)) ? |
|
Answer» Ethane |
|
| 21. |
TLV values of four pollutants A,B,C and D are 2ppm, 9 ppm, 20 ppm and 50 ppm. Among these four pollutants which one is the most toxic pollutant ? |
|
Answer» A |
|
| 22. |
Which one of the following is not an isoelectronic ion? |
|
Answer» `NA^(+)` |
|
| 23. |
What do you understand byallotropy ? |
| Answer» SOLUTION :Allotropy : Allotropy is the existence of an ELEMENT in more than one form, having the same chemical properties but different physical properties. The various forms of an element are called allotropes. For example, carbon exists in three allotropic forms: DIAMOND, GRAPHITE and fullerenes. | |
| 24. |
The restricted rotation about carbon-carbon double bond in 2-butene is due to |
|
Answer» <P>Overlap of one s and `sp^(2)` HYBRIDIZED orbitals |
|
| 25. |
Which one of the following alkaline earth metal carbonates is thermally the most stable ? |
|
Answer» `MgCO_(3)` |
|
| 26. |
Which of the following ion has maximum polarising power |
| Answer» Answer :B | |
| 27. |
When a reaction is conducted in an open vessel, the heat of reaction is represented as |
|
Answer» `DeltaH` |
|
| 28. |
Which of the following species cannot act as both Bronsted acid and base ? |
|
Answer» `H_(2)O` |
|
| 29. |
Which one of the following equations represents the variation of viscosity coefficient (q) with temperature (T)? |
|
Answer» `(ETA) = AE^(-E//RT)` |
|
| 30. |
What is one kilogram ? |
|
Answer»
|
|
| 31. |
The solubility productof a rare earth metal hydroxide M(OH)_3atroomtemperature is4.32 xx 10^(-14)its solubility is |
|
Answer» `2XX 10^(-3) M` `KsP = (s) (3s) ^(3)= 274 ^(4)=4.32 xx 10 ^(-14), S =2 xx 10 ^(-4) ` |
|
| 32. |
Which reagents are suitable for following transformations? |
|
Answer» (b) & (d) Dil. `H_(2)SO_(4)` or (i) Hg `(OAc)_(2),H_(2)O` (ii). `NaBH_(4)//OH^(ɵ)`. |
|
| 33. |
Which of the following is not organic compound? |
|
Answer» DNA |
|
| 34. |
What volume of hydrogen at STP required to reduce 7.95 grams of cupric oxide to give metal? |
|
Answer» SOLUTION :CUPRIC oxide is reduced to copper on treating with hydrogen `CuO+H_(2)to Cu+H_(2)O` 1 mole of CuO =1 mole of `H_(2)` 79.5 grams of CuO=22.4 L of `H_(2)` at STP 7.95 grams of CuO=? VOLUME of hydrogen required at STP `=(7.95)/(79.5) XX 22.4=2.24L` |
|
| 35. |
When Ferrous sulphate acts as reductant, its equivalent weight is |
|
Answer» TWICE that of its MOLECULAR WEIGHT |
|
| 36. |
Which of the following is NOT a redox reaction ? |
|
Answer» `CaCO_(3) RARR CaO + CO_(2)` |
|
| 37. |
Which among the following is softest metal and due to |
|
Answer» `Li lt Na gt K=Rb` |
|
| 38. |
Which method can be applied to separate a mixture of camphor and benzoic acid? |
|
Answer» Sublimation |
|
| 39. |
Which of the following statements is/are correct for alkyl halide ? |
|
Answer» In most unimolecular reactions of alkyl HALIDE `S_N1` reactions is favoured over E1 reaction |
|
| 40. |
Which of the following are soluble in water ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 41. |
What are the defects of Rutherford's model? |
| Answer» SOLUTION :According to J.C. Maxwell, whenever an electron is subjected to acceleration, it emits radiation and loses energy. As a result of this, its orbit should BECOME smaller and smaller and finally it should drop into the nucleus by FOLLOWING a spiral path. This means that atom would collapse and THUS Rutherford.s model FAILED to explain stability of atoms. Another drawback of the Rutherford.s model is that it gives no information about the electronic structure of an atom. | |
| 42. |
Which of the following compounds is used for water softening ? |
|
Answer» `Ca_3(PO_4)_2` `Na_2[Na_4 (PO_3)_6] = Na_6P_6O_18` `2CaCl_2 + Na_2 [Na_4 (PO_3)_6] to Na_2 [Ca_2(PO_3)_6] + 4NaCl` |
|
| 43. |
When 120mg of naphthalene C_(10)H_(8(s)) was burnt in a bomb calorimeter, the temperature rise was 3.05K. Calculate the calorimeter constant and molar standard internal energy change of combustion. Also report by how much will the temperature rise when 100mgof Phenol (C_(6)H_(5)OH_((s))) is burnt in the same calorimeter under the same conditions, if heat liberated is 3962.85J ? (DeltaH_(C)^(@) fo r C_(10)H_(8)=-5157kJ mol^(-1)) |
|
Answer» |
|
| 45. |
Who proposed that the attainment of stable electronic configuration in molecules are achieved by mutual sharing of electrons ? |
|
Answer» G.N. LEWIS |
|
| 46. |
Which of the IVA group element does not exhibit allotropy ? |
|
Answer» C |
|
| 47. |
The total number of electrons that take part in forming bonds in O_2 molecule according to V.B.T. |
|
Answer» 2 |
|
| 48. |
Which are alicyclic or closed chain or ring compounds? Give examples |
Answer» Solution :(a) ALICYCLIC (ALIPHATIC cyclic) compounds: These compounds CONTAIN carbon atoms joined in the form of a RING  (b) HETEROCYCLIC compounds: In a ring of compounds somethimes atoms other than carbon are also present in the ring is known as heterocyclic compounds. 
|
|
| 50. |
The solubility on any substance in water may be classified as : Molecular ability: AB(s/l/g) overset(aq)iffAB(aq) Ionic solubility : AB (s/l/g) overset(aq)iff A^(+)(aq)+B^(-)(aq) For a substance AB(s) the following thermodynamic informations are available at 300K: Molecular solubility: Delta H^(@)=+20kcal/mol, DeltaS^(@)=+40kcal/K-mole Ionic solubility: DeltaH^(@)=-25 kcal/ mol, DeltaS^(@)=-50 cal/K-mole The equilbrium constant for the ionic and molecular solubility of AB(s) in water at 300K are respectively: |
|
Answer» `E^(50//3),e^(-40//3)` |
|