Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following orders of relative strengths of acids is correct ? |
|
Answer» `FCH_(2)COOH gt ClCH_(2)COOH gt BrCH_(2) COOH` `FCH_(2) COOH gt ClCH_(2) COOH gt BrCH_(2)COOH` |
|
| 2. |
Viscosity of a liquid arises due to strong intermolecular forces existing between the molecules.Stronge the intermolecular forces, greater is the viscosity. Name the intermolecular forces existing in the following liquidsand arrangethem in the increasing order of their viscosities. Also give reason for the assigned order in one line. Water, hexane (CH_(3)OHCH(OH)CH_(2)OH) |
|
Answer» Solution :The INCREASING order of viscosity is : Hexane` LT `Water `lt` Glycerine REASON. In hexane, there are only London FORCES whereas in water and glycerine, there is hydrogen bonding which is stronger in case of glycerine (because of the PRESENCE of three-OH groups) than that in water. |
|
| 3. |
What is the boiling point of water at (i) higher altitudes, (ii) in pressure cooker ? |
| Answer» SOLUTION :(i) `GT 100^(@)C "" (II) LT 100^(@)C` | |
| 4. |
The type of isomerism observed in urea molecule is |
|
Answer» chain `{:(""O""OH),("||""|"),(H_(2)N-C-NH_(2) oversetrarr(larr)NH=C-NH_(2)),(""alpha""alpha):}` |
|
| 5. |
Write the equation for calculation of hydrolyses constant of salt of weak acid weak base solution. |
| Answer» SOLUTION :`K_h=K_w/(K_a XX K_b)` | |
| 6. |
The solutions of Schrodinger wave equation gives the allowed ________. |
|
Answer» ENERGY levels |
|
| 7. |
Which of the following statement is false about resonance? |
|
Answer» It increases the STABILITY of a molecule |
|
| 8. |
The Zn acts as sacrified of cathodic protection to prevent rusting of ironbecause: |
|
Answer» `E_(OP)^(@)` of `ZnltE_(OP)^(@)` of FE |
|
| 9. |
Which electronic level would allow the hydrogen atom to emit a photon but not to absorb a photon? |
|
Answer» 3 |
|
| 10. |
The structure of 4-methylpentene-2 is |
|
Answer» `(CH_2)_2 CH- CH_3CH = CH_2` |
|
| 11. |
The smallest stable cyclic alkyne contains ( no.of carbon atoms). |
|
Answer» |
|
| 12. |
What are the general formulae for monocyclic and bicyclic cycloalkanes ? |
| Answer» SOLUTION :`C_nH_(2N)` and `C_n H_(2n - 2)` | |
| 13. |
Which of the following metals dissolve in liquid NH_(3) |
| Answer» Solution :Be. doesn.t dissolve in `NH_(3)`. | |
| 14. |
Which one of the followingrepresents 180g of water? |
|
Answer» 5 MOLES of water =Mass of water / Molar mass of water 180g/18g `"MOL"^(-1)`=10 moles One mole of water contains `6.022xx10^(23)` water molecules 10 moles of water contains=`6.022xx10^(23)xx10=6.022xx10^(24)` water molecules |
|
| 15. |
Which of the following substances is not used for preparing lyophilic sols |
|
Answer» Starch |
|
| 16. |
To get n-type semicoductor from silicon, it should be doped with a substance with group no.…… |
|
Answer» 12 |
|
| 17. |
Which one of the following statements is incorrect with regard to ortho and para dihydrogen ? |
|
Answer» They are NUCLEAR spin isomers Para isomer - zero nuclear spin |
|
| 18. |
What type of interactions hold the molecules together in a polar molecular solid ? |
| Answer» SOLUTION :Dipole-Dipole ATTRACTIONS. | |
| 19. |
Type of hybridisation of central atom of gas (B): |
|
Answer» `SP^(3)` `I rarr PbCrO_(4) (ACOH` insoluble) `HNO_(2) +I^(-) +H^(+) rarr NO` (Gas H) |
|
| 20. |
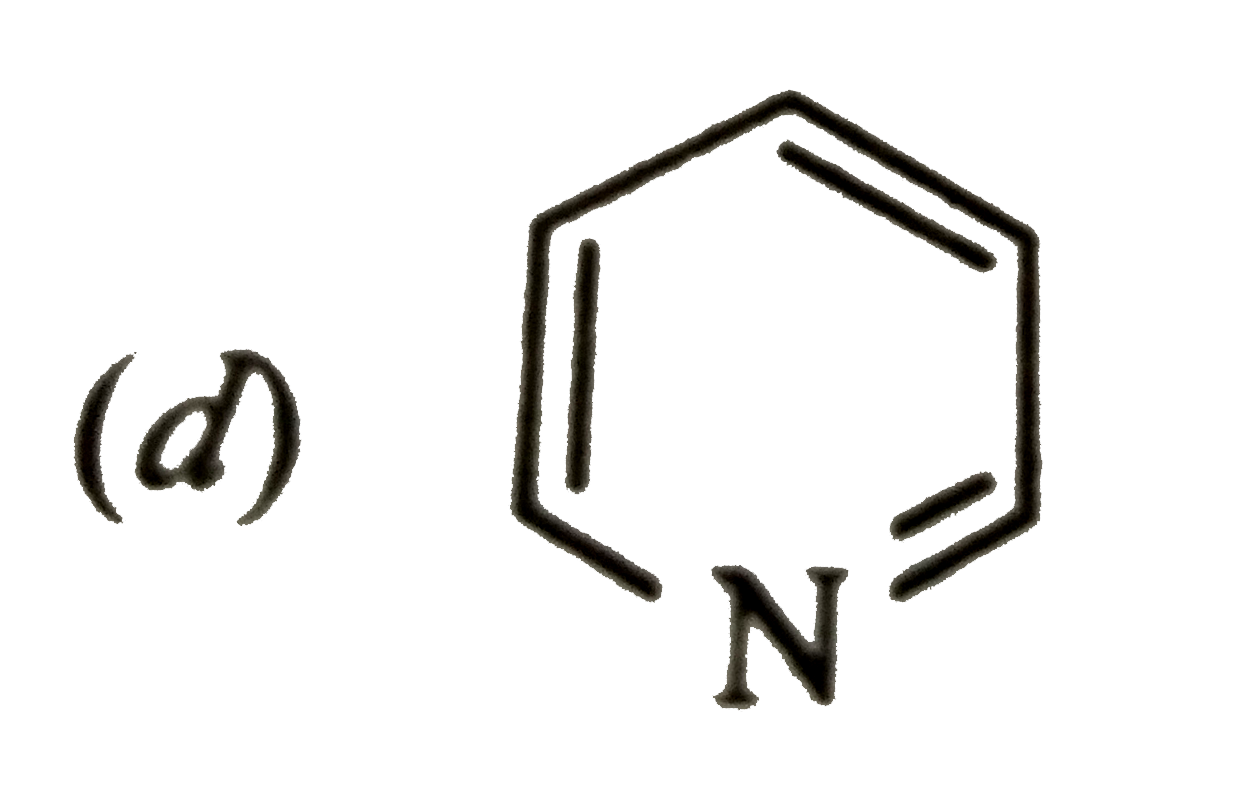
Whichof the following compoundsis not aromatic ? |
|
Answer»
|
|
| 21. |
Which law of thermodynamics gives information about exact entropy of a substance ? |
|
Answer» Zeroth Law `DELTA H = q_("rev") and Delta S= (Delta H)/( T) and Delta S= S_("FINAL state") -S_("initial state")` |
|
| 22. |
Which of the following reagents can be empolyedfor isomerisation of n-butane? |
|
Answer» HI/P |
|
| 23. |
Which of the following carbanion expected to be most stable? |
|
Answer» `p-NO_(2)C_(^)H_(4)overset(-)(C)H_(2)` |
|
| 24. |
When an element sulphur atom becomes a sulphide ion, "______________". |
|
Answer» there is no change in the composition of atom |
|
| 25. |
The wavelength associated with a golf ball weighing 200g and moving at a speed of 5 m/h is of the order |
|
Answer» `10^(-10)m` |
|
| 26. |
Which of the following compound will give three alkenes after dehydrohalogenation. |
|
Answer» `CH_3-CH_2-CH_2-CH_2-BR` |
|
| 27. |
Which of the following pair of solutions does not form a buffer solution? |
|
Answer» ` NaH_2PO_4 and Na_2HPO_4` |
|
| 28. |
Which of the following shows geometrical isomers ? |
|
Answer» `CH_(2) = CHCl` 
|
|
| 29. |
Which of the following transfer of electron pair from pi bond to its adjacent bond? |
|
Answer»
|
|
| 32. |
Two metals X and Y belong to the cond group of periodic table. X forms insoluble oxide but soluble sulphate. Y forms a soluble oxide but insoluble sulphate. Hydroxide of metal X is soluble in NaOH while that of metal Y is insoluble in NaOH. What are metals X and Y? |
|
Answer» X=Be,Y=BA `Be(OH)_(2)`-soluble in NAOH `Ba(OH)_(2)`-insoluble in NaOH |
|
| 33. |
Write the chemical equationwhen borax dissolve in water ? |
| Answer» | |
| 34. |
Which metal carbonates are majorly present in ash of plant herb ? |
| Answer» ANSWER :D | |
| 35. |
What are the equivalent volumes of oxygen and chlorine at STP? |
|
Answer» |
|
| 36. |
What will be bond angle C-O-H in alcohol if C and O atom possess sp^(3) hybridizations? |
|
Answer» `190^(@) 28'` |
|
| 37. |
Write the structural formula and carbon skeleton formula for all possible chain isomer of C_(6)H_(14) (Hexan) |
Answer» Solution :`C_6H_(14) ` has five POSSIBLE ISOMERIC STRUCTURE 
|
|
| 38. |
which of the following is not true about the ionic solids ? |
|
Answer» Bigger IONS from the close packed structure |
|
| 39. |
What will be the mass of one""^( 12)C atom in g? |
|
Answer» Solution :1 mol of `""^(12)C` atom , i.e. `6.022 xx 10^(23)` atoms weigh 12 G. `THEREFORE` Mass of one `""^(12)C` atom `=12/(6.022 xx 10^(23)) = 1.9923 xx 10^(-23)` g |
|
| 40. |
Which of the following explains the sequence of filling electrons in different subshells? |
|
Answer» HUND's rule |
|
| 41. |
Which bond is formed by overlapping of 2p_(x) with 2p_(y) ? Why ? |
| Answer» Solution :The BOND is not formed by this COMBINATION because `2p_(x) and 2p_(y)` are PARALLEL to each other. So no overlapping by each other. | |
| 43. |
What is formed when calcium carbide react with heavy water? |
|
Answer» `CD_2` |
|
| 44. |
Whoproposedthat electronhas wavenature ? |
|
Answer» Schrodinger |
|
| 45. |
What will be the wavelength of oxygen molecule in picometers moving with a velocity of 660 m s^(-1) (h = 6.6 xx 10^(-34) kg m^(2) s^(-1)) |
|
Answer» |
|
| 46. |
What is the difference between alkyl group and alkane? |
|
Answer» ONE CARBON is more in alkane |
|
| 47. |
The strength of the covalent bond in H_(2) , F_(2) and HF is in the order |
|
Answer» `H-HgtF-FgtH-F` Bond strengths `(KJ MOL^(-1)) ` 566436126 This is because they involve s-p,s-s and p-p OVERLAP respectively.Greater the overlap , stronger is the bond . |
|
| 48. |
Which among the following molecule obeys octet reule ? |
|
Answer» `NO` |
|
| 49. |
Using the data provided, calculate the bond energy( kJ mol^(-1))of a C-=C bond in C_(2)H_(2).That eneergy is ( take the bond energy ofC-H bond as350 kJ mol^(-1))2C(s) + H_(2)(g) rarr C_(2)H_(2)(g)" " DeltaH = 225 kJ mol^(-1) 2C(s) rarr 2C(g) " " DeltaH = 1410 kJ mol^(-1) H_(2)(g) rarr 2H(g)" " DeltaH = 330 kJ mol^(-1) |
|
Answer» 1165 (II) `2C(s) rarr 2C(g) , DeltaH = 1410kJ mol^(-1)` (iii) `H_92)9g)rar 2H(g), DeltaH = 330 KJ mol^(-1)` From eq. (i) `225= [2 xxDelta_(C(s) rarr C(g)) + BE_(H-H)]-[2BE_(C-H)+BE(C-=C)]` or `225=[ 1410 + 330]-[2 xx 350 k+ BE_(C-=C)]` or ` 225=1740- 700- BE_(C-=C)` or`BE_(C-=C) 1040-225 = 815kJ mol^(-1)` |
|
| 50. |
What is Green chemistry ? |
|
Answer» Solution :(i) Green CHEMISTY is chemical PHILOSOPHY encouraging the design of products and PROCESSES that reduces or eliminates the use and genration of hazardous substances. (ii) Efforst to control enviromental pollution RESULTED in development of science for the SYNTHESIS of chemicals favourable to enviroment . (iii) Green chemistry means science of enviromentally favourable chemical sysnthesis. |
|