Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which among the followings shows maximum oxidation state ? |
| Answer» Solution :`OVERSET(+7)(KMnO_4)` | |
| 2. |
underset(("Hydrated salt"))((A))underset("heated")overset("Strongly")rarr ubrace(B+C)_("Transparent glassy bead") H_(2)BO_(3) overset("Strongly")underset("heated")rarr C+D Find the number of water of crystallization in (A): |
|
Answer» 4 |
|
| 3. |
What are thermochemical equation ? |
| Answer» Solution :A thermochemical EQUATION is a balanced STOICHIOMETRIC chemical equation that includes the ENTHALPY CHANGE `(DeltaH)`. | |
| 4. |
The saponification number of fat or oil is defined as the number of mg of KOH required to saponify 1 g oil or fat. A sample of peanut oil weighing 1.5763 g is added to 25 mL of 0.421 M KOH. After saponification is complete, 8.46 mL of 0.2732 M K_(2)SO_(4) is needed to neutralise excess of KOH. What is the saponification number of peanut oil ? |
|
Answer» |
|
| 5. |
What is Hunsdiccker reaction? |
Answer» SOLUTION :SILVER salts of fatty acids when refluxed with BROMINE in `C Cl_(4)` gives bromo ALKANE.  . .
|
|
| 7. |
Which of the following is an element present in the d-block, but not a transition element? |
|
Answer» Cd |
|
| 8. |
Why alkali metals exhibit +1 oxidation state only in their compounds? |
| Answer» Solution :UNIPOSITIVE ions of alkali metals POSSESS stable inert gas ELECTRONIC configuration. Thus second ionisation potential of alkali metal is very high and they exhibit `+1` oxidation STATE only in their compounds. | |
| 9. |
What are carbocations? |
|
Answer» Solution :(i) Let us CONSIDER the hetrolytic fission of the bond C-X present in organic molecule. If the ATOM X has greater ELECTRONEGATIVITY than the carbon atom, the former takes away the bonding electron pair and becomes negatively charged while the carbon will lose its electron and thus acquire a positive charge. (ii) Such a cationic species CARRYING a positive charge on carbon are known as carbocation orcarbonium ion. 
|
|
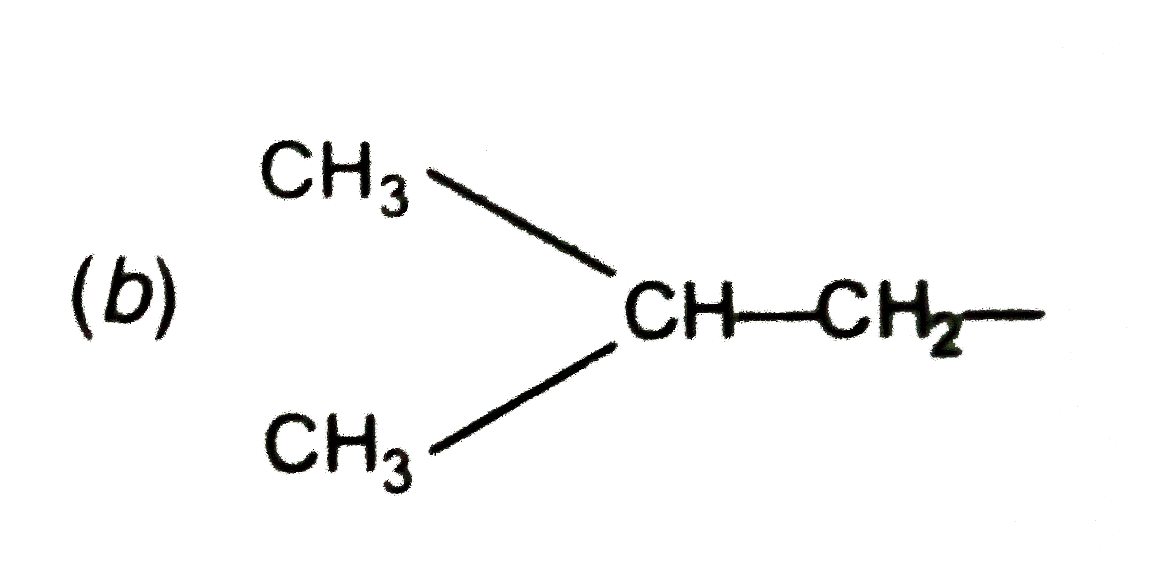
| 10. |
The structure of isoburtyl group in an organic compound is : |
|
Answer» `{:(""CH_(3)),("|"),(CH_(3)-C-),("|"):}` |
|
| 11. |
Which of the following elements will have the highest electronegativity ? |
|
Answer» CHLORINE |
|
| 12. |
Which of the following does not exist ? |
| Answer» Answer :A | |
| 13. |
The value of Rydberg constant is |
| Answer» Answer :A | |
| 14. |
Which of the following halides undergoes hydrolysis on warming with "water" // "aqueous" NaOH? |
|
Answer»
|
|
| 15. |
Write the resonance structure of CO_2 . |
|
Answer» |
|
| 16. |
Very slight decrease in atomic radius occurs in a transition series when compared with that in representative series. This is due to |
|
Answer» Shielding effect |
|
| 17. |
Which of the following os the incorrect name of CH_(3)NC? |
|
Answer» (1) Methyl ISOCYANIDE `CH_(3)NCoverset(H_(2)O)rarrCH_(3)NH_(2)+underset("Formic acid")(HCOOH)` |
|
| 18. |
Write the position isomers of diethyl ether. Write their structures and IUPAC names. Can these be also regarded as metamers ? |
|
Answer» Solution :Two position isomers (I and II) are possible of diethyl ether. `underset("Diethyl ether")(CH_(3)CH_(2)-O-CH_(2)CH_(3)) "" underset("1-Methoxypropane")(CH_(3)O-CH_(2)CH_(2)CH_(3))``{:(""CH_(3)),("|"),(CH_(3)O-CH-CH_(3)),("2-Methoxypropane (II)"):}` Both these can ALSO be regarded as METAMERS of diethyl ether because these differ in the number of carbon ATOMS on either side of the ethereal OXYGEN. |
|
| 19. |
The wavelength of m^(th) line balmer series for an orbital is 4103Å. What is the value of m? |
|
Answer» |
|
| 21. |
What is the volume strength of 100 ml of aqueous H_2O_2 solution containing 34 gm of H_2O_2 |
|
Answer» `5.6` VOL |
|
| 22. |
Which of the following compound is known as oil of wintergreen ? |
|
Answer» <P>METHYL SALICYLATE |
|
| 23. |
Which one of the following sets of ions represents a collection of isoelectronic species? |
|
Answer» `Ba^(+2), Sr^(+2), K^(+), CA^(+2)` |
|
| 24. |
Which of the following is water soluble compound of calcium ? |
| Answer» Answer :A | |
| 25. |
When BeCl_(2) is hydrolysed, white fumes of gas are given out. The intensity of fumesintensifies when a rod dipped in moist ammonia is brought near the mouth of the test tube. The gas which comes out during hydrolysis is |
|
Answer» `Cl_(2)` |
|
| 26. |
Which of th following is not true about the voids formed in 3 dimensional hexagonal close packed structure ? |
|
Answer» A tetrahdral void is formed when a SPHERE of the second layer is present above trinaglar void in the first layer. (d)is not true because octadedral void is combination of TWO trianglar voids one with vertex upward and the other with vertex downward and not their overlap. |
|
| 27. |
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride. |
|
Answer» Aniline |
|
| 28. |
The reducing power of a metal depends on various factors . Suggest the factor which makes Li, the strongest reducing agent in aqueous solution . |
|
Answer» SUBLIMATION enthalpy |
|
| 29. |
What is solubility of salt?Explain classification of salt based on solubility ? |
Answer» SOLUTION :Each salt has its own CHARACTERISTICS . The no. of MOLES of SOLUTE disslove in 1 lit. at definite TEMPERATURE is called solubility of salt. 
|
|
| 31. |
Which one of the following is paramagnetic and having 0.5 bond order ? |
|
Answer» `O_(2)^(-)` |
|
| 32. |
Which of thefollowingconfiguration representsatom of theelement havingthe highestsecondionizationpotential ? |
|
Answer» ` 1s^(2)2 s^(2)2p^(4)` |
|
| 33. |
Two elements 'A' and 'B' (atomic weights 75 and 16 respectively) combine to give a compound having 75.8% of 'A'. The compound has the formula (St. John's) |
|
Answer» XY |
|
| 34. |
Which one of the following hexachlorocyclohexane is least reactive and which one is most reactive for E2 reaction with a strong base for dehydrohalogenation. |
|
Answer» I LEAST & II least 
|
|
| 35. |
Which statement is correct about photochemical bromination of Butane? CH_(3)-CH_(2)-CH_(2)-CH_(3)overset(Br_(2).hupsilon)to |
|
Answer» 1-Bromobutane and 2-Bromobutanes are formed in equal amounts |
|
| 36. |
What is the formula mass of gypsum? |
|
Answer» SOLUTION :Formula of GYPSUM is `CaSO_(4)2H_(2)O` Formula mass of `CaSO_(4)2H_(2)O`=`(6xx12u)+(12xx1u)+(6xx16u)=180u`. |
|
| 37. |
Which of the following optionswill have compressibility factor greater than 1? |
|
Answer» `H_(2)` gas at its CRITICAL condition. |
|
| 38. |
Which of the following orbitals will have zero probability of finding the electron in the pz plane |
|
Answer» px |
|
| 39. |
Which of the following compounds of Be has polymeric structure |
|
Answer» `BeH_(2)` |
|
| 40. |
Which of the following statements are not true for hydrogen ? |
|
Answer» It exists as DIATOMIC molecule |
|
| 41. |
When the bond form according to VB theory ? |
| Answer» SOLUTION :If the ORBITALS of TWO atoms arrange an INTERNUCLEAR axes and overlap then covalent bond is form. | |
| 42. |
The correct increasing order of densities of alkali metals is : |
|
Answer» `LI lt K lt Na lt Rb` |
|
| 43. |
Writethe chemical equations for comubstionof propane |
|
Answer» Solution :Chemicalequationforcombustionof propane:Thegeneralcombustion reactionfor anyalkaneis `C_(n) H_(2n)+2 + ((3n+1)/( 2)) O_(2)to nCO_(2)+ (n+1)H_(2)O` `CH_(3) -CH_(2)-CH_(3) + 5O_(2)to 3CO_(2) + 4H_(2) O` `Delta H^(@) =- 101.6 kj` |
|
| 44. |
Which one of the following compounds can be deprotonated by OH fastest ? |
|
Answer» `HCOOH, pK_(a)=3.8` |
|
| 45. |
Which one of the following is not a path function |
|
Answer» Work |
|
| 46. |
Which elements having more positive and more negative electron gain enthalpy? |
| Answer» SOLUTION :ELEMENTS of noble gas having more POSITIVE and elements of halogens having more negative electron gain enthalpy. | |
| 47. |
What happens when a. Sodium metal is dropped in water? b. Sodium metal is heated in free supply of air? c. Sodium peroxide dissolves in water? |
|
Answer» Solution :(i) `H_(2)` gas is evolved which catches FIRE due to the exothermicity of the reaction . `2 Na (s) + 2 H_(2)O (l) to 2 NaOH (aq) + H_(2) (g)` (ii) `Na_(2)O_(2)` along with a small amount of `Na_(2)O` is formed . `2 Na(s) + O_(2)(g) to 2 UNDERSET("(minor)") (2 Na_(2)O(s))` `Na_(2)O(s)+ (1)/(2) O_(2) (s) to underset("(major)")(Na_(2)O_(2)(s))` (III) `H_(2)O_(2)` is formed `Na_(2)O_(2) (s) + 2H_(2)O (l) to 2 NaOH (aq) + H_(2)O_(2) (l)` |
|
| 48. |
Which d-orbital does not have four lobes and what is its shape called ? |
| Answer» SOLUTION :`d_(Z)^(2)` which has dough-nut SHAPE. | |
| 49. |
Which of the S-block element doesn't react with water. |
|
Answer» K |
|
| 50. |
What are the values of w and DeltaE, when a system absorbs 250Jof heat by expanding from 1 lit to 10 lit against 0.5 atm pressure and at constant temperature. |
|
Answer» Solution :`W=-P Delta v =- 0.5 (10 -1) =- 4.5 `lit - atm WOrk DONE, `w =- 4.5 XX 101 J =- 454 .5 J.` `Delta E =q - w = 250 -454.5 =-204 .5 J.` |
|