Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the importance of Le-chatelier's principle ? |
|
Answer» Solution :From the factors affecting to equilibrium LIKE temperature, pressure, concentration any ONE change than the reaction proceed in which direction can be predicted. Equilibrium constant is independent of initial concentration but in equilibrium system Le chatelier.s PRINCIPLE is useful to PREDICT the concentration of COMPONENTS in new equilibrium and equilibrium moves in which direction by change of concentration, temperature, pressure etc. |
|
| 2. |
What volume of carbondioxide is obtained at STP by heating 8g of 50% pure limestone? |
|
Answer» |
|
| 3. |
When benzene is treated with DCl at low temperatures a compound C_(6)H_(6)DCl, is formed. On warming the reactants, C_(6)H_(6) and DCl are re-formed. However, in the presence of AlCl_(3) an isomer of C_(6)H_(6)DCl is produced, which on warming, gives mainly C_(6)H_(5)D and HCl. Explain. |
| Answer» Solution :Since D is not incorporated into the BENZENE I ring in the absence of `AlCl_(3)` isomer C has no C-D `sigma` bond. Instead, the D end of DC is attracted to the `pi` cloud of benzene to form a loosely BOUND `pi` complex, indicated by an arrow in the structural formula. `pi` complexes are also called charge -transfer complexes, because the electron-rich benzene ring,t he donor molecule transfers some electron density to the electron-poor D of DCl, ACCEPTOR molecule. wit the catalyst `AlCl_(3)`, exchange of `D^(+)` for `H^(+)` occurs o WARMING. this exhange requires the formation of a C-D bond to give the `sigma` complex. with `D^(+)`. the benzene ring is reformed by losing an H rather than the D because the C-H bond is weaker than the C-D bond. | |
| 4. |
Which of the following exhibits geometrical isomerism ? |
|
Answer» 1,2-Dibromopropene |
|
| 5. |
Which of the following conformations of meso 2,3-dibromobutane will give bromoalkene with alcoholic KOH ? |
|
Answer» GAUCHE form 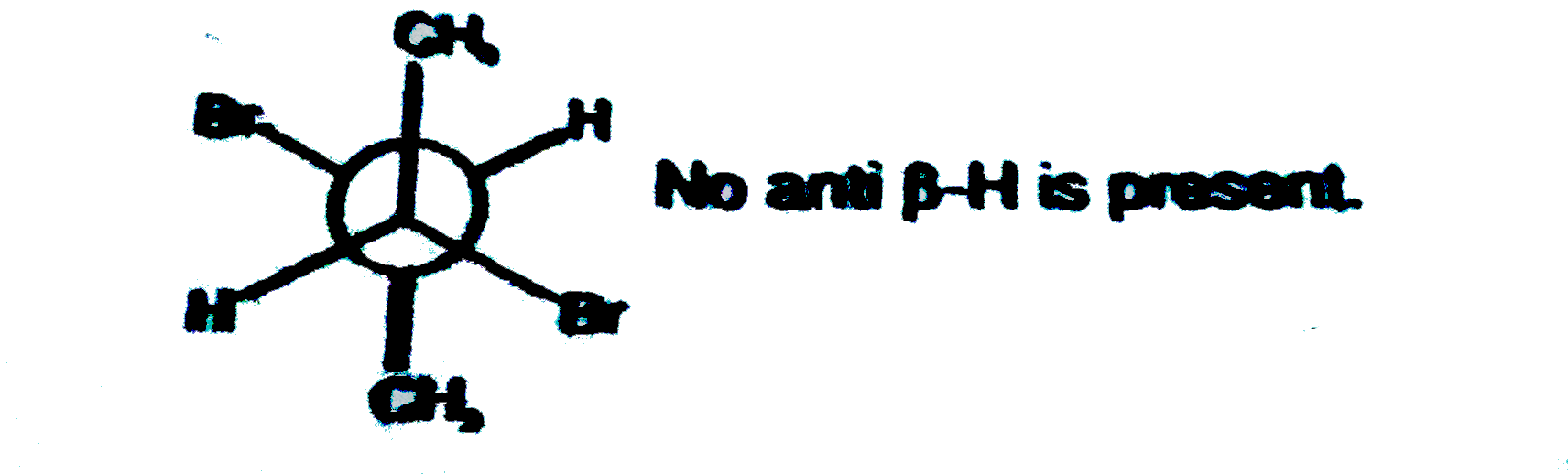
|
|
| 7. |
Which of the following pair functional groups represents ambident nucleophiles? |
| Answer» Solution :`-CN & - NO_2` | |
| 8. |
Write the energy level diagram of hydrogen molecule. |
Answer» Solution :Atomic number of hydrgen is 1=1s. 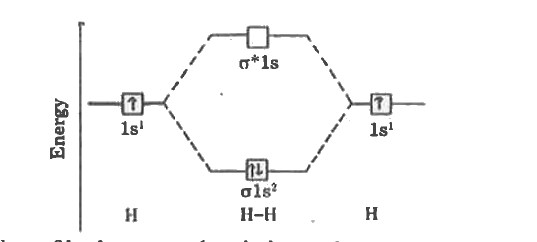 (a) ELECTRONIC configuration of hydrogen molecule is `SIGMA1S^(2)sigma^(**)1s^(@)` (b) Bond order `=(2-0)/(2)=1` (3) MAGNET property =DIAMAGNETIC. |
|
| 9. |
What is the binding force between molecules, if a substance is a gas under ordinary conditions of temperature and pressure ? |
| Answer» SOLUTION :VAN DER WAALS FORCES | |
| 10. |
Which of the following is mainly responsible for depetion of ozone layer? |
| Answer» Answer :A | |
| 11. |
Which of the following containing the positive resonance effect ? |
| Answer» Answer :D | |
| 12. |
Which element among the following canot exhibit variable electrovalency? |
|
Answer» `""_(29)Cu` |
|
| 13. |
Which of the following element's outermost orbits Ist electron has magnetic quantum number m = 0 ? |
| Answer» Answer :A | |
| 14. |
Which of the following is used for the seperation of ZnS and PbS during the froth floatation process ? |
|
Answer» KCl |
|
| 15. |
Which of the followingchanges decrease the vapour pressure of water kept in a sealed vessel ? |
|
Answer» Decreasing the quantity of water |
|
| 16. |
Which of the following has minimum flocculation value ? |
|
Answer» `PB^(2+)` |
|
| 17. |
What is the formula of carbon suboxide ? |
| Answer» Solution :`C_(3)O_(2)[O=C=C=C=O]` is a CARBON subdioxide | |
| 18. |
What is Isobars? |
| Answer» Solution :Atoms of different elements having the same MASS number but different ATOMIC NUMBERS are called isobars. | |
| 19. |
What is thesimilarityin 2p_(x ),2p_(y) and2p_(z) ? |
|
Answer» SOLUTION :n=2,L =1IS same Allthreehave samedumbbellshape. Theirshape sizeand ENERGYIS same. |
|
| 20. |
Whichare isoelectronicfrom thefollowing |
| Answer» Answer :D | |
| 21. |
Write the general electronic configuration of S-block elements. |
| Answer» SOLUTION :[Noble gas] `NS^(1 "to" 2)` | |
| 23. |
Under the same conditions the rates of diffusion of two gases are in the ratio 1:4. The ratio of their vapour densities is |
|
Answer» `2:1` |
|
| 25. |
The standard enthalpiesof formation ofCO_(2)(g), H_(2)O(l)and glucose (s)at 25^(@)C are-400 kJ //mol, - 300 kJ//mol and-1300kJ//molrespectively.The standard enthalpy of combustion per gram of glucose at25^(@)C is |
|
Answer» ` + 2900kJ` `Delta_(r)H^(@)=Delta_(c) H^(@) = [ 6 Delta_(F)(CO_(2)) + 6 Delta_(f)H^(@) ( H_(2)O)]- [ Delta_(f) H^(@) ( C_(6)H_(12)O_(6)) + 6 Delta_(f) H^(@) ( O_(2))]` `= 6 xx ( - 400) + 6 ( - 300) -( - 1300)` `= -2900 kJ` `= - ( 2900)/( 180 ) kJ //g =- 16.11 kJ //g` |
|
| 26. |
Which of the following cannot be used in the following raction as L ? B_(2)H_(6)+2Lto[BH_(3).L] |
|
Answer» `PF_(3)` |
|
| 27. |
Which one of the following pairs is not correctly matched |
|
Answer» Rutherford-Proton |
|
| 28. |
Which of theconfigurationof mostelectronegative elements is |
|
Answer» `[He] 2S^(1)` i.e.,`CS [He] 6s^(1)` |
|
| 29. |
Which of the following statements is true for the reaction given below ? |
|
Answer» P is a meso COMPOUND of 2,3-butanediol FORMED by syn addition. |
|
| 30. |
Which of the following reacts with metal by displacing the hydrogen atom? |
|
Answer» `CH_4` |
|
| 31. |
To a 25 ml H_(2)O_(2) solution, excess of acidified solution of potassium iodide was added. The iodine liberated required 20 ml of 0.3 N sodium thiosulphate solution. The volume strength of H_(2)O_(2) solution is |
|
Answer» `1.344` `(25)/(1000)XXN=(20xx0.3)/(1000)` `N=(1.2)/(5)=0.24N=1.344V` |
|
| 32. |
Which is the correct product formed from the bromination of nitrobenzene? |
|
Answer»
|
|
| 33. |
Which of the following compound is not chiral ? |
|
Answer» 1-chloropentane |
|
| 34. |
The velocityassociatedwith aprotonmovingin apotentialdifferenceof 100 v is4.37 xx 10^(5) ms^(-1).ifthe hockeyballof mass0.1kg ismovingwith thisvelocity, calculatethewavelengthassociatedwiththisvelocity |
|
Answer» Solution :`h= 6.626 XX 10^(34)J s=6.625 xx 10^(34)kg m^(2)s^(-10` `v=4.37 xx 10^(5) ms^(-1)m=-0.1 kg` Accordingto DEBROGLIE `lambda= (h)/(mv)= (6.626 xx 10^(34) kg m^(2) s^(1))/(0.1kg 4.37 xx 10^(5) ms^(1))` `=1.516xx 10^(38)m` `=1.516 xx 10^(26) pm` Note, thiswavelengthis least.Sothe BALL with high speeddoes not posseswavenature. |
|
| 35. |
Which one of the following has no stereogenic center? |
|
Answer»

|
|
| 36. |
The total number of isomers of C_(4)H_(10)O will be |
|
Answer» 4 (ii) `underset(("Butanol-2"))underset("SECONDARY butyl alcohol")(CH_(3)CH_(2)OVERSET(OH)overset(|)(CH)CH_(3))` (iii) `underset("2-Methyl propanol")underset("Iso butyl alcohol")(CH_(3)-underset(CH_(3))underset(|)(CH)-CH_(2)OH)` (iv) `underset(("2-MEthyl propanol-2"))underset("Tertiary butyl alcohol")(CH_(3)underset(CH_(3))underset(|)overset(CH_(3))overset(|)(CH)OH)` |
|
| 37. |
Which of the following can produce hydrogen from water ? |
|
Answer» Heated stannic oxide `3Fe+4H_(2)O ("hot")toFe_(3)O_(4)+4H_(2)` |
|
| 38. |
Why are liquids and gases categorised as fluids ? |
|
Answer» |
|
| 39. |
Which of the following alkanes give only one mono-chloro derivative |
|
Answer» 2, 3-dimethylbutane give only one MONO chloro Derivative. |
|
| 40. |
What are all the advantages of Kjeldahl's method? |
| Answer» SOLUTION :Kjeldahl.s METHOD: thismethodis carriedmuchmoreeasilythan theDumsmethodIt isusedlargelyin theanalysisoffoods andfertilizers . | |
| 41. |
The similarity between lithium and magnesium is observed due to... |
|
Answer» Size |
|
| 42. |
which of thefollowingrepresentsberyllateion? |
|
Answer» `[Be (OH)_(4) ]^(3-)` |
|
| 43. |
Which range includes the value of the equilibrium constant, K_(eq), for a system with DeltaG^(circ) lt 0 ? |
|
Answer» `-1 LT K_(EQ) lt 0` |
|
| 44. |
Which of the following sets contain only isoelectionic ions? |
|
Answer» <P>`Zn^(2+),Ca^(2+),Ga^(3+),Al^(3+)` |
|
| 45. |
Which of the following is not correct? |
|
Answer» Anhydrous `AICI_(3)` exists as `Al_(2)Cl_(6)` |
|
| 46. |
What is the limitation of framework model? |
| Answer» Solution :In framework model. (i) It is not indicate the ATOM (II) This model is not give information about ATOMIC SIZE. (III) This model emphasizes the pattern of bonds of a molecule | |
| 47. |
What is the correct from the following for the reaction H_(2) O_((l)) hArr H_(2) O_((g)) at 100^(@) C and 1 atm. Pressure ? |
|
Answer» `DELTA E =0` |
|
| 48. |
To neutralize compltely 20 ml of 0.1 M aqueous solution of phosphorus acid, the volume of 0.1 molal aqueous KOH solution required is |
|
Answer» 10 ml `2xx0.1xx20=1xx0.1xxV_(2)impliesV_(2)=40ml` |
|
| 49. |
What is functional group isomerism? Explain by suitable example |
|
Answer» Solution :Two or more COMPOUNDS having the same molecular formula but different functional groups are called functional isomers and this phenomenon is termed as functional group ISOMERISM Expample: The molecular formula `C_(3)H_(6)O` represents an aldehyde and a KETONE. `CH_(3)- CH_(2) - overset(overset(H)(|))(C )=O and underset("Propanal (Propionaldehyde)")(CH_(3)CH_(2)CHO) and `underset("Propanone (Acetone)")(CH_(3)- underset(underset(O)(||))(C )-CH_(3))` |
|
| 50. |
What is a vapour pressure of liquid ? What is relative lowering of vapour pressure ? |
|
Answer» <P> Solution :(i) The pressure of the vapour in equilibrium with its liquid is called vapour pressure of the liquid at the GIVEN temperature.(II) The relative lowering of vapour pressure is defined as the RATIO of lowering of vapour pressure to vapour pressure of pure SOLVENT. Relative lowering of vapour pressure `=(P _("solvent")-P _("solution"))/(P _("solvent"))` |
|







