Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following is an example for closed system? |
|
Answer» Hot water CONTAINED in a thermos flask |
|
| 2. |
Which of the following can not produce acetyl chloride ? |
|
Answer» calcium acetate and sulphuryl chloride |
|
| 3. |
What weight of zinc would be required to produce enough hydrogen by reacting with dilute HCl to reduce completely 8.5 g of copper oxide to copper ? |
|
Answer» `Zn+2HClrarrZnCl_(2)+H_(2)` `CuO+H_(2)rarrCu+H_(2)O` `underset(65g)(Zn)+underset(63.5+16)(2HCl)+underset((=79.5g))(CuO)rarrZnCl_(2)+Cu+H_(2)O` 79.5 G of CuO require Zn = 65 g 8.5 g of CuO require `Zn=(65)/(79.5)xx8.5=6.95g`. |
|
| 4. |
Write redox couples involved in the reactions (i) to (iv)given in question 31 . |
|
Answer» Solution :`(i) ZN(s)|Zn^(2+)(aq),CU^2+)(aq)|Cu(s)` `(ii) MG(s)|Mg^(2+)(aq),Fe^(2+)(aq)|Fe(s)` `(III)CI_(2)(g)|2CI^(-)(aq),2Br^(-)(aq)|Br_(2)(g)` `(iv) Fe(s)|Fe^(2+)(aq),Cd^(2+)(aq)|Cu(s)` |
|
| 5. |
Which of the indicated relationship is correct for the following exothermic reaction carried out at constant pressure? CO_((g)) + 3H_(2(g)) rarr CH_(4(g)) + H_(2)O_((g)) |
|
Answer» `DELTA E = Delta H` |
|
| 6. |
The standard oxidation potential of Zn ,Cu Ag and Ni electrode are +0.76 -034 -0.80 and +0.25 V respectively which of the following reaction will provide maximum volage ? |
|
Answer» `Cu + 2 Ag^(+) (aq)rarrCu^(2+)(aq)+2 Ag` `THEREFORE E.N.F =E_("cathode")E^(@)-E_("anode")^(@)` =+0.80 -(-0.76)=+1.56V thus option (b) iscorrect |
|
| 7. |
The reducing power of a metal depends on various factors. Suggest the factor which makes lithium, the strongest reducing agent in aqueous solution. |
|
Answer» SUBLIMATION ENTHALPY |
|
| 8. |
Write the structural formula of the following, (a) 2 - Butenamide B) Ethylethanote (c ) 2-Methyl-3-hexyne (d) Propanoic anhydric (e ) Pentan - 2,4 - dione |
|
Answer» Solution :(a) `CH_(3) - CH = CHCONH_(2)` (b) `CH_(3)COOC_(2)H_(5)` (C ) `CH_(3)CH_(2)C -= C UNDERSET(underset(CH_(3))(|))(C )HCH_(3)` (d) `CH_(3)-CH_(2)-CO-O-OC- CH_(2)-CH_(3)` (e ) `CH_(3)COCH_(2)COCH_(3)` |
|
| 9. |
Tick mark the wrong match. |
|
Answer» MORPHINE - narcotic |
|
| 10. |
The spectrum obtained from incandescent solids is |
|
Answer» CONTINUOUS |
|
| 11. |
Which one of the following statements about carbon monoxide is correct ? |
|
Answer» It has two lone PAIRS of electrons on oxygen atom hybridisation of C-atom . Because of donation of electron by O-atom to C-atom ,it has low polar character and HENCE low dipole moment ` (0.39 C m/0.117 D)`. |
|
| 12. |
Which of the following compound contain nitrogen atom in its -1 oxidation state ? |
|
Answer» `NH_(2)-NH_(2)` |
|
| 13. |
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 seconds respectively. The molecular mass of A is 49 u. Molecular mass of B will be |
|
Answer» 25.00 u |
|
| 14. |
Which of the following statements regarding the above reaction is correct ? |
|
Answer» A & B are enantiomers. 
|
|
| 15. |
Which of the following statements is/are false |
|
Answer» it is the isotropic nature of AMORPHOUS solids that all the bonds are of equal strength |
|
| 16. |
What is the difference between the forms .hydrolysis. and .hydration.. |
|
Answer» Solution :(iii) Hydrolysis Reaction : Due to high dielectric constant, it has a very strong hydrating tendency. It dissolves many ionic compounds. However, certain covalent and some ionic compounds are hydrolysed in water. `P_4O_(10(s)) + 6H_2O_((l)) to 4H_3PO_(4(aq))` `SiCl_(4(l)) + 2H_2O_((l)) to SiO_(2(s)) +4HCl_((aq))` `N_((s))^(3-) +3H_2O_((l)) to NH_(3(g)) + 3OH_((aq))^(-)` (IV) Hydrates Formation: From aqueous solutions many salts can be crystallised as hydrated salts. Such an association of water is of different types viz., (i)Coordinated water e.g., `[CR(H_2O)_6]^(3+) 3Cl^(-)` (II)interstitial water e.g., `BaCl_2. 2H_2O` (iii) hydrogen-bonded water e.g., `[Cu(H_2O)_4]^(2+) SO_4^(2-) . H_2O` in `CuSO_4 . 5H_2O` |
|
| 17. |
When acetylene is treated with ammonical cuprous chloride, a ............ precipitate of ............ is formed. |
| Answer» SOLUTION :RED, COPPER ACETYLIDE | |
| 18. |
To explain the nature of radiation, James Clark Maxwell in 1864 put forward .Electromagnetic wave theory.. This theory could explain the phenomena of interference and diffraction but could not explain phenomena of black body radiation and photoelectric effect. Also, after the advent of electromagnetic wave theory, Rutherford.s model of atom suffered a serious drawback. To explain the phenomena of black body radiation and photoelectric effect, Max planck in 1900 put forward Planck.s quantum theory. BAsed on this theory, Neils Bohr in 1913 put forward Bohr.s model of atom which could overcome the drawback of Rutherford.s model and also explain the line spectral elements, especially the line spectra of hydrogen and hydrogen-like particles. Kinetic energy of the electron ejected when yellow light of frequency 5.2 xx 10^(14) s^(-1) falls on the surface of potassium metal (threshold frequency = 5 xx 10^(14) s^(-1)) is |
|
Answer» `1.325 XX 10^(-13) J` |
|
| 19. |
To explain the nature of radiation, James Clark Maxwell in 1864 put forward .Electromagnetic wave theory.. This theory could explain the phenomena of interference and diffraction but could not explain phenomena of black body radiation and photoelectric effect. Also, after the advent of electromagnetic wave theory, Rutherford.s model of atom suffered a serious drawback. To explain the phenomena of black body radiation and photoelectric effect, Max planck in 1900 put forward Planck.s quantum theory. BAsed on this theory, Neils Bohr in 1913 put forward Bohr.s model of atom which could overcome the drawback of Rutherford.s model and also explain the line spectral elements, especially the line spectra of hydrogen and hydrogen-like particles. Which of the following statements is incorrect about electromagnetic wave theory ? |
|
Answer» ENERGY is emiited continuously from the source |
|
| 20. |
What does the following conditions reval ? i= 1 , lt 1 and I gt 1. |
|
Answer» Solution :For a solute that does not DISSOCIATE or ASSOCIATE the Van't Hoff factor is EQUAL to 1(i=1) and the molar mass For the solutes that associates to form higher oligomers in solution the Van't Hoff factor will be less than one `(I lt 1) `and the observed molar mass will be GREATER than the actual molar mass For solutes that dissociates into their consituent ions the Van't Hoff factor will be more than one ` ( i gt 1) ` and the oberved molar mass will be less than the normal molar mass. |
|
| 21. |
When excess of eletroyte is added to a colloid it |
|
Answer» Coagulats |
|
| 22. |
Which components are present in stratosphere? |
| Answer» Solution :TROPOSPHERE is made of turbulent, dusty zone containing air, much WATER VAPOUR and clouds. | |
| 23. |
Which Boiling Point is greater ? Give example. |
|
Answer» Solution :For `H_(2)O` : `underset(100^(@)C(273 K))("NORMAL Boiling POINT")gt underset(99.6^(@)C(372.6K))("Standard Boiling Point")` Normal Boiling point = 1 atm pressure Standard Boiling point = 1 bar pressure 1 bar pressure `LT` 1 ATOM pressure. |
|
| 24. |
Which methods are used to make water germ free? |
|
Answer» PASSING `Cl_2` |
|
| 25. |
What is the ("Mol wt")/("Eq.wt") ratio of Fe_(2)(SO_(4))_(3) being converted into Fe(OH)_(3) |
|
Answer» No change in oxidation Number EQ. wt of `underset(("as SALT"))(Fe_(2)(SO_(4))_(3))=("MOL. wt. of "Fe_(2)(SO_(4))_(3))/(6)` |
|
| 26. |
When borax is heated in a Bunsen burner flame with CaO on a loop of platinum wire , ayellow coloured Co(BO_2)_2 bead is formed. |
| Answer» Solution :False STATEMENT :(BLUE COLOURED bead is FORMED ) | |
| 27. |
Which of the following conditions favours the existence of a substance in the solid state ? |
|
Answer» HIGH TEMPERATURE |
|
| 28. |
When 1 litre of IM HCl is mixed with 1 litre of 1M NaOH, the rise in temperature was found to be T_1. In another experiment 1 litre of 0.5 M NaOH is mixed with 1 lit. of 0.5 M HCl. The rise in temperature was found to be T_2. Then |
|
Answer» `T_1 GT T_2` |
|
| 29. |
Two what type of system the following belong ? (i) Tree (ii) Pond(iii) Animals (iv) Tea placed in a kettle(v) Tea placed in thermos flask(vi) Tea placed in a cup. |
| Answer» Solution :(i) Open SYSTEM (II) Open system (iii) Open system (IV) CLOSED system (v) isolated system (VI) open system. | |
| 30. |
The vander waals constants a and b of a real gas are 3.6L^2 atm mol^(-2) and 0.05L mol^(-1)respectively. If 200g of gas (molecular mass 40) is placed in 10L vessel at 300K, {:("Column-I ","Column-II "),("A) Pressure correction (atm) ","P) 0.25 "),("B) Free space for the molecules to move about(L) ","Q) 0.0125 "),("C) Actual volume of the gas molecules per mole (L) ","R) 0.9 "),("D) Effective volume occupied by total gas molecules (L) ","S) 9.75 "):} |
|
Answer» `= (an^2)/(V^2) = (3.6 XX (200/40)^2)/((10)^2) = 0.9`. b) Free space = `V_i = V = nb = 10 - 10 - 0.25 = 9.75` c) Actual volume of gas MOLECULES = `5 xx V_m` `b = 4 V_m implies V_m = b//4 = (0.05)/(4) = 0.0125//"mole"` d) effective volume = `nb = 5 xx 0.05 = 0.25`. |
|
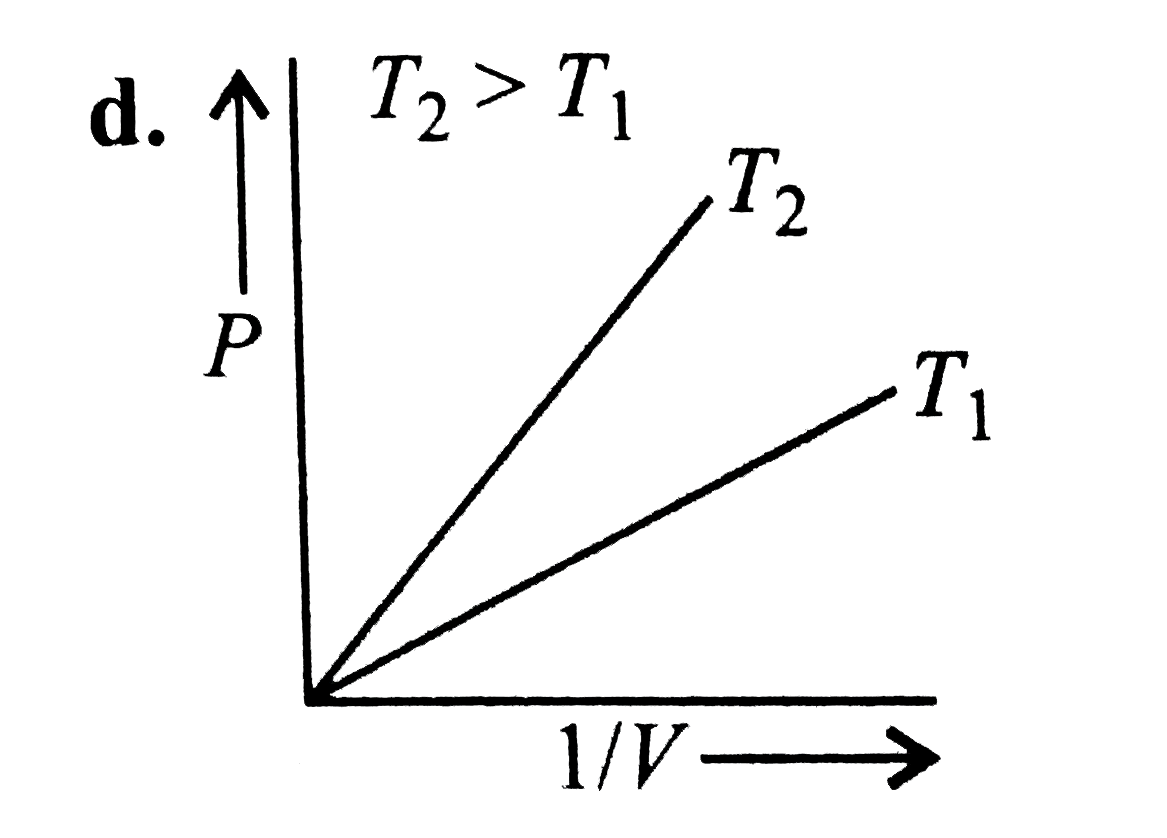
| 31. |
Which of the following graphs is consistent with ideal gas behaviour? |
|
Answer»
|
|
| 33. |
Which of the following conditions will favour maximum formation of the product in the reaction A_(2) (g) + B_(2) (g) hArr X_(2) (g), Delta _(r) H=-X kJ |
|
Answer» LOW temperature and high pressure |
|
| 34. |
What starting material gives 5- and 6- membered rings through aldol cyclizations? |
|
Answer» ALPHA, beta- unsaturated aldehydes |
|
| 35. |
Wrute following reaction : (i) Ethyne + (Cl_(2) " or "Br_(2) + H_(2)O) rarr (ii) Hydration reaction of 1-butyne. (iii) Reaction of acetenelene with HCN. (iv) Hydrogenation of ethyne in presence of Lyndlar catalyst (Pd, BaSO_(4)). (v) Complete combustion of ethyne in presence of air. (vi) Reaction of propyne with alkaline KMnO_(4). (vii) Ozononolysis of ethyne. |
Answer» Solution : (iii) `underset("Ethyne")(HC-=CH+HCN) OVERSET(Ba(CN)_(2))rarr underset("(Vinyl Cynide)")underset("Acylonitrile")(CH_(2)=CHCN)` (IV) `underset("Ethyne")(HC-=CH)+H_(2) overset(Pd, BaSO_(4))rarr underset("Ethene")(CH_(2)=CH_(2))` (v) `CH-=CH+(5)/(2)O_(2) rarr 2CO_(2) + H_(2)O + 1300 kJ` (vi) 
|
|
| 36. |
What kind of side effects observed, if among of fluoride in drinking water is more than 10 ppm. |
| Answer» Solution : If the concentration of `F^-` is more than 10 PPM, it is hazardous to BONES and TEETH. | |
| 37. |
Thestep in which CI-CI bond homolysisoccurs is called …………… |
| Answer» SOLUTION :initiationstep | |
| 38. |
Write two reactions to explain amphoteric nature of water. |
|
Answer» SOLUTION :Water is amphoteric in nature . It acts as an ACID in presence of strong base and as a base in presence of strong acids. `underset("Base"_1)(H_2O_((l))) + underset("Acid"_2)(H_2S_((AQ)))to underset("Acid"_1)(H_3O_((aq))^+) + underset("Base"_2)HS_((aq))^(-)` `underset("Aacid"_1)(H_2O_((l)))+underset("Base"_2)(NH_(3(aq))) tounderset("Aacid"_2)(NH_(4(aq))^(+))+ underset("Base"_1)(OH_((aq))^(-))` Due to amphoteric character , water undergoes self IONISATION as following `underset("Acid"_1)(H_2O_((l)))+underset("Base"_2)(H_2O_((aq))) hArr underset"(Conjugate acid)" underset("Acid"_2)(H_3O_((aq))^(+))+ underset"(Conjugate acid)"underset("Base"_1)(OH_((aq))^(-))` |
|
| 39. |
Which of the following statements are correct? A) CH_2 = CH - CH = CH_2 underset(Br_2)overset(-40^(@)C)to B) CH_2 = CH - CH = CH_2 underset(Br_2)overset(-40^(@)C)to |
|
Answer» In the MAJOR PRODUCT is 3,4-dibromobut-1-ENE and minor product is 1,4-dibromobut-2-ene |
|
| 40. |
Which of the following is redox reaction? |
|
Answer» 1-Butyne `underset(BaSO_(4))overset(Pd//C-H_(2))to` |
|
| 41. |
Whatis interference ? |
| Answer» SOLUTION :The COMBINATION of twoor moreelectromagneticwaveformsto FORMA resultan WAVE inwhichduedisplacementis eitherreinforcedor CANCELLED. | |
| 42. |
White fumes appear around the bottle of anhyd. AICI_3 due to the formation of |
| Answer» ANSWER :A | |
| 44. |
Which of the following statements indicates that law of multiple proportionsis being followed . |
|
Answer» Sample of carbon dioxide taken from any source will always have carbon and oxygen in the RATIO `1:2`. Thus, the masses of oxygen combine with a fixed mass of carbon in CO and `CO_(2)` are 16 and 32 respectively these masses of oxygen bear simple ratio of `16:32`or `1:2` to each other. This is an example of law of MULTIPLE proportion. |
|
| 45. |
Which is used as autocatalyst:- |
|
Answer» `Al_(2)O_(3)` `5H_(2)C_(2)O_(4)+2KMnO_(4)+3H_(2)SO_(4)to2MnSO_(4)+K_(2)SO_(4)+10CO_(2)+8H_(2)O` |
|
| 46. |
Which of the following is incorrect regarding atomic radii ? |
|
Answer» `Na^(+)gt AL^(3+)lt Mg^(2+)` |
|
| 47. |
The wrong statement in the following is |
|
Answer» Sulphonation of benzene takes place only with HOT concentrated sulphuric acid |
|
| 48. |
Why Andrews Graph required ? |
|
Answer» Solution :It is ISOTHERMAL curve of, Pressure `to` VOLUME of fixed temperature of GAS. It is USEFUL to derive liquification of gas in proper situation. |
|
| 49. |
Which one of the following pairs of structures represent the phenomenon of resonance? |
|
Answer» `H_(2)C=CH-OVERSET(overset(O)(||))(C)-H, Coverset(+)(H_(2))-CH=overset(overset(O^(-))(|))(C)-H` B) `CH_2 = CH - overset(+)(CH)Cl , overset(+)(CH_2) - CH = CH - Cl` C) `(CH_3)_2 CH - overset(O)overset(||)C-O^(-) , (CH_3)_2CH- overset(O^(-))overset(|)C=O` |
|
| 50. |
Which of the following reactions of methane incomplete combustion : |
|
Answer» `2CH_4+O_2 overset"Cu/523 K / 100 atm"to 2CH_3OH` |
|