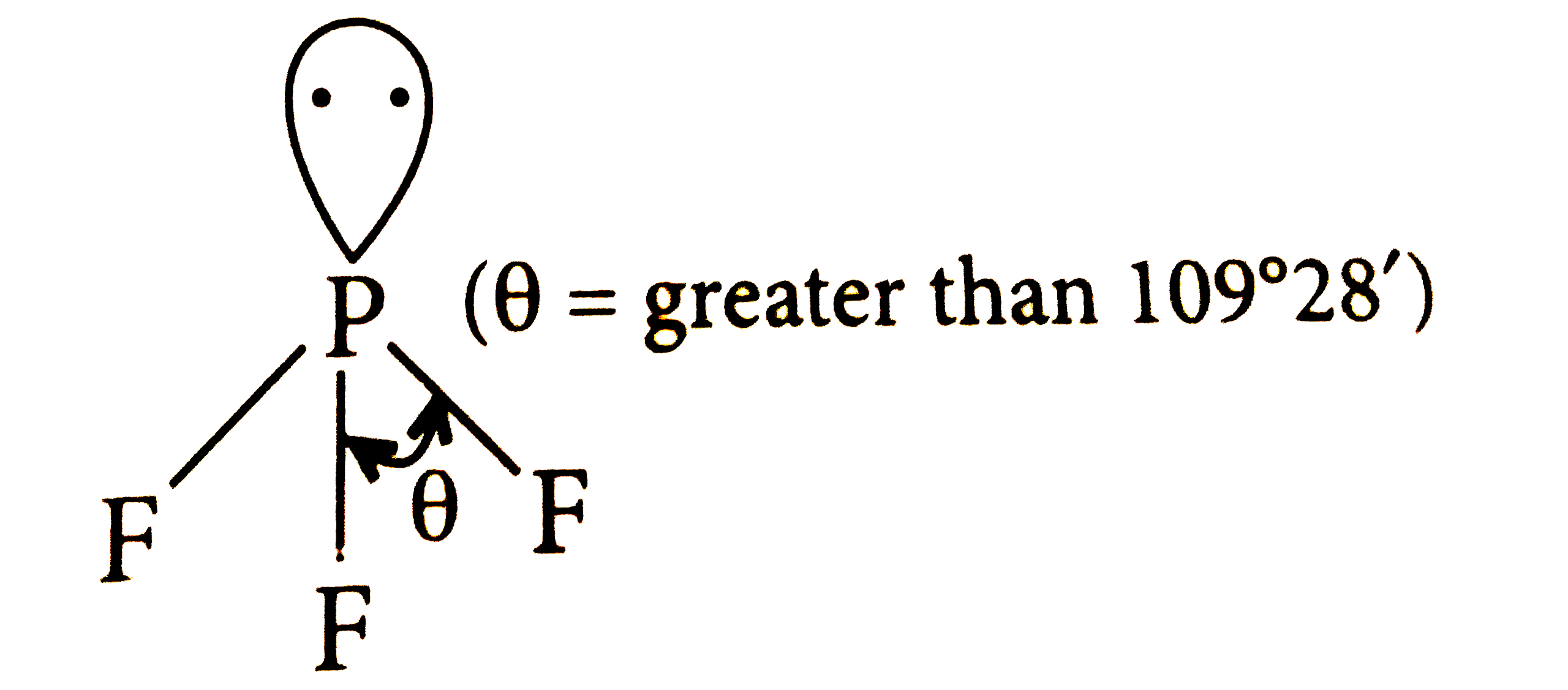Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What are the causes for deviation of real gases from ideal behaviour. |
| Answer» Solution :The CAUSES are : (a) The VOLUME occupied by molecules is negligle (b) the INTERMOLECULAR forces of ATTRACTION are NEGLIGIBLE. | |
| 2. |
Using the equation of state pV = nRT , show that at a given temperature density of a gas is proportional to gas pressure p. |
|
Answer» Solution :IDEAL GAS equations, pV = nRT `therefore p=((n)/(V))RT ""` …..(a) but, mole of gas (n) `= (m)/(M)` where, m = mass of gas is constant M = molecular mass of gas is ALSO constant `therefore p=(mRT)/(MV) ""` ……(b) but, `(m)/(V)=("mass of gas")/("volume of gas")=` density of gas (d) Which is put in eq. (b) According to this, `p=(dRT)/(M)` Thus, `p prop d ""` .....(at constant T and V) ..At constant temperature and volume, PRESSURE of fixed AMOUNT of gas is directly proportional to its density... |
|
| 3. |
Which of the following relates to photon both as wave motion and as a stream of particles ? |
|
Answer» `E=mc^(2)` |
|
| 4. |
Write the IUPAC name of CH_(3)-underset(Cl)underset(|)(CH)-CH_(2)-CH=CH_(2) |
|
Answer» 2-chloro-pent-4-ene |
|
| 5. |
The total number of orbitals associated with the principle quantum number n=3 is |
|
Answer» 9 |
|
| 6. |
Which one of the following is most reactive towards nucleophilic substitution reaction ? |
|
Answer»

|
|
| 7. |
Which of the following contains the greatest number of oxygen atoms? |
|
Answer» 1 G of O |
|
| 8. |
Which of the following reagents cannot be used for converting ? |
|
Answer» `CH_(3)COOH//H_(2)SO_(4)` |
|
| 9. |
Two solid A "and" B are present in two different container having same volume and same temperature following equilibrium are established: In container (1)A(s)hArrD(g)+C(g)P_(T)=40 "atm at equilibrium" In container (2)B(s)hArrE(g)+F(g)P_(T)=60 "atm at equilibrium" If excess of A "and" B are added to a third container having double the volume and at same temperature then, the total pressure of this container at equilibrium is: |
| Answer» Solution :N//A | |
| 10. |
Which of the followingsets of quantumnumbersrepresentsthe highestenergyof an atom ? |
|
Answer» n=3 ,l= 0 m=0 ,s=`(1)/(2)` |
|
| 11. |
Write the Lewis dot structure of CO molecule. |
Answer» SOLUTION :
|
|
| 12. |
Which of the following statements about amorphous solid is incorrect ? |
|
Answer» They MELT over a range of TEMPERATURE |
|
| 13. |
Which of the following is used in cakes or pastries to making them light and fulffy ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 14. |
What is the value ofDeltaH in the following reaction at 300 K ? CH_(4)(g) 2O_(2)(g) rarr CO_(2)(g)+2H_(2)O(l), DeltaU = 885389J //mol. |
| Answer» SOLUTION :`DELTAH = DeltaU + Deltan_(g)RT = 885389+ ( -2) XX 8.314 xx 300 = - 880400 J mol^(-1)` | |
| 15. |
When is a cation highly polarising ? Which alkali metal cation has the highest polarising power ? |
| Answer» Solution :A cation is HIGHLY POLARISING if its charge/SIZE ratio is very high. Because of its small size ,`Li^(+)` ion has the highest polarising power AMONG the ALKALI metal ions. | |
| 17. |
What is the maximum nad minimum oxidation states for Na Mg AI Sn and Mn? |
|
Answer» Solution :The minimum oxidation state for all metals is 0 the MAXIMUM oxidatin state for s and p block elements is EQUAL to the numbre of ELECTRONS present inn the valence shell thus maximum oxidation stateof Na=+1 that of MG is +2 that of AI is +3 and that of k `ll` Sn is +4 However for d block elemtys the maximum oxidation state is equal to hte sum of (n-1) d and ns electons thus maximum oxidation state of Mn`(3d^(5) 4S^(2))=5+2=+7` |
|
| 18. |
What is the volume (in lit) of carbon dioxide liberated at STP, when 2.12 grams of sodium carbonate (mol. wt = 106) is treated with excess dilute HCl? |
|
Answer» `2.28` |
|
| 19. |
What type of structure is possessed byH_(2)O_(2) molecule? Draw it and label the varous bond angles and bond length . Comment on the dipole moment of H_(2) O_(2)molecule . |
|
Answer» Solution : As ` H_(2) O_(2)` is not a linearbut has a bent STRUCTURE as SHOWN in , therefore , bond MOMENTS do not cancel out and as a result , it has a net dipole MOMENT of 2.1 D |
|
| 20. |
Which of the following statement is/are incorrect about of 'Y' and 'Z'? |
|
Answer» The pink colour is due to adsorption of `KMnO_(4)` on `BaSO_(4)` surface |
|
| 21. |
Which of the following statement (s) is/are correct? Statement (i) : The entropy of isolated system with P-V work only is always maximized at euilibrium. Stetement (ii) : It is possible for the entropy of close system to decreases substantilly in an irreversible process. Statement (iii) : Entropy can be created but not be destroyed. Statement (iv) : DeltaS system is zero for reversible process in an isolated system. |
|
Answer» Statements `I,II,iii` , |
|
| 22. |
Which of the following colour changes shown during redox titrations is not correct ? |
|
Answer» `Cr_(2)O_(7)^(2-)` oxidises the indicator diphenylamine to produce blue colour showing end point. |
|
| 23. |
Which pair of species are formed when hydrogen peroxide is react with suspension of PbS ? |
|
Answer» `PBO+ SO_2 + H_2O` |
|
| 24. |
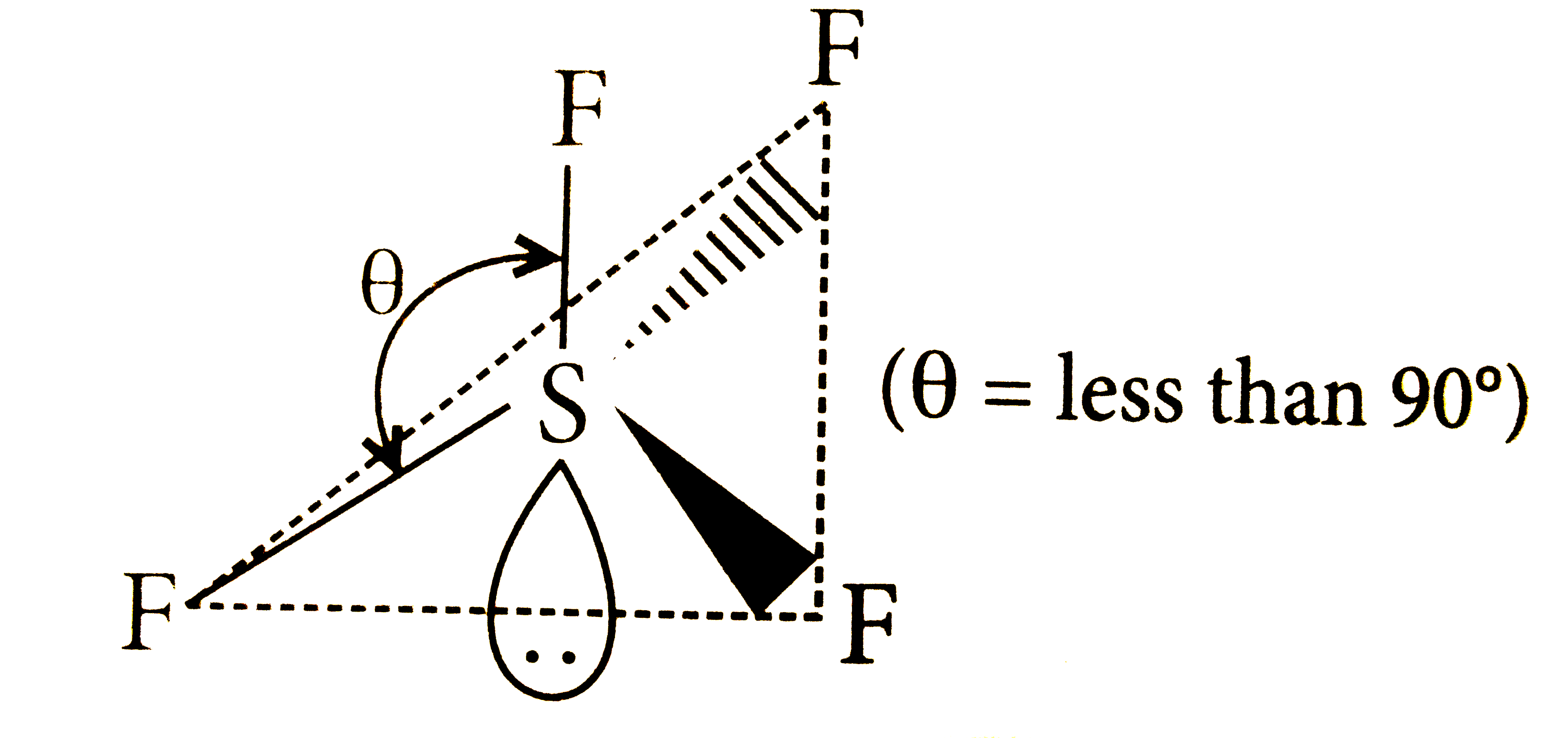
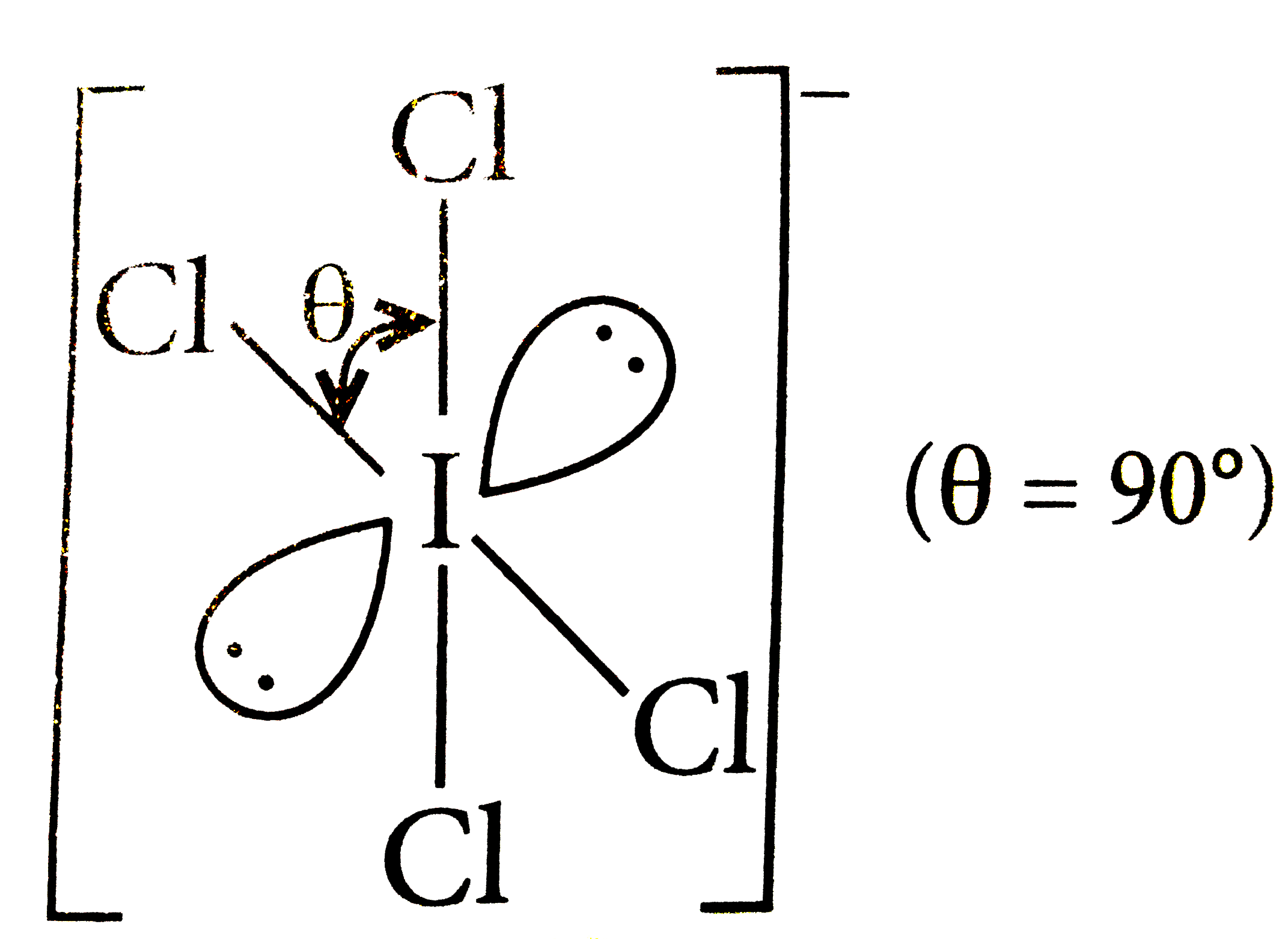
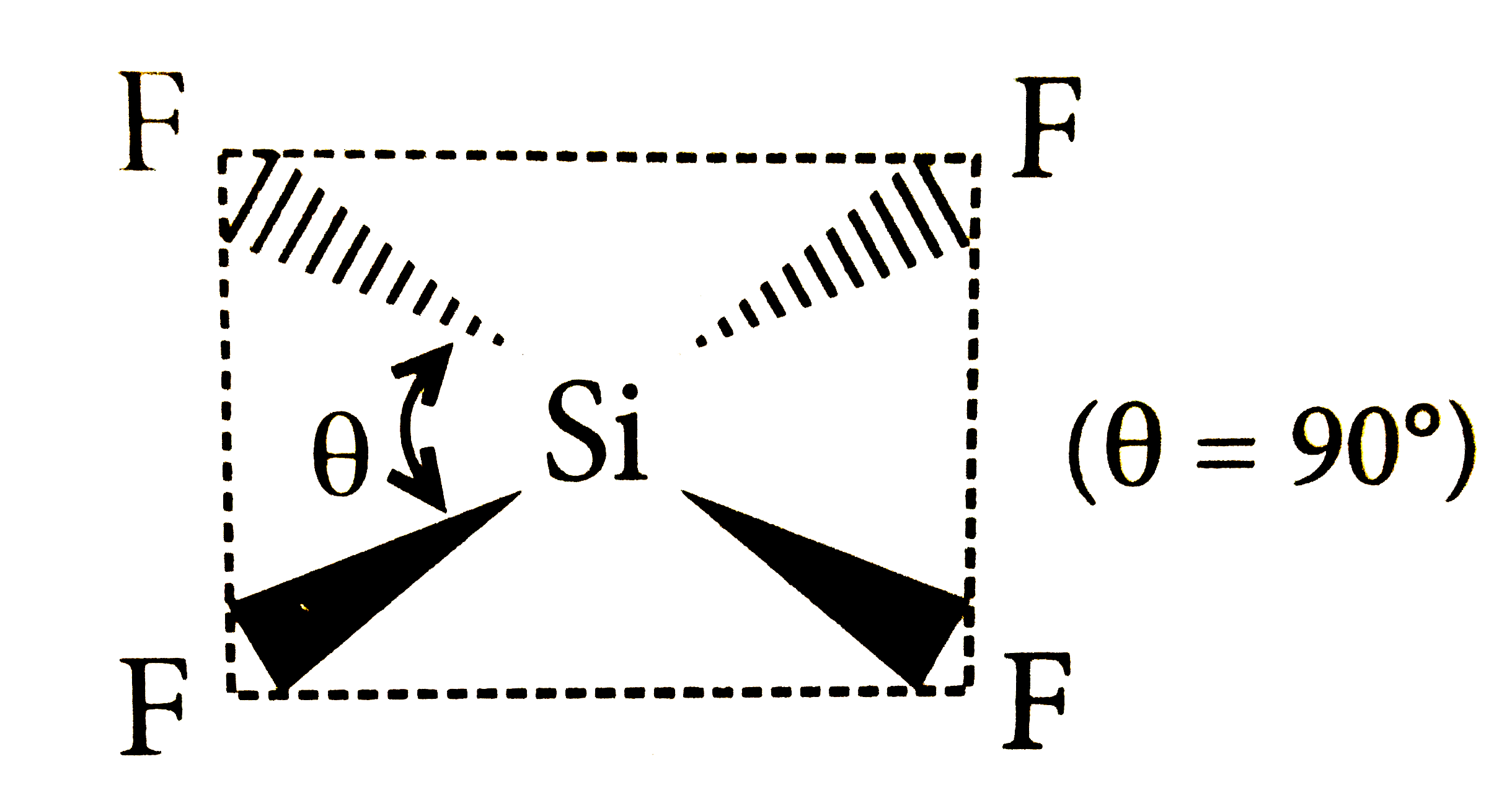
Which of the following structure is correctly drawn according to fundamental idea of VSEPR theory? |
|
Answer»
|
|
| 25. |
The wave length having wave number 4000 cm^(-1) is |
|
Answer» `2.5 mu m` |
|
| 26. |
What is the prescribed standard of desirable limit of TDS (Total Dissolved Solids) of drinking water according to BIS? |
| Answer» SOLUTION :500 ppm | |
| 27. |
Which of the following reactions does not involve the change in oxidation state of metal? |
|
Answer» `VO^(-2) rarr V_(2)O_3` |
|
| 28. |
Writethe value of K_p and K_C equation for CaCO_(3) (s) hArr CaO(s) + CO_(2)(g) |
| Answer» Solution : A pure solid ALWAYS has the same CONCENTRATION at a given temperature, as it does not expand to fill its CONTAINER i.e., it has the same no. of moles of its VOLUME. Therefore the concentration of pure solid is a constant. So the EXPRESSION if `K_(c) "and " is K_(p) is K_(c) = [CO_2].K_p = P_(CO_3)` | |
| 29. |
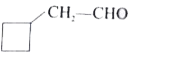
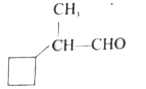
Which one of the following is called 2-cyclobutylpropanal? |
|
Answer»
|
|
| 30. |
Which chemical is used to remove fluoride from water? |
|
Answer» `MG Cl_2` |
|
| 31. |
What is the role of Fe(OH)_3 in the contact process ? |
|
Answer» To REMOVE COLLOIDAL IMPURITIES |
|
| 32. |
What is the maximum numaber of emission lines obtained when the excited electron of a H atom in n=6 drops to the ground state ? |
|
Answer» Solution :No. of LINES produced when electron from nth shell drops to ground state `Sigma (n_(2)-n_(1))=Sigma (6-1)= = Sigma 5=5+4+3+2+1=15` There are produced DUE to FOLLOWING transitions : `{:(6 rarr 5,5 rarr 4,4 rarr 3,3 rarr 2,2 rarr 1),(6 rarr 4,5 rarr 3,4 rarr 2,3rarr1,),(6 rarr 3,5 rarr 2,4 rarr 1,,),(6 rarr 2,5 rarr 1,,,),(6 rarr 1,,,,),(("5 lines"),("4 lines"),("3 lines"),("2 lines"),"(1 lines)"):}` |
|
| 33. |
When methane is burnt in oxygen to produce CO_(2) and H_(2)OFind the Change in oxidation number ? |
|
Answer» Oxidation no. CHANGE `=+4 -(-4) =+8` |
|
| 34. |
What is the IUPAC name of |
|
Answer» 3-Ethyl-5-methyl-1-n-propylbenzene |
|
| 35. |
The wave function Phi_(n,l,m_(1)) is a methematical function whose value depends upon shperical upon spherical polar coordinates (r,theta,phi) of the electron and characterized by the quantum numbers, n,l and m_(1).Here r is distance from nucleus, theta is colatitude and phi is azimuth. in the mathematical functions given in the table, Z is atomic number, a_(0) is Bohr radius. . Q. For hydrogen atom the only CORRECT combination is : |
|
Answer» (I) (iv) (R) `E=-(Z^(2))/(n^(2))xx13.6eV` `DeltaE_(2-4)=(1)/(16)xx13.6-(-(1)/(4)xx13.6)` `=(3)/(16)xx13.6eV` `DeltaE_(2-6)=-(1)/(36)xx13.6-(-(1)/(4)xx13.6)` `=(8)/(36)xx13.6eV` `(DeltaE_(2-4))/(DeltaE_(2-6))=(3//16xx13.6eV)/(3//36xx13.6eV)` `=(3xx36)/(16xx8)=(27)/(32)` `thereforeDeltaE_(2-4)=(27)/(32)DeltaE_(2-6)` |
|
| 36. |
The wave function Phi_(n,l,m_(1)) is a methematical function whose value depends upon shperical upon spherical polar coordinates (r,theta,phi) of the electron and characterized by the quantum numbers, n,l and m_(1).Here r is distance from nucleus, theta is colatitude and phi is azimuth. in the mathematical functions given in the table, Z is atomic number, a_(0) is Bohr radius. . Q. For the given orbital in column 1, the only correct combination forany hydrogen-like species is: |
|
Answer» (IV) (iv) (R) 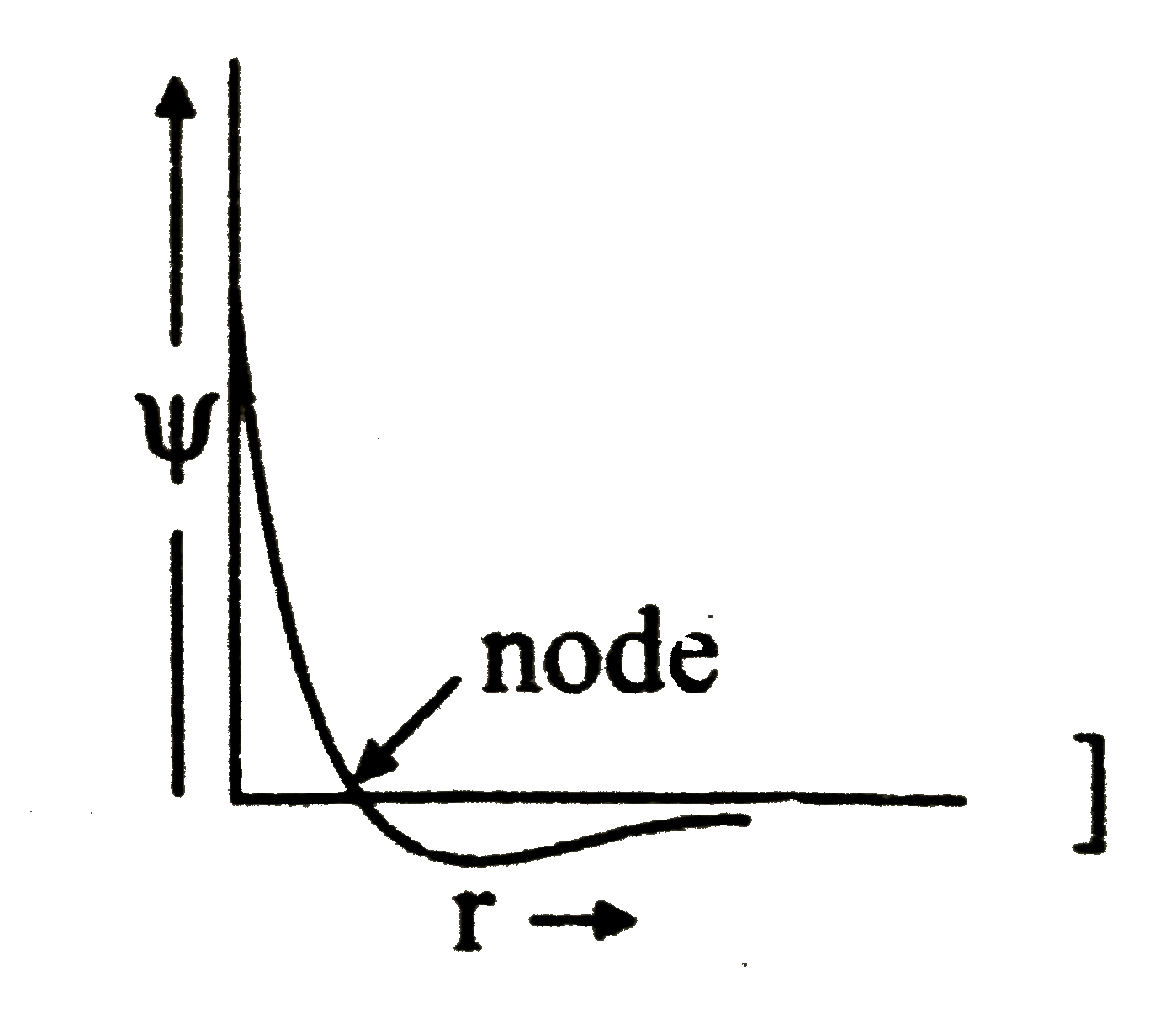 Number of RADIAL NODE in `2s=n-l-1` `=2-0-1=1` |
|
| 37. |
The wave function Phi_(n,l,m_(1)) is a methematical function whose value depends upon shperical upon spherical polar coordinates (r,theta,phi) of the electron and characterized by the quantum numbers, n,l and m_(1).Here r is distance from nucleus, theta is colatitude and phi is azimuth. in the mathematical functions given in the table, Z is atomic number, a_(0) is Bohr radius. . Q. For He^(+) ion, the only INCORRECT combination is: |
|
Answer» <P>(II) (ii) (Q) |
|
| 38. |
Who is the first member of alkene series ? Give information about its bond and structure. |
|
Answer» Solution :General formula of alkene is `C_(n)H_(2n)`. If n = 1 `1^(st)` member is methane. Age of methene is too LESS. THEREFORE, methane is not TAKEN as first member of alkene. `1^(st)` member of alkene is `C_(2)H_(4)` bond structure of ETHENE. |
|
| 39. |
Which type of hybridization is called (a) tetrahedral (b) trigonal ? |
| Answer» SOLUTION :`SP^(3)`(B) `sp^(2)` | |
| 40. |
Which of the following statements is correct? Dielectric constant of H_(2)O_(2)? |
|
Answer» Zeolites have a more closed struture than feldspar. |
|
| 41. |
The strength of sigma bond formed by s-s, s-p & p-p overlaps are in the order |
|
Answer» `s-s GT s-p gt p-p` |
|
| 43. |
Which of the following possess zero dipole moment |
|
Answer» trans-2,3-dichloro-2-butene |
|
| 44. |
Which of thefollowingatomsshould have thehighest1 st electron affinity ? |
|
Answer» F |
|
| 45. |
What type of interations hold the molecules together in a polar molecular solid ? |
| Answer» SOLUTION :DIPOLE - dipole ATTRACTIONS. | |
| 46. |
Which of the following oxide react with quick lime and produce Ca(PO_(4))_(2) ? |
| Answer» Solution :`6CaO+P_(4)O_(10) to 2Ca_(3)(PO_(4))_(2)` | |
| 47. |
Which one of the following is used to test primary amines? |
|
Answer» Schiff's TEST |
|
| 48. |
whichof thefollowing reactionsindicate arerrangementreactioninvolvingmodificationof functionalgroup ? |
|
Answer» `CH_(3)- OVERSET(CH_(3))overset(|)CH-CH_(2) Ohoverset(H^(+))hArrCH_(3) - underset(CH_3)underset(|)overset(CH_3)overset(|)C-OH` |
|
| 49. |
Write the equation to calculate the energy of n^(th) orbit. |
|
Answer» Solution :`E_n=((-1312.8)Z^(2))/(N^(2))kJmol^(-1)` Where Z=atomic NUMBER,n=principle quantum number. |
|
| 50. |
Which effect is observed on velocity distribution if temperature increases. |
| Answer» Solution :Due to TEMPERATURE INCREASES then GRAPH BECOMES wide and flat and most probable speed increases and number of MOLECULES increases. | |