Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is used for the destruction of colloids |
|
Answer» Dialysis |
|
| 2. |
Which of thefollowingprocessesdo notinvolveabsorption ofenergy ? |
|
Answer» `S ( G) + e^(-) to S^(-)(g)` |
|
| 3. |
Which one is non-viable particulate pollutant? |
| Answer» SOLUTION :Fumes | |
| 4. |
Which metal in powdered from suspended in oil is used paint for mirrors? |
|
Answer» Ag |
|
| 5. |
Whichof the following does not contain the peroxide ion ? |
|
Answer» `K_(2)O ` |
|
| 6. |
Violet coloured complex obtained in the detection of sulphur is: |
|
Answer» `Na_(2)[Fe(NO)(CN)_(5)]` |
|
| 7. |
What is the above CN^(-) and N_(2)? |
|
Answer» Both are isoelectronic |
|
| 8. |
Which of the following substituents has +M (Mesomeric) effect? |
| Answer» Answer :C | |
| 9. |
Which of the following conditions is/are favourable for the feasibility of a reaction? |
|
Answer» `DELTA H= - ve, T Delta S= +ve` `Delta H lt 0, T Delta S lt 0` `rArr Delta H - T Delta S lt 0` if `T Delta S lt Delta H` `Delta H gt 0, T Delta S gt 0` `rArr Delta H - T Delta S lt 0` if `T Delta S gt Delta H` |
|
| 10. |
Which of the following types of carbon atoms possess highest electronegativity |
|
Answer» `SP^(3)` |
|
| 11. |
Which hybridization occurs in the square planar, octahedral and linear shape ? |
| Answer» Solution :Respectively `DSP^(2), SP^(3) d^(2) or d^(2) sp^(3) and `sp | |
| 12. |
Write the resonance structure of CO_(2). |
| Answer» SOLUTION :`:OVERSET(..)O=C=overset(..)O:HARR:overset(o+)O-=C-UNDERSET(..)overset(..)O:^(ө)harr""^(ө):underset(..)overset(..)O-C-=-overset(+)O:` | |
| 13. |
Which of the following when used as a fuel have the maximum tendency to resist knocking ? |
|
Answer» STRAIGHT CHAIN alkanes |
|
| 14. |
Weight of one C_(60)H_(122) molecule is |
|
Answer» `3.72 XX 10^(23)G` |
|
| 15. |
Which of the following is not a nucleophile ? |
|
Answer» `OH^(-)` |
|
| 16. |
The schrodinger wave equation for hydrogen atom is Psi_(2s)=1/(4sqrt2pi)(1/a_0)^(1//2) [2-r_0/a_0]e^(-2//a_0) where a_0 is Bohr radius. If the radial node in 2s be at r_0, then find r in terms of a_0 |
|
Answer» `r_0=2a_0` `Psi_(2x)^2 =0` at NODE then , `[(2-r_0)/a_0] =0 RARR r_0 =2a_0` |
|
| 17. |
Which one of the following is the correct statmenet |
|
Answer» BORIC acid is a protonic acid 
|
|
| 18. |
Which will be the least stable resonating structure : |
|
Answer» `CH_(2)=CH-OVERSET(o+)CH-overset(Theta)CH-O-CH_(3)` |
|
| 19. |
Whenthe energyof electrontakenzero? |
| Answer» SOLUTION :When theelectronis freefrom the EFFECT of nucleuss TENN= `PROP`atthentimeenergy. | |
| 20. |
Two flasks A and B have equal volumes. A is maintained at 300 K and B at 600 K. while A contains H_(2)gas, B has an equal mass of CH_(4)gas. Assuming ideal behaviour for the both gases find the ratio of (u_(av))_(A): (u_(av))_(B). |
|
Answer» |
|
| 21. |
The units which are obtained from basic units are called .......... |
|
Answer» DERIVED units |
|
| 22. |
Which of the following tests could be performed to distinguish between 1-butyne and 2-butyne. ? |
|
Answer» `Ag^(+)//NH_(3)` <BR>`Br_(2)//C Cl_(4)` |
|
| 23. |
Which one of the following is an example for Clathrate hydrate? |
|
Answer» `CuSO_(4).5H_(2)O` |
|
| 24. |
X is heated with soda lime and gives ethane. X is |
|
Answer» ethanoic acid |
|
| 25. |
Weight percentage of gypsum in cement is approximately |
|
Answer» 2 |
|
| 26. |
What are the similar properties in Beryllium and Aluminium? |
| Answer» SOLUTION :The similar PROPERTIES in Be and AI are: (a) Both Be and AI form covalent compounds EX: `BeCI_(2)` and `AICI_(3)` (b) Both Be and AI have same electronegativity (i.e., Be = 1.5, AI = 1.5 (c ) Both `BeCI_(2)` and `AICI_(3)` used as catalyst in Fridel - Craft.s reaction because both act as LEWIS acid. (d) Both Be and AI form an oxide coating . Hence, they donot react with water. (e ) Both `Be_(2)C` and `AI_(4)C_(3)` react with water to liberate methane gas. | |
| 27. |
What is the formula of the binary compound of boron and sulphur? |
| Answer» Solution :Boron on strong HEATING with sulphur at `1200^@C` GIVES boron SULPHIDE `B_2S_3` | |
| 28. |
The thermite reaction used ofr welding of metals involves the reaction 2Al(s) + Fe_(2)O_(3)(s) rarr Al_(2)O_(3)(s) + 2Fe(s) What is Delta H^(@)at 25^(@)C for this reaction ? Given that the standard heats of formation of Al_(2)O_(3) and Fe_(2)O_(3) are - 1675.7 kJ and - 828.4 kJ mol^(-1) respectively. |
|
Answer» Solution :We are given(i) `2Al(s) + (3)/(2) O_(2)(g) rarr Al_(2)O_(3)(s) , Delta _(r) H^(@) = - 1675.7 K J mol^(-1)` (II) `2Fe(s) + (3)/(2) O_(2)(g) rarr Fe_(2)O_(3)(s) , Delta _(r) H^(@) = - 828 .4kJ mol^(-1)` We aim at `2Al(s) + Fe_(2)O_(3) (s), rarr Al_(2)O_(3)(s) + 2Fe(s), Delta_(r) H^(@) = ?` Equation (i) - Equation (ii) gives `2Al(s) + Fe_(2)O_(3)(s) rarr Al_(2)O_(3)(s)+2Fe(s), Delta_(r)H= - 1675.7 - ( 828.4)` `= - 847.3 kJ mol^(-1)` ALTERNATIVE Method We aim at`2Al(s) + Fe_(2)O_(3)(s) rarr Al_(2)O_(3) +2Fe(s)lt Delta_(r) H^(@) = ?` `Delta _(r) H = ` Sum of `Delta_(r) H^(@)` of products `-` Sum of `Delta_(r) H^(@) =?` `= [ Delta_(F) H^(@) (Al_(2)O_(3))+ 2 xx Delta_(f ) H^(@) (Fe)] - [ 2 xx Delta _(f)H^(@) (Al)+Delta_(f) H^(@) ( Fe_(2)O_(3))]` `= [ 1675.7 +0] - [ 0+ ( -828.4)]` `= -847.3 kJ mol^(-1)` |
|
| 29. |
Which fo the following is the most stabl compound? |
|
Answer» `Ph_(3) OVERSET(+)(C )` |
|
| 30. |
Which out of the following will give basic solution ? |
|
Answer» AMMONIUM acetate |
|
| 31. |
Which of the following elements has the highest ionisation energy |
|
Answer» `[Ne] 3s^(2) 3p^(1)` |
|
| 32. |
Which of the following gas(es) always show positive devlation from ideal behavlour? |
|
Answer» `CH_(4)` |
|
| 33. |
Xe reacts with F_(2) at different ratio to give different types of xenon fluorides. Xe+ Fe rarr XeF_(2) (2:1) Xe +F_(2) rarr XeF_(4) (1:5) Xe + F_(2) rarr XeF_(6) (1: 20)The sbape and hybridisation of XeF_(4 ) is |
|
Answer» tetrahedral and`sp^(3)` |
|
| 34. |
Xe reacts with F_(2) at different ratio to give different types of xenon fluorides. Xe+ Fe rarr XeF_(2) (2:1) Xe +F_(2) rarr XeF_(4) (1:5) Xe + F_(2) rarr XeF_(6) (1: 20) Which of the following option is correct regrading XeF_(2) |
|
Answer» two fluorine OCCUPY equatorial position |
|
| 35. |
What is meant by the conjugate acid-base pair ? Find the conjugate acid/base for the following species: HNO_2 , CN^(-) , HClO_4 , F^(-), OH^(-) , CO_3^(2-) and S^(2-) |
Answer» Solution :Example : The ionic equilibrium is as under by ionization of HCl in WATER.  In this reaction `Cl^-` is a conjugate base of HCl and HCl is a conjugate ACID of `Cl^-`. HCl and `Cl^-` is a conjugate acid/base pair. Defination : The acid-base pair that DIFFERS only by one proton is called conjugate acid-base pair. Conjugate acid: `CN^(-)` of HCN, `F^-` of HF, `OH^-` of `H_2O, CO_3^(2-)` of `HCO_3^(-)`and `S^(2-)` of `HS^(-)` . Conjugate acid : `HNO_2` of `NO_2^-` and `HClO_4` of `ClO_4^-` |
|
| 36. |
Thermodynamics does not deal with |
|
Answer» the FEASIBILITY of a chemical reaction |
|
| 37. |
What is mole ? |
|
Answer»
|
|
| 38. |
The standard free energy change for a reaction is -212.3 kJ mol^(-1). If the enthalpy change of the reaction is -216 kJ mol^(-1) , calculate the entropy change for the reaction. |
|
Answer» |
|
| 39. |
Which one of the following undergoes nitration most readily ? |
|
Answer» Acetophenone |
|
| 40. |
White fumesappear around the bottle of anhydrousaluminium chloride. Give reason. |
|
Answer» SOLUTION :Anhydrous aluminiumchlorideis partially hydrolysedby ATMOSPHERIC moistureto liberateHCl gas. MoistHClappears white in colour. `AlCl_(3)(s) + 3H_(2)O(l)RARR Al(OH)_(3) + 3HCl(G)` |
|
| 41. |
Which of the following substance is used in the making of tools ? |
|
Answer» `Be_4C` |
|
| 42. |
Which one of the following is the strongest base in liquid phase? |
|
Answer» `(C_(2)H_(5))_(3)N` |
|
| 43. |
Which of the following alkali metal is having least melting point ? |
|
Answer» Na |
|
| 44. |
What is formed when oxalic acid is dehydrated by conc. H_(2)SO_(4) ? |
|
Answer» `C + CO_(2)` |
|
| 45. |
Which one of the following will decrease the pH of 50 mL of 0.01 M hydrochloricacid ? |
|
Answer» Addition of 50 ML of 0.01 M HCl (a) `N_(1)V_(1)+N_(2)V_(2)=N_(3)(V_(1)+V_(2))` `50xx0.01+50xx0.01=N_(3)(50+50)` or `N_(3)=(0.5+0.5)/(100)=10^(-2), pH=2` (No change) (b) `50xx0.01+50xx0.02=N_(3)(50+50)` or, `N_(3)=(0.5+0.1)/(100)=6xx10^(-3)` `pH=-log(6xx10^(-3))=2.22` (pH increases) (c) `50xx0.01+150xx0.002=N_(3)(50+150)` or, `N_(3)=(0.5+0.3)/(200)=4xx10^(-3)` `pH=-log(4xx10^(-3))=2.39` (pH increases) (d) `50xx0.01+5xx1=N_(3)(50+5)` or, `N_(3)=(0.5+5)/(55)=(55)/(5)=10^(-1)` pH = 1 (pH decreases) |
|
| 46. |
When 6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the residual solution is found to weigh 33 g. The number of moles of carbon dioxide released in the reaction is ………….. . |
|
Answer» 3 The amount of `CO_(2)` RELEASED, x = 3.3 G No. of MOLES of `CO_(2)` released = `3.3//44` = 0.075 mol |
|
| 47. |
Write the major product of the following reaction. |
Answer» SOLUTION :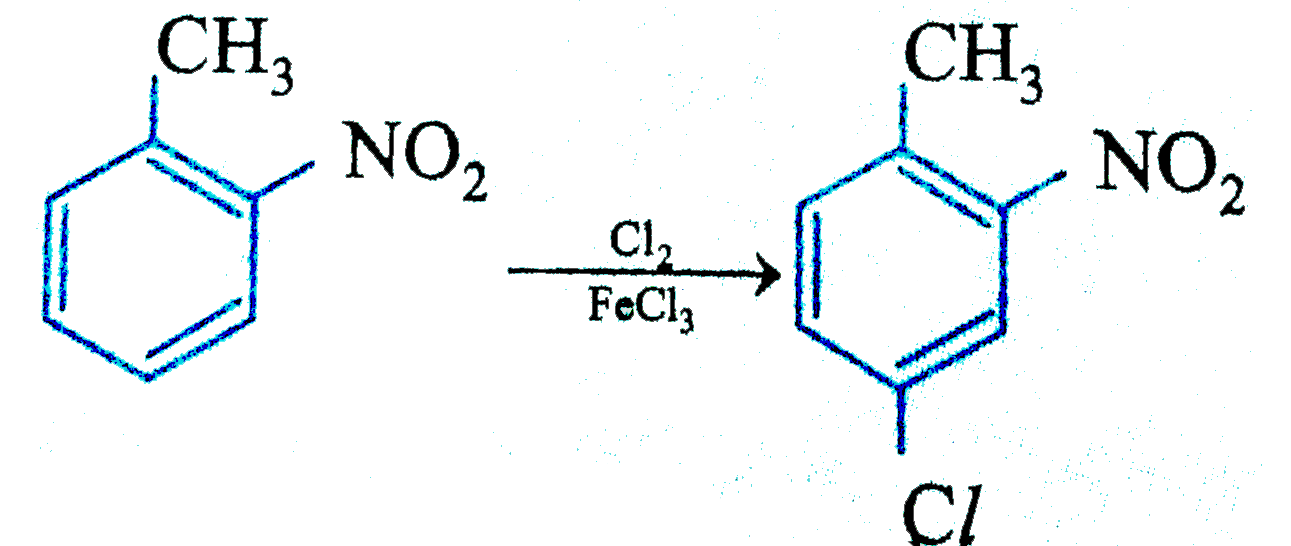 In 2-nitrotoulene, METHYL group and nitro group direct the electrophile into para POSITION of the methyl group which is META to nitro group. Hence the major product of the reaction is 4-chloro-1-methyl-2-nitrobenzene. |
|
| 48. |
The value ofDelta_(f)H^(c-) for NH_(3) is -91.8 kJ mol^(-1). Calculate enthalpy change for the following reaction : 2NH_(3)(g) rarr N(2)(g) + 3H_(2)(g) |
| Answer» Solution :GIVEN `:(1)/(2)N_(2)(G) +(3)/(2)H_(2)(g) rarr NH_(3)(g), Delta_(f)H^(@) = -91.8 kJ MOL^(-1)` | |
| 49. |
What is London forces? |
| Answer» Solution :It is a weak FORCE of attraction between non-polar MOLECULES like `O_(2)` or `N_(2)` (i.e. between like ATOMS). | |
| 50. |
When copper is added to a solution of silver nitrate, silver is precipitated. This is due to i) oxidation of silver ii) oxidation of copper iii) oxidation of silver iv) reduction of silver ion The correct combination is |
|
Answer» iii and IV are correct |
|