Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is hydration reaction ? |
| Answer» Solution :Dihydrogen gas adds to alkenes and alkynes in the presence of FINELY DIVIDED catalysts like platinum, palladium or nickel to FORM alkanes. This process is CALLED HYDROGENATION. | |
| 2. |
Which of the following compounds exists in only aqueous solution- |
|
Answer» `Li_3PO_4` |
|
| 3. |
Which of the following is not characteristic of chemisorption? |
|
Answer» Adsorption MAY be multimolecular LAYER |
|
| 4. |
The vapour density of a gas is 11.2. The volume occupied by 11.2 g of the gas is NTP is |
|
Answer» Solution :(b) Molecular weight of gas = 2`xxVD=2xx11.2=22.4` Hence, number of MOLES of gas taken=`(11.2)/(22.4)=(1)/(2)` Hence, volume of gas at NTP=`(1)/(2)xx22.4L=11.2 L` |
|
| 5. |
You are given two compound First, you name both of them the same yet, are they really same structure, or they some how different ?Carefully inspection should convince you that they are indeed different. Now, how their physical and chemical properties differ? Generally (i) cis-isomer has more diplomoment than trans-isomer (ii) cis-is less stable than trans (iii) boiling points of cis isomers are than those of trans isomers (remember boiling points are directly related to polarity which is determined by dipolemoment) chemical reactivities of cis and trans also differ from each other. Arrange the following compounds in the increasing order of the boiling poiints CH_(3)-underset(A)CH=CH-CH_(3)(cis),CH_(3)-underset(B)CH=CH-CH_(3)(trans) CH_(3)-underset(C)CHCl=CH(cis),CH_(3)-underset(D)CH=CHCl(trans) |
|
Answer» `BltAltCltD` |
|
| 6. |
You are given two compound First, you name both of them the same yet, are they really same structure, or they some how different ?Carefully inspection should convince you that they are indeed different. Now, how their physical and chemical properties differ? Generally (i) cis-isomer has more diplomoment than trans-isomer (ii) cis-is less stable than trans (iii) boiling points of cis isomers are than those of trans isomers (remember boiling points are directly related to polarity which is determined by dipolemoment) chemical reactivities of cis and trans also differ from each other. Which of the following compounds exhibit geometrical isommerism? |
| Answer» Answer :1 | |
| 8. |
Which of the following molarity of KMnO_4 solution can prouduce atleast 1 gm each of CO_2 and water when 2 litre of KMnO_4 reacts with excess of oxalic acid, according to the equation : 2KMnO_4 + 5H_2C_2O_4+3H_2SO_4 to 2MnSO_4 + K_2SO_4 + 10 CO_2+8H_2O |
|
Answer» `1/18 M` |
|
| 9. |
Which of the following set of quantum numbers of an electron will have the highest energy |
Answer»  As we know increasing ORDER of energy is as .... ` Is LT 2s lt 2p lt 3s lt 3p lt 4s lt 3d lt 4p lt 5s lt 4d lt 5p lt 6s `therefore 3d lt 4p lt 5s lt 4d` ELECTRON of 4d, (means n = 4, Z = 2) has highest energy, from the given list. |
|
| 10. |
Which group of atoms have nearly same atomic radius- |
| Answer» Answer :C | |
| 11. |
Which of the following is strongly acidic ? |
|
Answer» Phenol 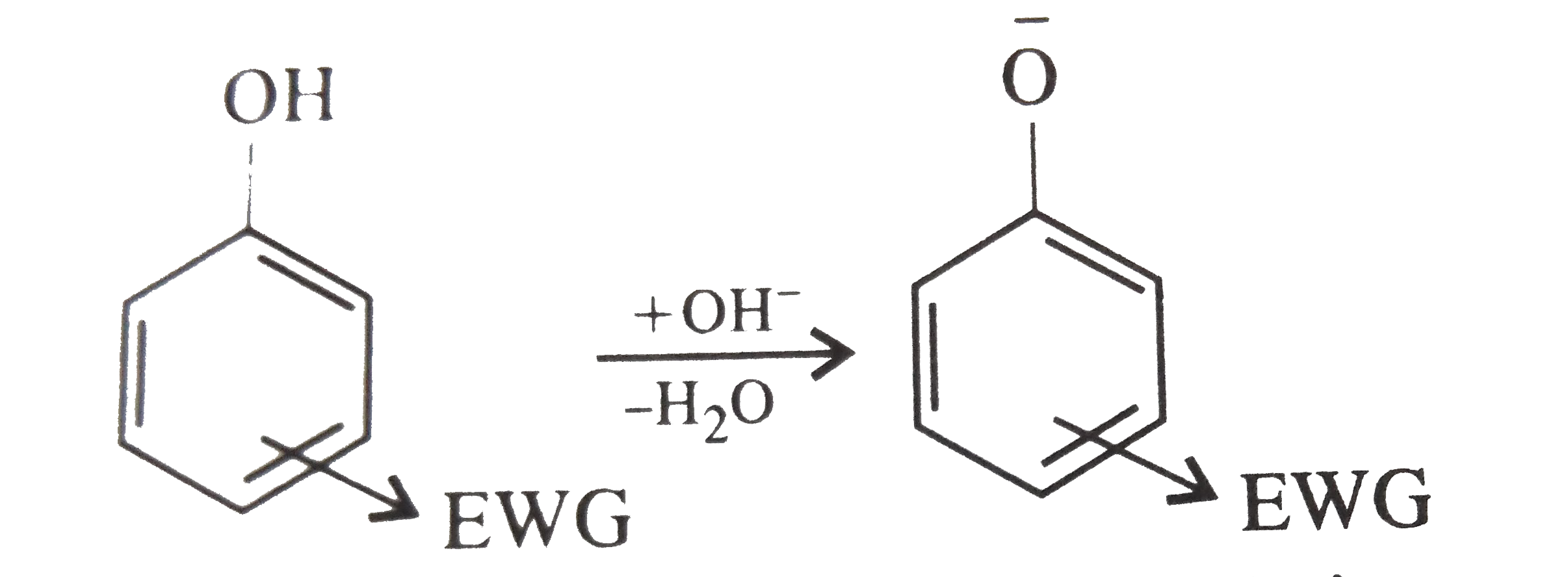 Electron-donating group such as `-CH_(3)` destabilizes the phenoxide ion by intensifying the negative charge relative to phenol. This tend to decrese the acidic strength of phenols 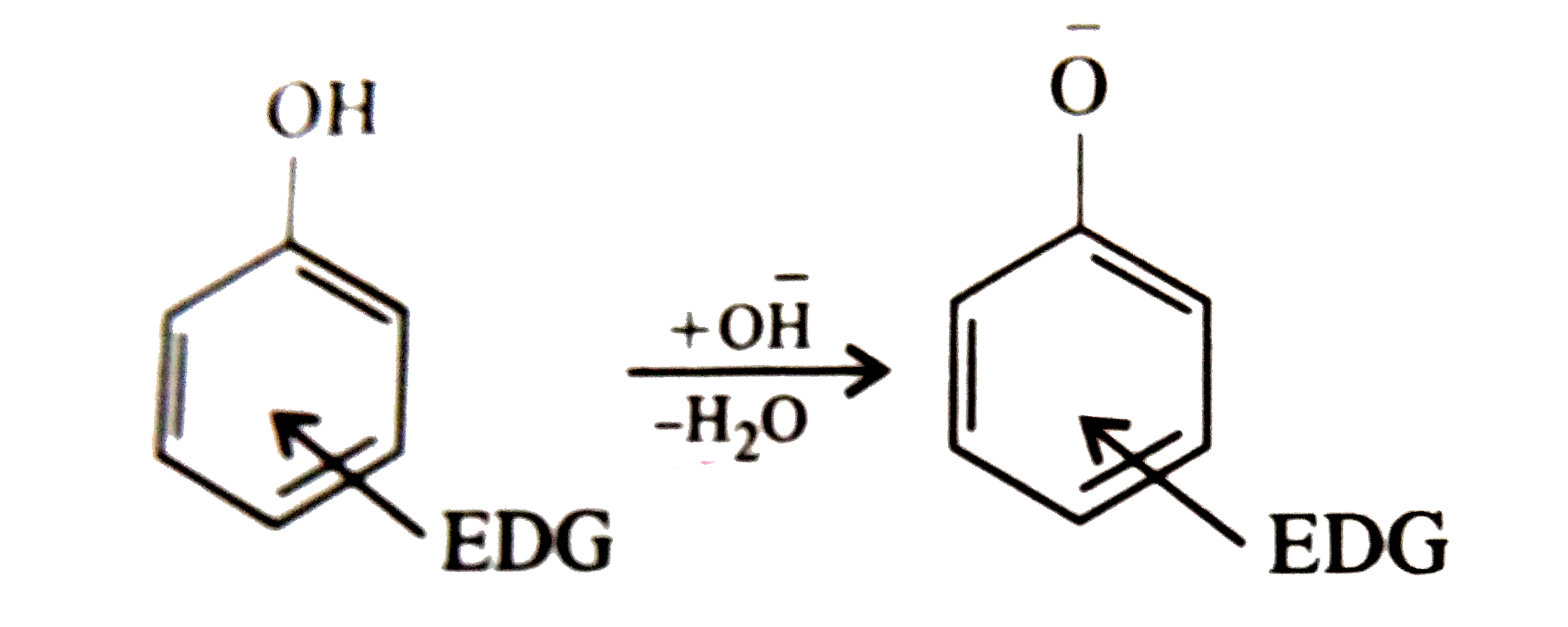 In cresols presence of `-CH_(3)` group decreases the acidic character while in nitrophenol presence of `-NO_(2)` increases the acidic character. Thus p-nitrophenol is most acidic. |
|
| 12. |
Which of the following compounds gives prussian blue colour in Lassaigne's test? |
|
Answer» `C_(6)H_(5)NH_(2)` |
|
| 13. |
Which are the methods of purification os organic compounds? |
| Answer» SOLUTION :Once an ORGANIC compound is extracted from a natural SOURCE or synthesised in the laboratory it is essential to purify it. The common techniques used for purification of organic compounds are as follows: (i) Sublimation (ii) Crystallisation (iii) Distillation (IV) DIFFERENTIAL extraction (v) Chromatogrphy | |
| 14. |
Write the IUPAC names of the following monofunctional compounds (a) CH_(3)CH_(2)CH_(2)Coverset(overset(OH)(|))(H)CH_(3) (b) CH_(3)-O-CH_(2)-CH_(2)-CH_(3) (c ) CH_(3)CH_(2) CH_(2)COOH (d) CH_(3) - overset(overset(CH_(3))(|))(N)-C_(2)H_(5) (e ) CH_(3)CH_(2)CN |
|
Answer» |
|
| 15. |
Which values can be obtained from the information represented by the vapour pressure curve of a liquid ? Normal boiling point Normal freezing point Enthalpy of vaporisation |
|
Answer» A only 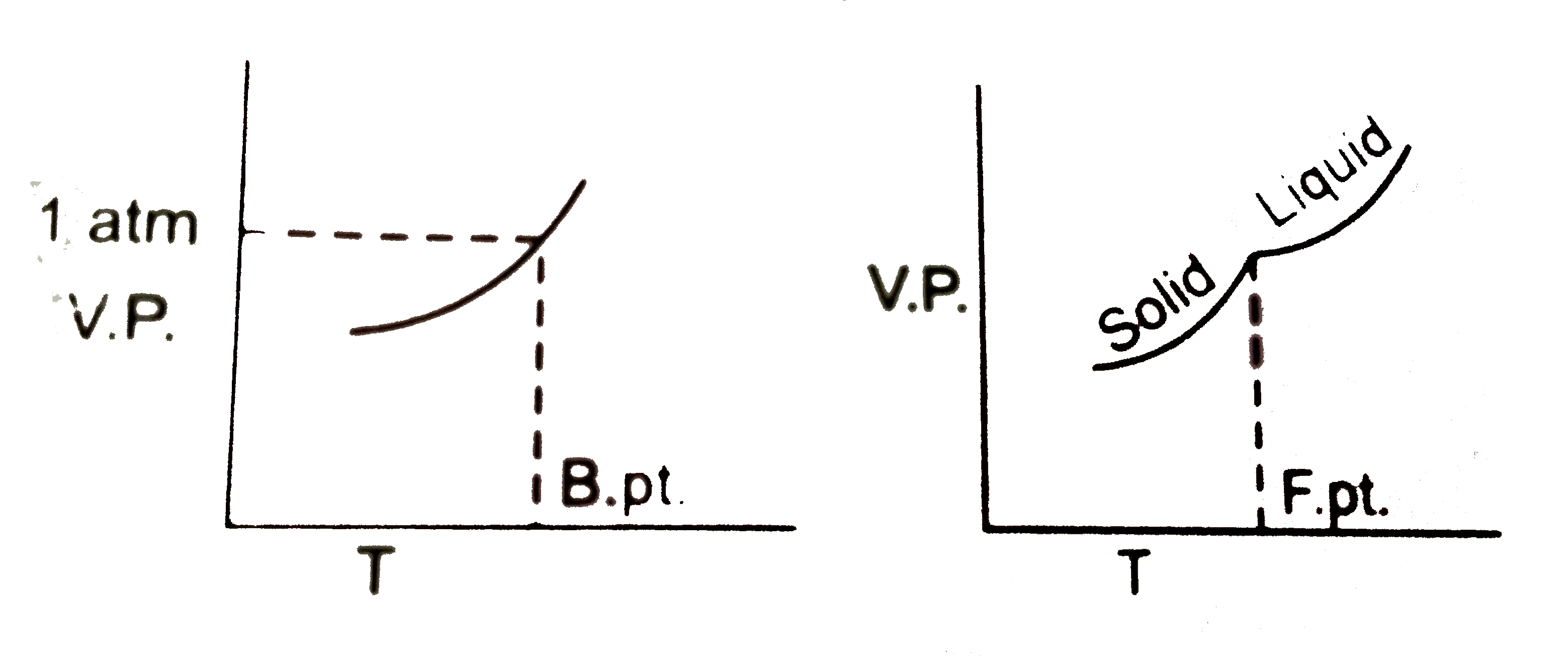
|
|
| 16. |
The substance used for regenerating the exhausted permutit is |
|
Answer» 100vol. `H_2O_2` |
|
| 17. |
What is meant by the term bond order ? |
|
Answer» Solution :The stability of the molecule can be restated in terms of bond order. A positive bond order `(" i.e. " N_(B) gt N_(a) )` MEANS a stable molecule. A NEGATIVE bond order `("i.e. " N_(b) ltN_(a))` means on unstable molecule. A ZERO bond order `("i.e. "N_(b) = N_(a))` means bond not possible. |
|
| 18. |
Who gave modern periodic law by observing X - ray spectra of element . |
| Answer» SOLUTION :HENRY MOSELEY in 1973 | |
| 19. |
The unit of viscosity is |
|
Answer» POISE |
|
| 20. |
When 400 mL of 0.2 N solution of a weak acid is neutralized by a diluate aqueous solution of sodium hydroxide under standard conditions, 4.4kJ of heatis liberated .Therefore, the standard enthalpy of neutralization of this weak acid in Kjeqiv^(-1) is |
|
Answer» `-11` `:. ` 100 ML of 1 MLOF 1N acid solution ( 1g EQ) willproduce heat`=( 4.4)/(400) xx ( 1000) /(0.2) =55kJ` `i.E.,DeltaH _("neut.") =-55kJ e quiv^(-1)` |
|
| 21. |
What is theIUPACnameofficialname andsymbol of theelementwith atomicnumber 110 ? |
| Answer» SOLUTION :IUPACNAME :Ununnilium(Unn) : Officialname :Darmstadtium , SYMBOL: DS | |
| 22. |
What volume of 0.10 M sodium formate solution should be added to 50 ml of 0.05 M formic acid to produce a buffer solution of pH 4.0 ? pK_(a) for formic acid is 3.80 . |
|
Answer» Solution :`pH=pK_(a) + log.(["SALT"])/(["Acid"]) :. 4=3.8+log.(["Salt"])/(["Acid"]) ORLOG.(["Salt"])/(["Acid"]) = 0.2 or(["Salt"])/(["Acid"])=1.5849` or `("No. of moles of salt")/("No. of moles of acid")=1.5849`(`:'` Volume is same for both) No. of moles of acid PRESENT `=(0.05)/(1000)xx50=2.5xx10^(-3)` `:.` No. of moles of salt `=1.5849 xx 2.5xx10^(-3)=3.962xx10^(-3)` Hence, volume of 0.1 M sodiumformate required `=(1000)/(0.1) xx 3.962xx10^(-3)=39.62 ml` |
|
| 23. |
Which lanthanoide compounds is used as a most powerful liquid lasers after dissolving it in selenium oxychloride |
|
Answer» CERIUM oxide |
|
| 24. |
Which gas is responsible for acid-rain ? |
|
Answer» `CO_2` `2H_2 O_((l)) + 4NO_(2(g)) + O_(2(g)) to 4HNO_(3(aq))` This acidic clouds STRETCHED with the flow of wind and it rains where the situation is PROPER. |
|
| 25. |
The stability of peroxide and superoxide of alkali metals increase as we go down the group. Explain giving reason. |
|
Answer» Solution : The stability of peroxides and SUPEROXIDES increases as the size of metal ion increases. `KO_(2) lt RbO_(2) lt CsO_(2)` LI gives only monoxide, Na gives peroxide and K, Rb and Cs give superoxide also. Peroxide ion and superoxide ion combine with large size of alkali metals. Stability increases as the size of cation increases. `underset("Oxides")(O_(2)^(2-)) overset((1)/(2)O_(2))to underset("Peroxide")(O^(2-)) overset(O_(2)) tounderset("Super oxide")( 2O_(2)^(-))` |
|
| 26. |
Value of C_(v) for mono atomic and diatomic gas are respectively .......... . |
|
Answer» `(1)/(2) R, (3)/(2) R` |
|
| 27. |
Write the drawbacks in Mendeleef's periodic table that let to its modification. |
|
Answer» i) Some elements having similar properties were placed inn different groups whereas some elements having dissimlar properties were placed in the same group. e.g. Alkali metals such as Li, Na, K, ETC. [(A group are grouped togther with coinage metals such as CU, Ag, Au (IB group) through their properties are quite different. Chemically similar elements such as Cu(IB group) and HG(IIB group) have been placed in different groups. ii) Some elements with higher atomic weights are placed before the elements with lower atomic weights in order to maintain the similar chemical nature of the elements. i.e., `._(18)^(39.9)Ar` and `._(19)^(39)K`, `._(27)^(58.9)Co` and `._(28)^(58.7)Ni` etc. iii) Isotopes did not find any place in the periodic table. However, according to Mendeleel's classification, these should be placed at different places in the periodic table. (All the above three defects were however removed when modern periodic law based on atomic number was given). iv) Position of hydrogen in the periodic table is not fixed but is controversal. v) Position of elements of group VIII could not be MADE clear which have been arranged i three trends without any justification. vi) It could not explain the even and odd series in IV, V and VI long periods. vii) Lanthanides and actinides which were discovered later on have been given proper positions in the main frame of periodic table. |
|
| 28. |
Which of the following compounds gives faster electrophilic aromatic substitution than benzene ? |
|
Answer»
|
|
| 29. |
What is the value of Gas constant R in Joule "Kelvin"^(-1) mol^(-1) ? |
|
Answer» `0.0821` |
|
| 30. |
The rms velocity of hydrogen is sqrt(7)times to that of nitrogen. If T is temperalure of the gas, then |
|
Answer» `T_(H_2) = T_(N_2)` |
|
| 31. |
The salts of which ions always neutral ? (pH = 7) |
| Answer» Solution :If the POSITIVE ions are `Na^(+), K^(+)` and NEGATIVE ions are `Cl^(-), NO_3^(-)` and `SO_4^(2-)` than the solution is always neutral. | |
| 32. |
The value of universal constant depends upon |
|
Answer» TEMPERATURE of the gas |
|
| 33. |
Which one of the following is the functional group of ketone? |
|
Answer» -CHO |
|
| 34. |
Which one of the following carbanions is least stable? (a) CH_3 CH_2^(-) (b) HC -= C^(-) (c ) (C_6 H_5)_3 C^(-) (d) CH_3^(-) (e) (CH_3)_3 C^(-) |
| Answer» | |
| 35. |
What is non-electrolyte ? |
| Answer» Solution :A compound whose aqueous solution or melt does not conduct ELECTRICITY is called a NON ELECTROLYTE. SUGAR, Glucose. | |
| 36. |
Which among the folloiwing series is obtained in both absorption and emission spectrums? |
|
Answer» LYMAN series |
|
| 37. |
The total number of electrons present in all the 's' orbitals, all the 'p' ortitals and all the 'd' orbitals of cesium ion are respectively |
|
Answer» 8, 26,10 |
|
| 38. |
Which is a characteristic reaction of an unsaturated compound? |
|
Answer» ADDITION REACTION |
|
| 39. |
Which one of the following is an example of disproportionation reaction? |
|
Answer» `3Cl_(2)(g)+6OH^(-)(aq)rarrClO_(3)^(-)(aq)+5CL^(-)(aq)+3H_(2)O(l)` |
|
| 40. |
Which of the following deplete ozone layer? |
|
Answer» `SO_2` |
|
| 41. |
What is the name of TiO_(2) according to stock notation nomenclature ? |
|
Answer» TITANIUM oxide (IV) |
|
| 42. |
Which values can be obtained from the information represented by the vapour pressure curve of a liquid ? 1. Normal boiling point 2. Normal freezing point 3. Enthalpy of vaporisation |
|
Answer» 1 only |
|
| 43. |
The two ions A^+ and B^- have radii 88 and 200 pm respectively. In the close packed crystal of compound AB, predict the coordination number of A^+ |
|
Answer» Solution :`r_+/r_(-) =(R(A^+))/(r(B^-))="88pm"/"200pm"=0.44` it lies in the range 0.414 to 0.732 HENCE, the coordination NUMBER of `A^+`=6 |
|
| 44. |
What is the formula of gypsum? What happens when it is heated? |
| Answer» Solution :`CaSO_(4). 2H_(2) O`. When heated to 393 K, it GIVES PLASTER of pairs `(CaSO_(4). 1//2H_(2)O)` but at 473 K it gives dead burnt plaster `(CaSO_(4))` | |
| 45. |
Which of the following expression is correct ? |
|
Answer» `V = a (z-b)` |
|
| 46. |
Why are electrongain enthalpies of Be andMgpositive ? |
| Answer» SOLUTION :THEYHAVE fullyfilled s- orbitalsand hencehave no tendency to ACCEPT an additionalelectron. Consequently,VALENCESHELL. That iswhyelectrongain enthalpies Beand Mg arepositive. | |
| 47. |
What is the stability order of these elements in their compounds in +2 oxidation states? Why? |
|
Answer» SOLUTION :The stability order: `Pb^(2+)gtSn^(2+)gtGe^(2+)`. This is due to INERT pair EFFECT. |
|
| 48. |
What is the IUPAC nomenclature of the given compound? |
|
Answer» 5-Ethynyl-1, 6-heptadiene |
|
| 49. |
Using the equation of state, pV=nRT, show that at a given temperature, density of a gas is proportional to the gas pressure, p. |
| Answer» Solution :. `(M(dRT)/(P)" or "d=(MP)/(RT)."If T"="constant",d PROP P)` | |
| 50. |
Which of the following has the highest tendency to give the reaction,M^(+)(g)overset(Aqueous)underset(Medium)toM^(+)(aq) |
|
Answer» Na |
|