Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Name an alkali metal carbonate which is thermally ustable and why? Give its decompostion reaction. |
|
Answer» Solution :`Li_(2)CO_(3)` is THERMALLY UNSTABLE because it is covalent. It decomposes to FORM `Li_(2) O` and `CO_(2)` `Li_(2) CO_(3) to Li_(2) O + CO_(2)` |
|
| 2. |
Name a suitable technique of separation of the components from a mixture of calcium sulphate and camphor. |
|
Answer» SOLUTION :A mixture of `CaSO_(4)` and camphor can be separated by the following two methods : (i) Camphor is sublimable but `CaSO_(4)` is not, therefore, sublimation of the mixture gives camphor on the sides of funnel while `CaSO_(4)` is left in the china dish. (ii) Camphor is soluble in organic solvents LIKE `CHCl_(3), C Cl_(4)` etc. while `CaSO_(4)` is not. Therefore, when the mixture is shaken with the solvent, camphor goes into solution while `CaSO_(4)` remains as RESIDUE. It is filtered and evaporation of solvent gives camphor. |
|
| 3. |
Name a suitable technique of separation of the components from a mixture of calcium, sulphate and camphor. |
| Answer» SOLUTION :SUBLIMATION. Because camphor can sublime whereas `CaSO_(4)` does not. | |
| 4. |
Name a suitable technique of separation of the components from a mixture of calcium sulphate and camphor |
| Answer» Solution :Sublimation technique is SUITABLE for separation of the COMPONENTS from a MIXTURE of calcium sulphate and camphor | |
| 5. |
Name a suitable method for separation of components from a mixture of camphor and calcium sulphate. |
|
Answer» |
|
| 6. |
Name a substance which could be added to cement without affecting its quality. |
| Answer» SOLUTION :FLY ASH. | |
| 7. |
Name a substance which acts as halogen carrier during the chlorination of benzene. |
| Answer» SOLUTION :`FeCl_3` | |
| 8. |
Name a salt that can be added to AgCl so as to produce cation vacancies. |
| Answer» SOLUTION :`CdCl_(2) or SrCl_(2)` | |
| 9. |
Name a pair of elements which exhibits diagonal relationship. |
| Answer» SOLUTION :LITHIUM (LI) and MAGNESIUM (MG). | |
| 10. |
Name a mixture used : (i) by all living beings (ii) in te construction for building materials (iii) as a food. |
|
Answer» |
|
| 11. |
Name a member of group 14 element in the periodic table which is used as semi conductor. |
|
Answer» |
|
| 12. |
Name a functional group whose IUPAC name has only a prefix but no suffix. Give one example. |
Answer» SOLUTION :NITRO `(-NO_(2))` GROUP has only a prefix but no suffix. 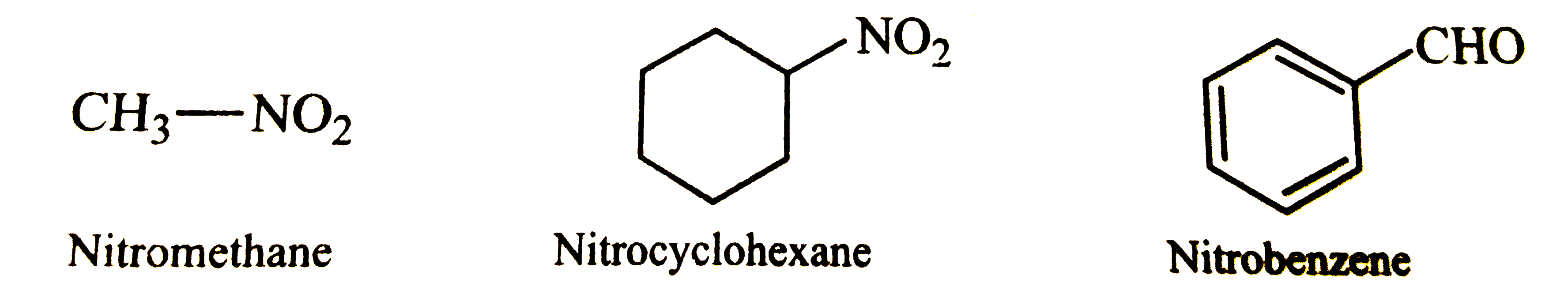
|
|
| 13. |
Name a compound whose empirical formula and molecular formula are equal. |
|
Answer» |
|
| 14. |
Name a compound which does not contain halogen but gives positive Beilestein test. |
|
Answer» |
|
| 16. |
NaI+AgNO_(3)toAgI+NaNO_(2) AgI+Fe to FeI_(2)+Ag FeI_(2)+Cl_(2) to FeCl_(2)+I_(2) (atomic mass of Ag=108, I=127, Fe=56, N=14, Cl=35.5). The above reaction is carried out by taking 75g of NaI and 255 kg of AgNO_(3). Therefore, the number of moles of iodine formed is- |
|
Answer» 0.5 |
|
| 17. |
NaHCO_(3) and NaOH cannot coexist in a solution because of |
|
Answer» common ION effectdue to common `Na^(+)` ions `NaHCO_(3)+NaOH rarr Na_(2)CO_(3)+H_(2)O` |
|
| 18. |
NaH produce H_(2) gas when |
|
Answer» It REACTS with water `NaHoverset("ELECTROLYSIS" )rarrNa+1/2H_(2)` |
|
| 19. |
Na_((g)) +Cl_((g)) to+Cl^(-) ""_((g)) Thisreactionis |
|
Answer» EXOTHERMIC |
|
| 20. |
NaCZ (Aq) + Ag NO3 (Aq) rarr AgC+ NaNO,(Aq). The change takes place in the reaction |
|
Answer» LOSS of `e^(-)` |
|
| 21. |
NaCl_((aq)) +AgNO_(3 (aq)) to AgCl darr +NaNO_(3(aq))thechange taking placehereis |
|
Answer» Loss of `BARE` |
|
| 22. |
NaCl solution is added to a saturated solution of PbCl_(2) . What will happen to the concentration of Pb^(2+) ions ? |
| Answer» SOLUTION :`Pb^(+)` ion CONCENTRATION will decrease to KEEP `K_(SP)` constant. | |
| 23. |
NaClwhenheatedwith conc: H_(2) SO_(4) andMnO_(2)liberates_______ |
|
Answer» HYDROGENGAS |
|
| 24. |
NaCl shows Schottky defects and AgCl Frenkel defects. Their electrical conductivity is due to |
|
Answer» MOTION of IONS and not the motion of ELECTRONS |
|
| 25. |
NaCl is not hygroscopic but common table salt is hygroscopic. Why? |
| Answer» Solution :Table SALT contains CALCIUM chloride and magnesium chloride as IMPURITES which are deliquescent in nature. THUS COMMON table salt is hygroscopic. | |
| 26. |
NaCl gives a golden yellow colour to the Bunsen flame which is due to. |
|
Answer» SUBLIMATION of metallic NA to give yellow vapour |
|
| 27. |
NaCl crystals require energy ( called lattice energy ) to break into ions. Then why NaCl is soluble in water ? |
| Answer» Solution :Dissolution of NaCl is a two step PROCESS.The 1st step involvingdissociation of NaCl into ionsrequires, called lattice energy but the 2nd step involves hydration of `NA^(+)` and`Cl^(-)` IONS in which energy is released called hydration energy. THUS, overall process is accompanied by release of energy. Hence NaCl dissolves in WATER. | |
| 28. |
NaCl does not exhibit space isomerism due to |
|
Answer» PRESENCE of ions |
|
| 29. |
NaCl crystals have some yellow colour. This is due to the presence of _________ |
|
Answer» |
|
| 30. |
NaCl crystal belongs to the crystal system |
|
Answer» hexagonal |
|
| 31. |
NaCl and CsCl have similar formula. Then why they have different structures ? |
| Answer» Solution :The radius ratio of NaCl `(r_(Na^+)//r_(Cl^-))` LIES in the RANGE 0.414 -0.732. HENCE , it has a coordination number of 6, i.e., octahedral arrangement . On the other HAND, the radius ratio of CsCl `(r_(CS^+)//r_(Cl^-))` lies in the range 0.732-1.0 . Hence, it has a coordination number of 8, i.e., it has a body-centred cubic arrangement. | |
| 32. |
NaBH_(4) + I_(2) to X + Y + X BF_(3) + NaH overset(450K)(to) X + P BF_(3) + LiAIH_(4) to X + Q + R X,Y,Z,P,Q and R reaction are {:(X,Y,Z,P,Q,R):} |
|
Answer» `{:(Na_(4)B_(4)O_(7),NAI,HI,HF,LiF,AIF_(3)):}` `2BF_(3) + 6NaH overset(450 k)(to) underset((X))(B_(2)H_(6)) + underset((F))(6NaF)` `4BF_(3) + 3LiAIH_(4) to underset((x))(2B_(2)H_(6)) + underset((Q))(3LiF) + underset((R ))(3AIF_(3))` |
|
| 33. |
NaBH_(4) + I_(2)rarr X uarr + Y uarr + 2Nal X + C_(2)H_(5)OH rarr Y uarr + D X + HCl rarr Y uarr + E D gives following colour with flame |
|
Answer» Red |
|
| 34. |
Na_(2)SO_(4).10H_(2)O(s)hArrNa_(2)SO_(4).5H_(2)O(g) K_(P)=2.43xx10^(-10) atm^(5) incorrect statement is- |
|
Answer» If partial pressure of `H_(2)O` in CONTAINER is `3.5xx10^(-2)` atm amount of `Na_(2)SO_(4).5H_(2)O` decreases. |
|
| 35. |
Na_(2)SO_(4) is water soluble but BaSO_(s) is insoluble because |
|
Answer» the hydration energy of `Na_(2)SO_(4)` is higher than that of its lattice energy |
|
| 36. |
Na_(2)O + CO_(2) to |
| Answer» Solution :`Na_(2)O + CO_(2) overset("FUSE")(to) Na_(2)CO_(3)` | |
| 37. |
Na_(2)CO_(3).10H_(2)O when exposed to air looses water of crystallisation and gives finally |
|
Answer» `NaCO_(3). 4H_(2)O` |
|
| 38. |
Na_(2)CO_(3) + Fe_(2)O_(3) rarr A + CO_(2)What is A tn the reaction? |
|
Answer» `NaFeO_(2)` |
|
| 39. |
Na_(2)B_(4)O_(7).10H_(2)O overset(conc. HCl)rarr A overset(160^(@)C)rarr B. Compound 'B' is |
|
Answer» `H_(2)B_4O_7` |
|
| 40. |
Na_(2)B_(4)O_(7).10H_(2)O can also be represented as |
|
Answer» `Na_(2) [B_(4) O_(5) (OH)_(4)].8H_(2)O` `Na_(2)B_(4)O_(7).10H_(2)O` `B_(2)H_(6)+2NH_(3)overset(120^@C)rarrB_(2)H_(6).2NH_3` |
|
| 41. |
Na_(2)B_(4)O_(7)+H_(2)Ooverset("Conc. HCl")underset(Delta)rarrNaCl+X Xoverset(Delta)rarrY. The product Y in the reaction is |
|
Answer» CRYSTALLINE B |
|
| 42. |
Na_(2)CO_(3) can be manufactured by Solva's process but K_(2)CO_(3) cannot be prepared because |
|
Answer» `K_(2) CO_(3)` is moresoluble |
|
| 43. |
Na_2 B_4 O_7.10 H_2 O can also be represented as |
|
Answer» `Na_2 [B_4 O_5 (OH)_4 ]. 8 H_2 O ` |
|
| 44. |
Na reacts with phenol |
|
Answer» `H_(2)` gas |
|
| 45. |
Na shows which type of color flame ? |
|
Answer» Yellow |
|
| 46. |
Sodium is heated in air at 300^(0)C to form X, X absorbs CO_(2) and forms Na_(2)CO_(3) and Y. Which of the following is Y. |
|
Answer» `H_(2)` |
|
| 47. |
Na^+ has higher value of ionization energy than Ne. So ionisation energy of Na^+ is more than Ne. |
|
Answer» |
|
| 48. |
Na and Mg crystallise in BCC and FCC type crystals, than the number of Na and Mg atoms present in the unit cell of their respetive crystal is |
|
Answer» 4 and 2 No. of atoms per unit cell in FCC structure = 4 |
|
| 49. |
Na and K have low density, because... |
|
Answer» they have SMALLER size. |
|
| 50. |
N^(3-),O^(2-),F^-,Na^+, Mg^(2+),Al^(3+) what is comman in them ? |
| Answer» SOLUTION :They are ISOELECTRONIC (10 ELECTRONS). | |