Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Number of moles of O_2 gas evolved bythe decomposition of 1 lit of 1N H_2O_2 solution is |
|
Answer» 0.5 |
|
| 2. |
How many moles of KMnO_(4) are required to oxidise one mole of ferrous oxalate in acidic medium ? |
|
Answer» 7.5 moles |
|
| 3. |
Number of moles of KMnO_(4) required to oxidize one mole of Fe(C_(2)O_(4)) in acidic medium is _____ |
|
Answer» 0.6 |
|
| 4. |
Number of moles of K_(2)Cr_(2)O_7 , reduced by one mole of Sn^(2+) ions is 1/x.The value of x is |
|
Answer» Thus ,1 moles of `Sn^(2+)`reduces `1/3` moles of `K_(2)Cr_(2)O_7` |
|
| 5. |
Number of moles of K_(2)Cr_(2)O_(7) that can be reduced by 1 mole of Sn^(2+) ions is : |
|
Answer» `1//3` `Cr_(2)O_(7)^(2-)+14H^(+) +3Sn^(2+) to 2Cr^(3+)+7H_(2)O+3Sn^(4+)` `THEREFORE 1 " mole of " Sn^(2+)` will reduce `1//3 " mole of " K_(2)Cr_(2)O_(7)` |
|
| 6. |
Number of moles of K_(2)Cr_(2)O_(7) reduced by one mole of Sn^(2+) is : |
| Answer» SOLUTION :N//A | |
| 7. |
Number of moles of KMnO_(4) required to oxidize one mole of Fe(C_(2)O_(4)) in acidic medium is |
|
Answer» `0.6` |
|
| 8. |
Number of moles of K_(2)Cr_(2)O_(7) reduced by 1 mole of Sn^(2+) is ____ |
|
Answer» `1/6` |
|
| 9. |
Number of moles of hydrogen peroxide that can oxidise one mole of lead sulphide to lead sulphate is |
|
Answer» 1 |
|
| 10. |
Number of moles of FeSO_4 that can reduce one mole of permanganate in aqueous acid solution is |
|
Answer» 2 |
|
| 11. |
Number of moles of electrons shared in the formation of a dioxygen molecule . |
|
Answer» 2 |
|
| 12. |
Number of moles of Cul (K_(sp) = 5 xx 10^(-12)) that will dissolve in 1 L of 0.1 M Nal solution is |
|
Answer» `2.2 XX 10^(6) ` `S xx 0.1 = 5 xx 10 ^(-12)= S = 5 xx 10 ^(-11) ` |
|
| 13. |
Number of molecules of an ideal gas present in a flask of volume 2 L are 10^(23). The mass of each gas molecule is 6.64xx10^(-23)g and the root mean square velocity of the molecules is 4.33xx10^(4)cm*s^(-1). Hence- |
|
Answer» pressure of the GAS is 3.27 atm |
|
| 14. |
Number of molecules in which steric inhibition of resonance is observed is |
|
Answer» In b,c,d,e the bulky GROPUS present at ortho position inhibit DELOCALISATION. |
|
| 15. |
The number of molecules in one litre of air at STP is |
|
Answer» `(6.02 XX 10^(23))/(32)` |
|
| 16. |
Number of metals presents in 'alnico' alloy is |
|
Answer» |
|
| 17. |
Number of meso isomers of he given compound is |
Answer» 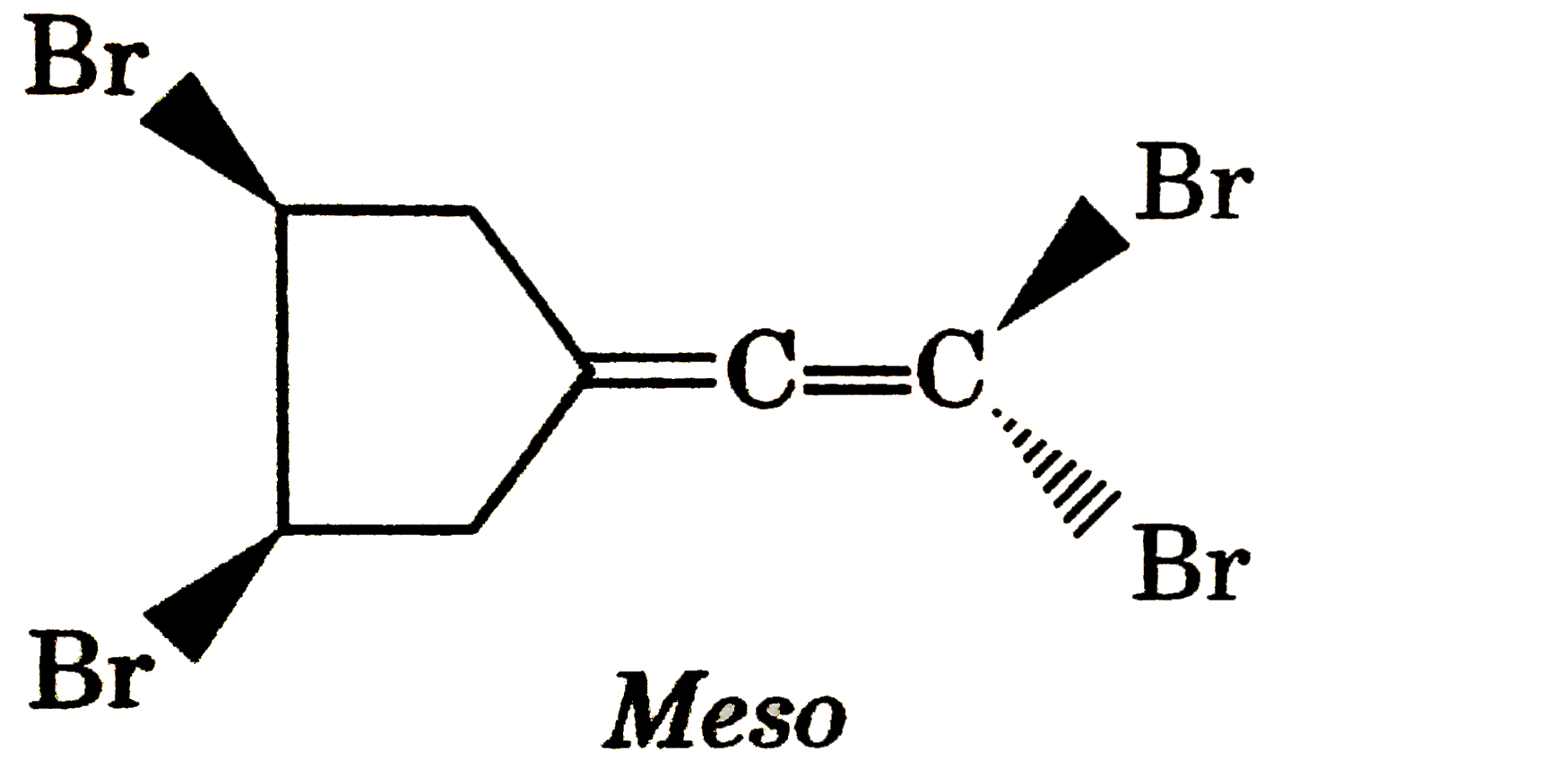
|
|
| 18. |
Number of meso somer of the given compound is : |
|
Answer» 1 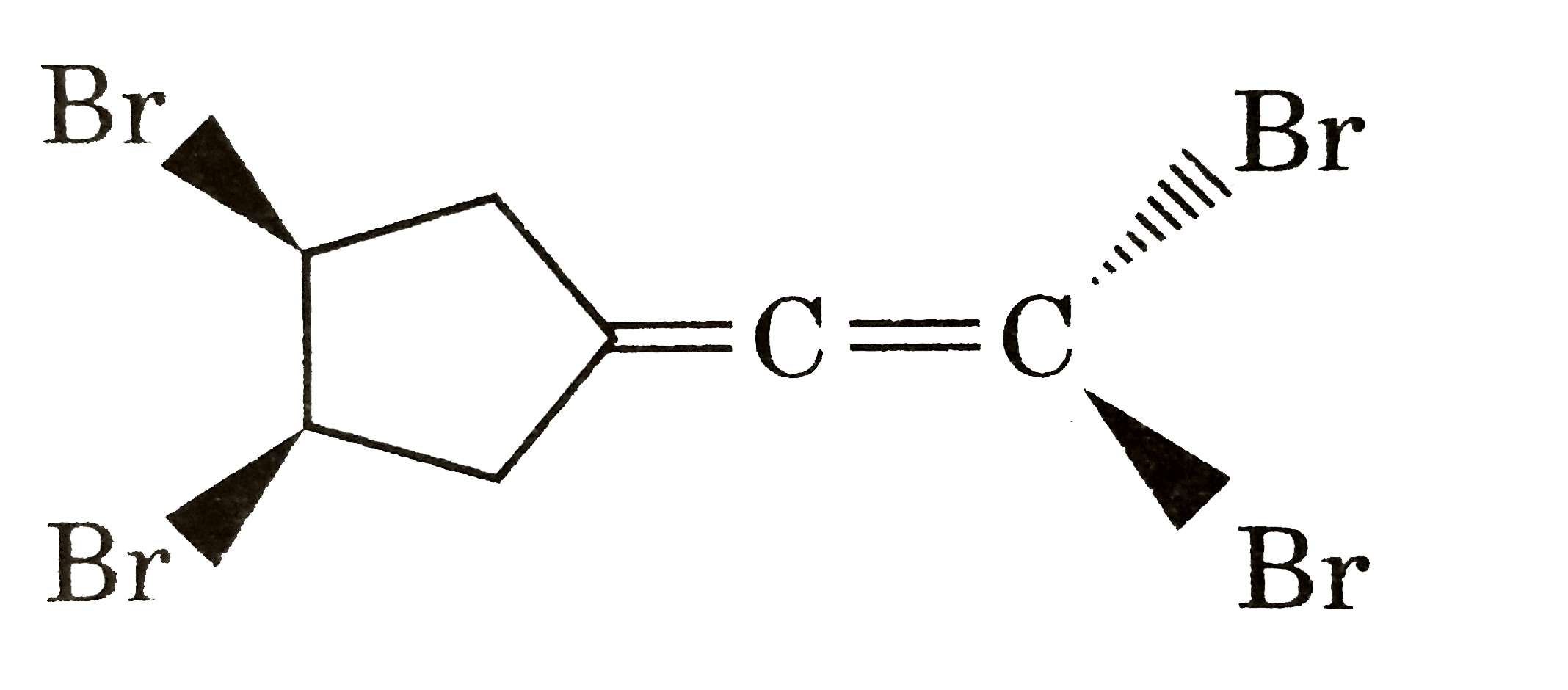
|
|
| 19. |
Number of measurable parameters of a gas is |
|
Answer» 1 |
|
| 20. |
Number of lone pair (s) present in the structure ofHNO_(3) is |
Answer» 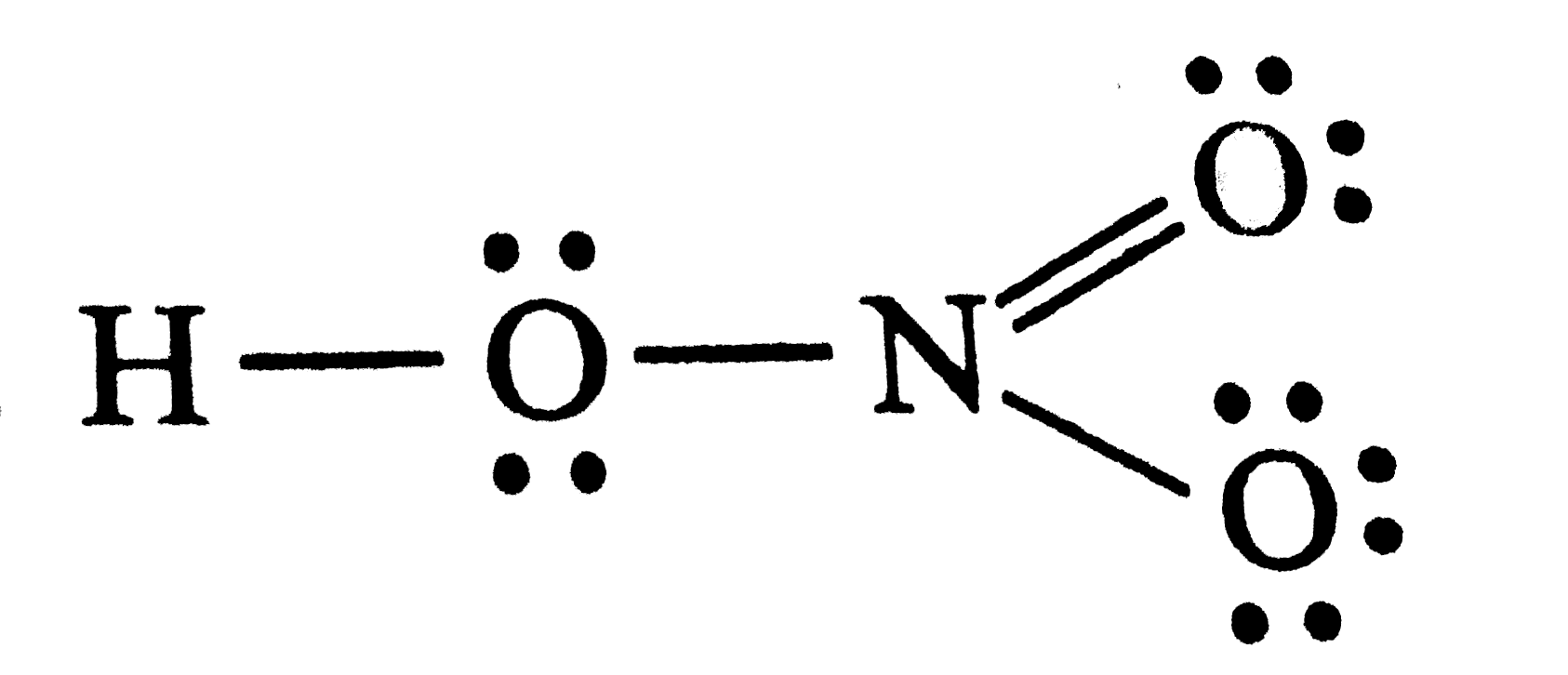 THUS , these are 7 LONE PAIRS of electons |
|
| 21. |
Number of linear atoms in acetylene molecues is a maximum of |
|
Answer» 1 |
|
| 23. |
Number of isomers possible for dichlorocyclohexane |
|
Answer» 8 3-geomentric isomers (1,2,1,3,1,4) 1-optical ISOMER(1,3) |
|
| 24. |
Number of isomers possible for dichloro cyclohexane |
|
Answer» 8  1, 3 dichloro CYCLO hexane |
|
| 25. |
Number of isomers for the compound dihydroxy benzene |
|
Answer» 1 
|
|
| 26. |
Number of nucleons in D_(2) molecule is |
|
Answer» 1 |
|
| 27. |
Number of hexagonal rings in borax. |
Answer» 
|
|
| 29. |
Number of HCl molecules present in 10 mL of 0.1 N HCl solution is |
|
Answer» `6.022 XX 10^(23)` |
|
| 30. |
Number of H_(2) molecules attached to each H_(2)O molecule through hydrogen bonding is |
|
Answer» is LINKED to 4 other MOLECULES tetrahedrally . |
|
| 31. |
Number of g of oxygen in 32.2 g Na_(2)SO_(4)* 10H_(2)O (Mol. omega t = 322) is ......... |
|
Answer» `16.0` |
|
| 32. |
Number of gram equivalents of solute in 100ml of 5NHCl solution is |
|
Answer» 50 |
|
| 33. |
Number of geometrical isomers for the for the following compound will be : |
|
Answer» |
|
| 34. |
Number of faraday required to convert 1 mol Cr_(2)O_(7)^(2-) to Cr^(3+) ion is.... |
|
Answer» |
|
| 35. |
Number of elements present in 5th period is..... |
|
Answer» 8 |
|
| 36. |
Number of electrons transferred from one atom to another during bond formation in SrS (Strontium Sulphide) |
|
Answer» 1 |
|
| 37. |
Number of electrons taken up by nitrogen atom when a NO^(+)ion is reduced to NH_(2)OHis: |
|
Answer» |
|
| 38. |
Number of electrons transferred from one Al atom during bond formation in Aluminium fluoride |
|
Answer» 1 |
|
| 39. |
Number of electrons present in P^(-3) ion having 15 protons a r e ...... |
|
Answer» 15 |
|
| 40. |
Number of electrons involved in the reduction of Cr_(2)O_(7)^(2-) ion in acidic solution to Cr^(3+)is |
| Answer» Solution :`{:(Cr_(2)O_(7)^(-2)RARR, 2Cr^(3+)),(14H^(+),+7H_(2)O),(+6e^(-),):}` | |
| 41. |
Number of electrons forming bonds in O_(2) molecule according to valency bond theory. |
|
Answer» 2 |
|
| 43. |
Number of electron-rich hydrides among the following are : CH_(4),NH_(3),PH_(3),H_(2)O,H_(2)S,BH_(3),HF,AlH_(3),AsH_(3). |
|
Answer» |
|
| 44. |
Number of delocalised electrons in C_6H_6molecule is |
|
Answer» 9 |
|
| 45. |
Number of correct statements are : DeltaU =nC_(Vm)Delta T is applicable for ideal gas undergoing reversible process but not irreversibe (No chemical and phase change) DeltaS =nC_(Vm)Delta T is applicable for ideal gas undergoing reversible process but not irreversible (No chemical and phase change) DeltaS=nC_(Vm)ln"(T_(2))/T_(1)+nRln'(V_(2))/V_(1) is applicable for ideal gas undrgoing reversible as well as irreversibel process (No chemical and phase change ) DeltaH=DeltaU+Delta(PV) isapplicable for chemical and phase change carried out reversibly or irreversibly PV^(gamma) = constant for reversible and irreversible adiabatic process involving ideal gas. |
|
Answer» |
|
| 46. |
Number of collinear carbons in 2,4-hexa diyne |
|
Answer» 3 |
|
| 47. |
Number of chiral centers generated during monochlorination in the above reaction: |
|
Answer» 1 |
|
| 48. |
Number of chiral centres present in above compound. Number of teoretically possible stereoisomer of above compound. Degree of unsaturation (DBE) of above compound. |
|
Answer» (b) Steresisomer = `2^(N)=2^(6)=64` where n stereogenic area (c ) SIX (THREE `pi` bond and ONE monocyclic RING one bicyclic ring) |
|
| 49. |
Number of chiral carbons inDiosgenin are : |
Answer» 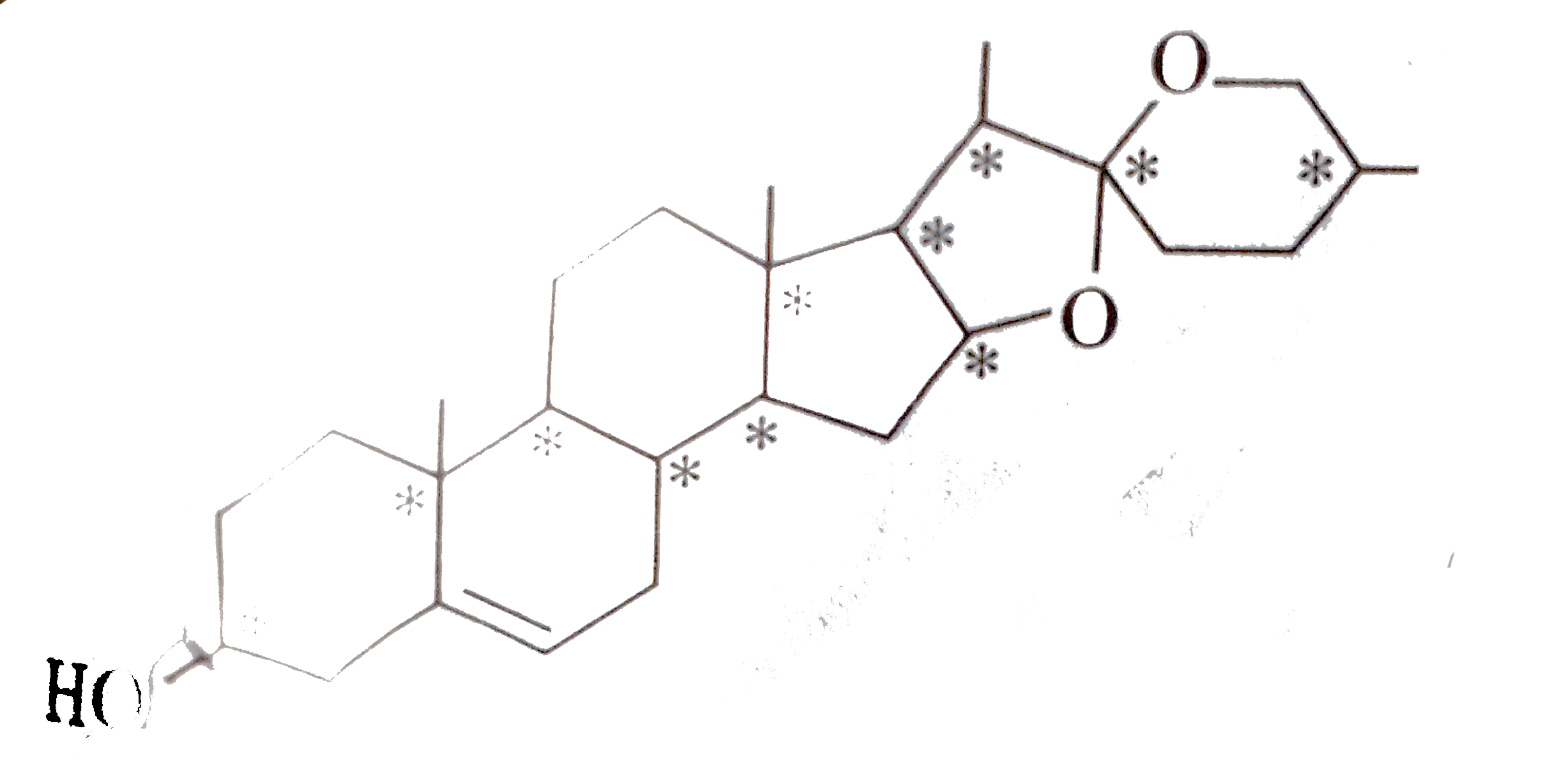
|
|