Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Poly substitution is a major drawback in(a)Reimer Tiemann reaction(b)Friedel Crafts acylation(c)Friedel Crafts alkylation(d)acetylation of aniline. |
|
Answer» REIMER Tiemann reaction |
|
| 2. |
Pollution can be controlled if : |
|
Answer» all automobiles MUST be FITTED with exhaust system scatalytic converters |
|
| 3. |
Pollutant of automobile exhausts that effects nervous system /produces mental diseases is |
|
Answer» Mercury |
|
| 4. |
Pollutant of automobile exhausts that affects nervous system and producers mental diseases is |
|
Answer» LEAD |
|
| 5. |
PolarizingpowerCd^(2+) on the anions is stronger then that of Ca^(2+) ion. This is because |
|
Answer» ATOMIC number of CD is greater than that of CA |
|
| 6. |
Polarity in a molecule and hence the dipole moment depends prl marily on electro- negativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment ? |
|
Answer» `CO_(2)` `CO_(2)` being symmetrical so zero DIPOLE moment.  Among HI, `SO_(2) and H_(2) O` dipole moment is highest for `H_(2)`O as in it the central ATOM process 2 LONE pairs. 
|
|
| 7. |
Polarity in a molecule and hence the dipole moment depends primarily on electronegativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment ? |
|
Answer» `CO_(2)` 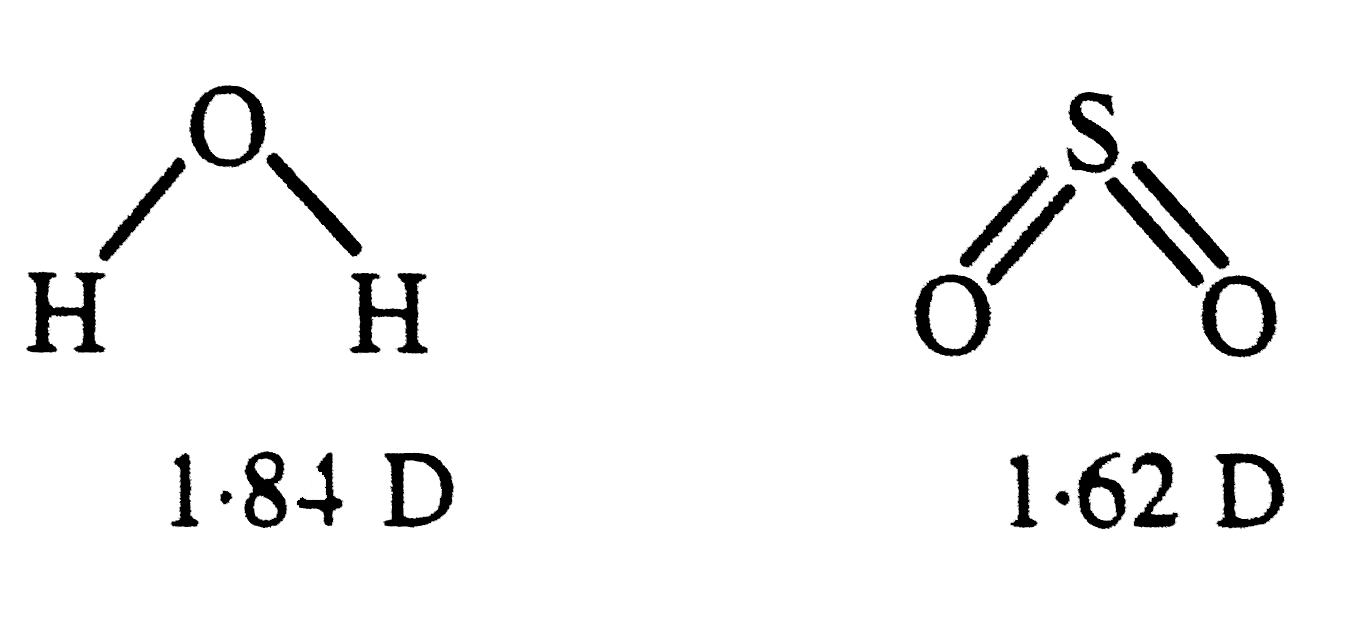
|
|
| 8. |
Polarization of electrons in acroleinmay be written as |
|
Answer» `H_(2)^(DELTA-)C=CH-underset(delta+)OVERSET(overset(O)(||))(C)-H` |
|
| 9. |
Poisonous has 'Lewisite' is obtained by the reaction of |
|
Answer» `CH -= CH` and `AsCl_(3)` |
|
| 10. |
Poisonous gas present in the exhaust fumes of car is |
|
Answer» Ulraviolet RAYS |
|
| 11. |
Poisonous gas present in the exhaust fumes of an automobile is |
|
Answer» `CO_2` |
|
| 13. |
Poise stands for |
|
Answer» `1 "dynes CM sec"^(-2)` |
|
| 14. |
Point out the wrong statement. in a given period of the periodic table, the s-block elements has in general, a lower value of..... |
|
Answer» Electronegativity |
|
| 15. |
Point out the incorrect statement about resonance? |
|
Answer» RESONANCE structures should have EQUAL energy |
|
| 16. |
Point out the false statements in the following: (a) Lead shows allotropy. (ii) Carbon shows sp^(3) hybridisation in both diamond and graphite. (c) Tin exists in three allotropic forms. These are white. Grey and brittle. (d) +2 oxidationstate is more common in tin than in lead. (f) Carbon has themaximum tendency to catenate among theelements of group-14. (f)Both CO_(2) and SiO_(2) are gaseous at room temperature (g) Elemental silicon does not have graphites-like structure. (h) Stannous chloride can act as an oxidising agent inreactions. (i)C Cl_(4) can act as Lewis acid. |
|
Answer» Solution : (a) Lead is the only element in group-14 which does nto show any allotorpy. (B) Carbon shows `sp^(3)` hybridisation only in diamond. In graphite, the carbon atom sare `sp^(2)` hybridised. (d) +2 oxidation state is more COMMON in lead than in tin due to greater inert pair effect. (f) only `CO_(2)` is a gas while `SiO_(2)` is a solid. (h) Stannous chloride `(SnCl_(2))` canact as reduc ing agent in its reactions because the element tin (SN) can increase its oxidation number from +2 to +4but cannot decrease it (i) `C Cl_(4)` cannot act as Lewis acid since carbon atom has novacant orbitals to accept electrons. |
|
| 17. |
Point out the false statements from the following and rectify them : (a) The members in a homologous series can be represented by same general formula. (b) C_(n)H_(2n) is the general formula for the members of alkane familt. (c) Prefixes n-, iso-, tert-, neo- etc. are used in the IUPAC names of the compounds. (d) Neohyfrocarbon contains a tertiary carbon atom. (e) Propane consists of two primary and one secondary carbon atoms. (f) Chemical properties of compounds are those of the functional groups. (g) CHO group gets priority over COOH group in writing the IUPAC names of the compounds. |
|
Answer» Solution :(b) `C_(n)H_(2n)` is the general formula for the members of the alkane familt. (C) Prefixes n-, iso-, tert-, neo- ETC. are used in the common NAMES of the COMPOUNDS. (d) Neohydrocarbon contains a quaternary carbon atom. (g) `COOH` group gets priority over `CHO` group in writing the IUPAC name of the compound. |
|
| 18. |
Point out the false statement |
|
Answer» Colloidal SOLS are homogeneous |
|
| 19. |
Point out the difference between London dispersion forces and dipole-dipole forces. |
| Answer» SOLUTION :`1:1,KE=sqrt(3RT//M)` | |
| 20. |
Pointout hewrongstatementin a givenperiod of theperiodicitable, the s- blockelements has , ingenerala lowervalueof |
|
Answer» ELECTRONEGATIVITY |
|
| 21. |
(pm)-2 Butanol is optically inactive. Give reasons. |
| Answer» Solution :`(pm)`-2 Butanol is a racemic MIXTURE and hence contains equal amounts of the two ENANTIOMERS, i.e, (+)-2- butanol and (-)-2- butanol. Whereas (+)-2-butanol rotates the PLANE of polarized light toward right, (-)-2-butanol rotates the plane of polarized light towards left but to the same extent. The OVERALL rotation is zero and hence `(pm)`-2-butanol is optically INACTIVE. | |
| 22. |
Plots of log K Vs (1)/(T) plots shows an intercept of 2 on y-axis with a slope of 45^(@) for the studied reactions. Assuming that Delta H^(@) and Delta S^(@) astemperature independent, select the correct statement |
|
Answer» `DeltaG^(@)= -2.75 K.cal` `DeltaH^(@)-T Delta S^(@)= -2.303 RT logK` `log_(k)=((-Delta H^(@))/(2.303R))(1)/(T)+(DeltaS^(@))/(2.303R)` slope= `(-DeltaH^(@))/(2.303R)=TAN 45^(@)` `DeltaH^(@)= -2.303 xx 2 xx 1= -4.606` cal Intercept = `(DeltaS^(@))/(2.303R)=2` `DeltaS^(@)=9.212 cal implies DeltaG_(P)=DeltaH^(@)=T Delta S^(@)` `= -4.606-298 xx 9.212 xx 10^(-3)` `DeltaG^(@) = -2.75` k cal `log_(10)^(k)=(-DeltaG^(@))/(2.303RT)=(2.75 xx 10^(3) cal)/(2.303 x 2 xx 298)=2` `K=10^(2)=100` |
|
| 23. |
Plot of Maxwell's distribution of velocities is given below : Which of the following is correct about this plot? |
|
Answer» `f_1 GT f_2` |
|
| 24. |
Plots for 2s orbital are: X,Y,and Z are respectively. |
|
Answer» `R,R^(2)and4pir^(2)R^(2)` |
|
| 25. |
Plot of log x/m against log p is a straight line inclined at an angle of 45^(@) . When the pressure is 0.5 atm and Freundlich parameter, k is 10.6, the amount of the solute adsorbed per gram ofadsorbent will be ( log 5 =0.0990) |
|
Answer» 1 g or `LOG(x)/(m)log k+(1)/(n) logp` `therefore` plot of log x/m vs log p is a straight LINE with slope=1/n and intercept = log k. Thus, `(1)/(n)= tan theta = tan 45^(@)=1 " or " n=1` At p=0.5 atm and k=10 x/m =10(0.52)=5 When m=1g `therefore` x (i.e., amount adsorbed PER gram )=5 g |
|
| 26. |
Plot a graph with respect to Henry's law stating the solubility of HCl gas in the solution in cyclohexane |
|
Answer» Solution :ACCORDING the Henry's law `P_("SOLUTE") =K_HX_("solute") ` ` P_("solute ") ""-` Vapour PRESSURE `K_H "" `- Empirical constant ` X_("Solute ") "" - ` MOLE fraction ` (##SUR_CHE_XI_V02_C09_E03_005_S01.png" width="80%"> |
|
| 27. |
Platinum (atomic radius = 1.38 Å) crystallises in a cubic closed packed structure. Calculate the edge length of the face-centred cubic unit cell and the density of the platinum (Pt = 195) |
|
Answer» Solution :`r = (a)/(2sqrt2)` (for fcc), `a = 2 sqrt2 r = 3.9 Å` DENSITY `= (ZM)/(a^(3)N_(0)) = (4 xx 195)/((3.9 xx 10^(-8))^(3) xx 6.023 xx 10^(23)) = 21.83 g//cm^(3)` |
|
| 28. |
Platinised asbestos helps in the formation of SO_(3) from SO_(2) and O_(2). But, if even a small amount of As_(2)O_(3) is present the platinised asbestos does not help in the formation of SO_(3). As_(2)O_(3) acts here as |
|
Answer» A POSITIVE catalyst |
|
| 29. |
Plaster of Paris is used to joint fracture bones and keeping them immobilize. |
| Answer» SOLUTION :TRUE STATEMENT | |
| 30. |
Plaster of Paris is represented as |
|
Answer» `CaSO_(4) . 2H_(2)O` |
|
| 31. |
Plaster of Paris is obtained by heating ……….at ……….and its formula is ……. . |
| Answer» Solution :gypsum `(CaSO_(4). 2H_(2)O) , 393 K , CaSO_(4).1//2 H_(2)O` | |
| 32. |
Plaster of Paris is hardened by |
|
Answer» Liberating `CO_(2)` |
|
| 33. |
What is plaster of Paris and how is it prepared? |
|
Answer» `CaSO_4 . H_2O` |
|
| 34. |
Plaster of Paris is ………. |
|
Answer» `CaSO_(4)*2H_(2)O` |
|
| 35. |
Plaster of paris hardens by |
|
Answer» Giving off `CO_(2)` |
|
| 36. |
Plants such as Pinus, Juniparus, Pyrus and Vitis metabolize nitrogen oxides |
|
Answer» |
|
| 37. |
Plank's constant has the dimensions of |
|
Answer» Force |
|
| 38. |
Planck's constant has the units of |
|
Answer» WORK |
|
| 39. |
Place the following systems in order of increasing randomness : (a)1 mol of a gas X (b) 1 mol of a solid X (c) 1 mol of a liquid X. |
|
Answer» Solution :Order of increasing RANDOMNESS: 1 MOL of solid X `LT` 1 mol of liquid X `lt` 1 mol of gas X. |
|
| 40. |
pK_(b) of aq. NH_(3) is 4.74 hence pH of 0.01 M NH_(3) solution is |
|
Answer» 3.37 |
|
| 41. |
pK_b ofCN^(-)is 4.7Calculate (a) hydrolysis constant and (b) degree of hydrolysis of 0.5M aqueous NaCN solution. |
|
Answer» Solution :`pk_b `of ` CN^(-) = 4.7 ` therefore `k_b `of ` CN^(-) = 10^(-4.7) = 2 xx 10^(-5)` Hydrolysis constant (K) is given in TERMS of ionic PRODUCT of WATER (`K_w`) as `k_h = (k_w)/(k_a) =k_w xx (k_b)/(k_w)=k_b = 2 xx 10^(-5)` Degreeof hydrolysis(H )is givenas , h`= sqrt( k_h //c) = sqrt(2 xx 10^(-5) //0.5 ) = 6.3 xx 10^(-3)` |
|
| 42. |
pK_(a) of weak acid (HA) and pK_(b) of weak base (BOH) are 3.2 and 3.4 respectively. The pH of their salt (AB) solution is |
|
Answer» `7.0` `=7-0.1=6.9` |
|
| 43. |
Pka of O_(2)N-CH_(2)COOH is 1.68. How many compounds given below have a pKa value greater than 1.68 ""underset((I))(CH_(3)COOH)""underset((II))(Cl-CH_(2)COOH)""underset((III))(Cl-underset(underset(Cl)(|))overset(overset(Cl)(|))(C)-COOH)""underset((IV))(O_(2)N-CH_(2)CH_(2)COOH)"underset((V))(H_(3)C-overset(overset(O)(||))(C)-COOH)"" |
|
Answer» |
|
| 44. |
pK_a of pure water at certain temperature is 13.4. What is the hydronium ion concentration of water at that temperature ? |
| Answer» SOLUTION :`2 XX 10^(-7) M` | |
| 45. |
P^(K_(a)) of O_(2)N-CH_(2)COOH is 1.68. How many compounds given below have a pKa value greater than 1.68 ""underset((I))(CH_(3)COOH)""underset((II))(Cl-CH_(2)COOH)""underset((III))(Cl-underset(underset(Cl)(|))overset(overset(Cl)(|))(C)-COOH)""underset((IV))(O_(2)N-CH_(2)CH_(2)COOH) ""underset((V))(H_(3)C-overset(overset(O)(||))(C)-COOH)""(##AKS_ELT_AO_CHE_XI_V01_D_C03_E02_029_Q01.png" width="80%"> |
|
Answer» |
|
| 46. |
pK_a of acetic acid is 4.76 and pk_b is NH_4 OH is 4.75. Calculate the hydrolysis constant and degree of hydrolysis of ammonium acetate. |
| Answer» Solution :`k_h = 3.2 xx 10^(-5) ,H= 5.7 xx 10^(-3)` | |
| 47. |
Pinacolone is |
|
Answer» 2, 3-Dimethyl, 2, 3-butanediol |
|
| 48. |
Picric acid is |
|
Answer» 2,4,6-Trinitrotoluene 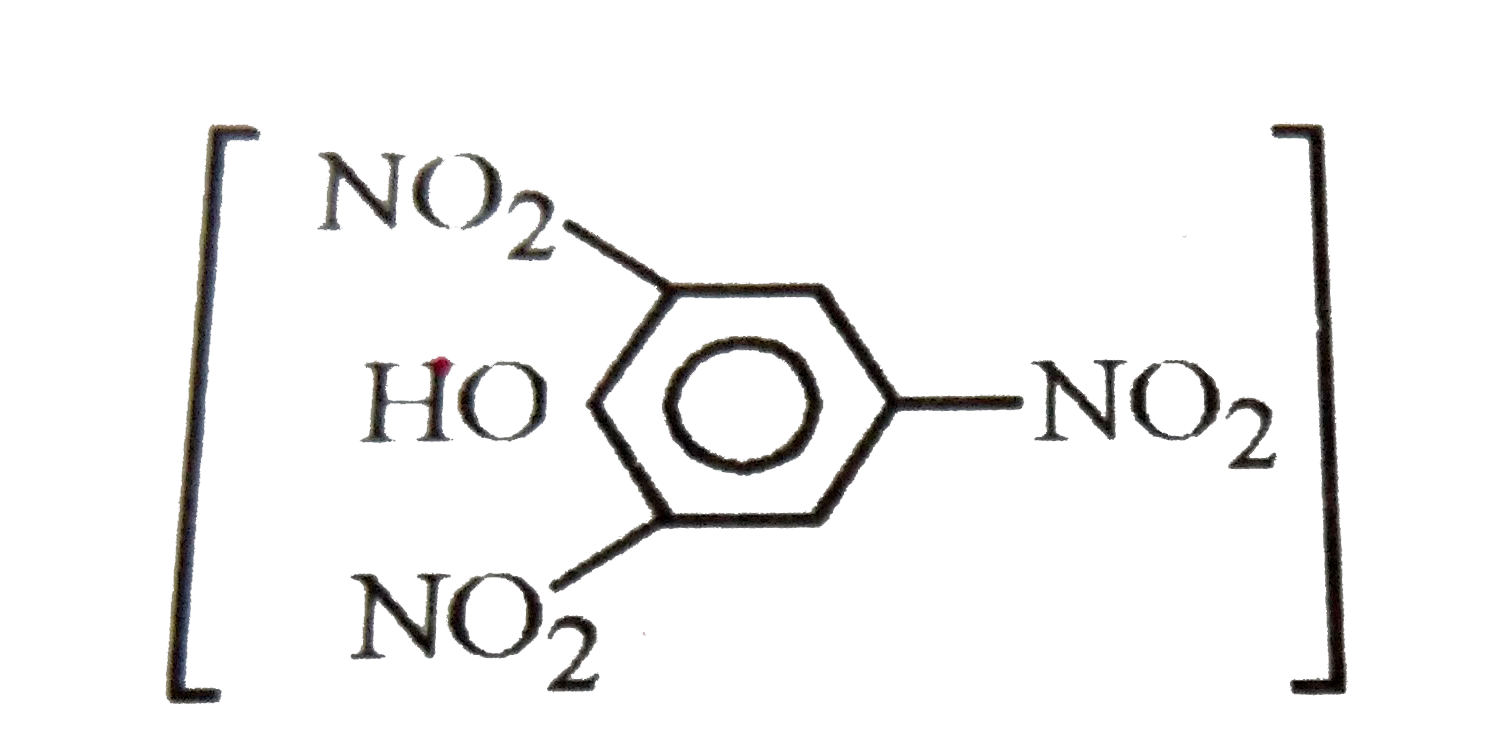
|
|
| 49. |
Pickup the correct statements |
|
Answer» Borazone a crystalline FORM of BNis even harder than diamond |
|
| 50. |
Pick up the incorrect statement(s) ? |
|
Answer» CO which is major pollutant resulting from the COMBUSTION of fuels in automobiles PLAYS a major role in photochemical smog. |
|