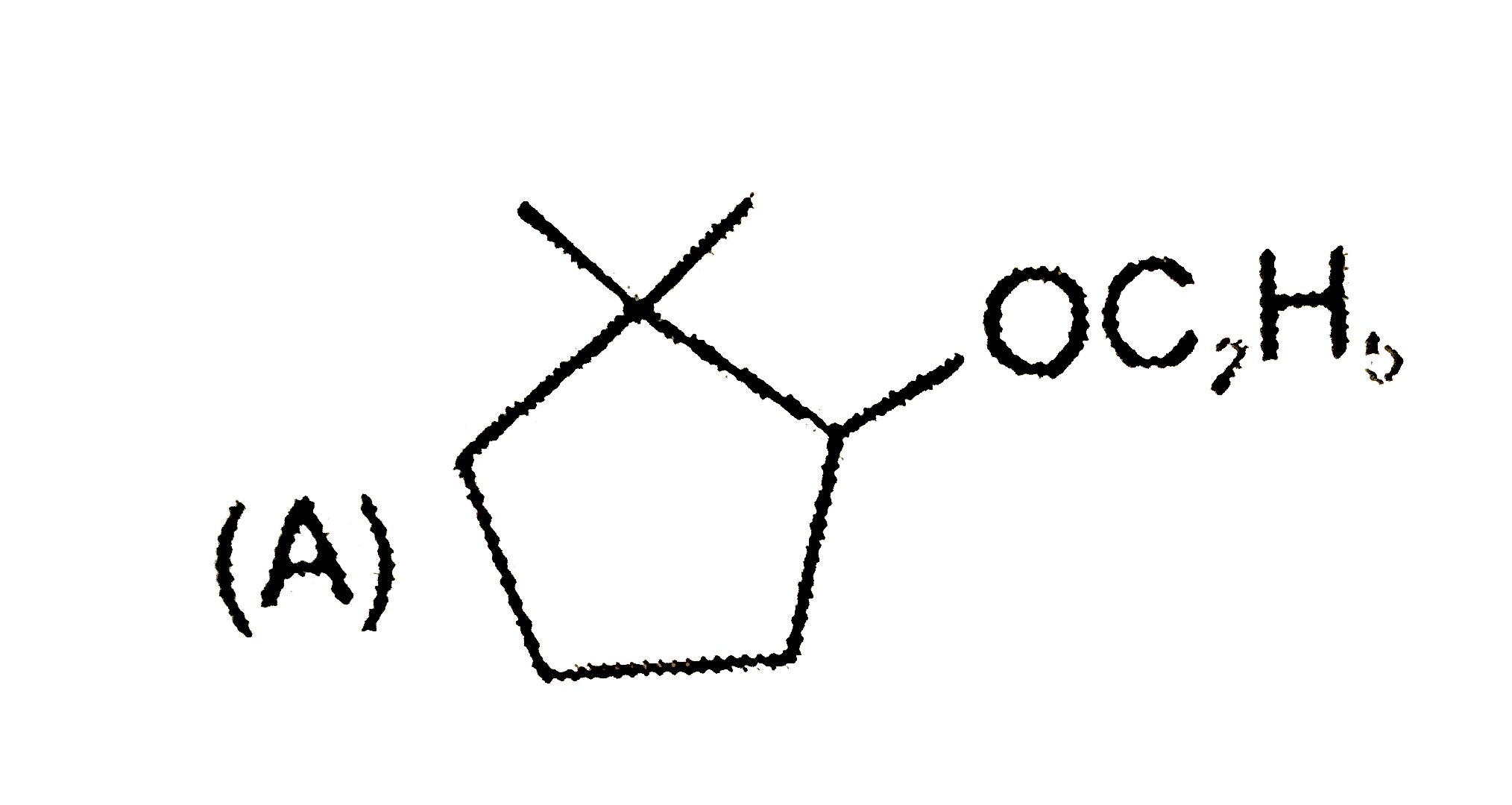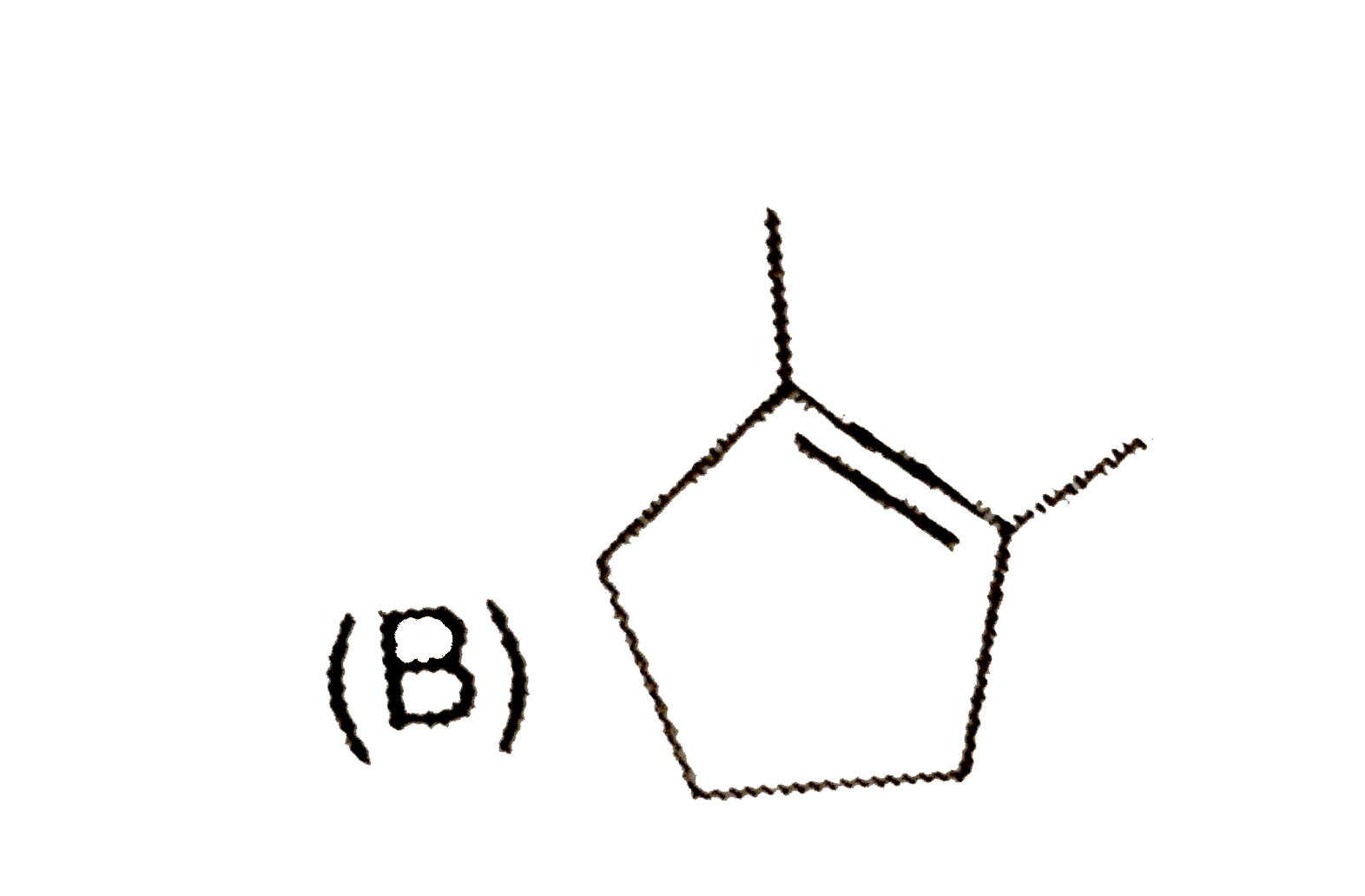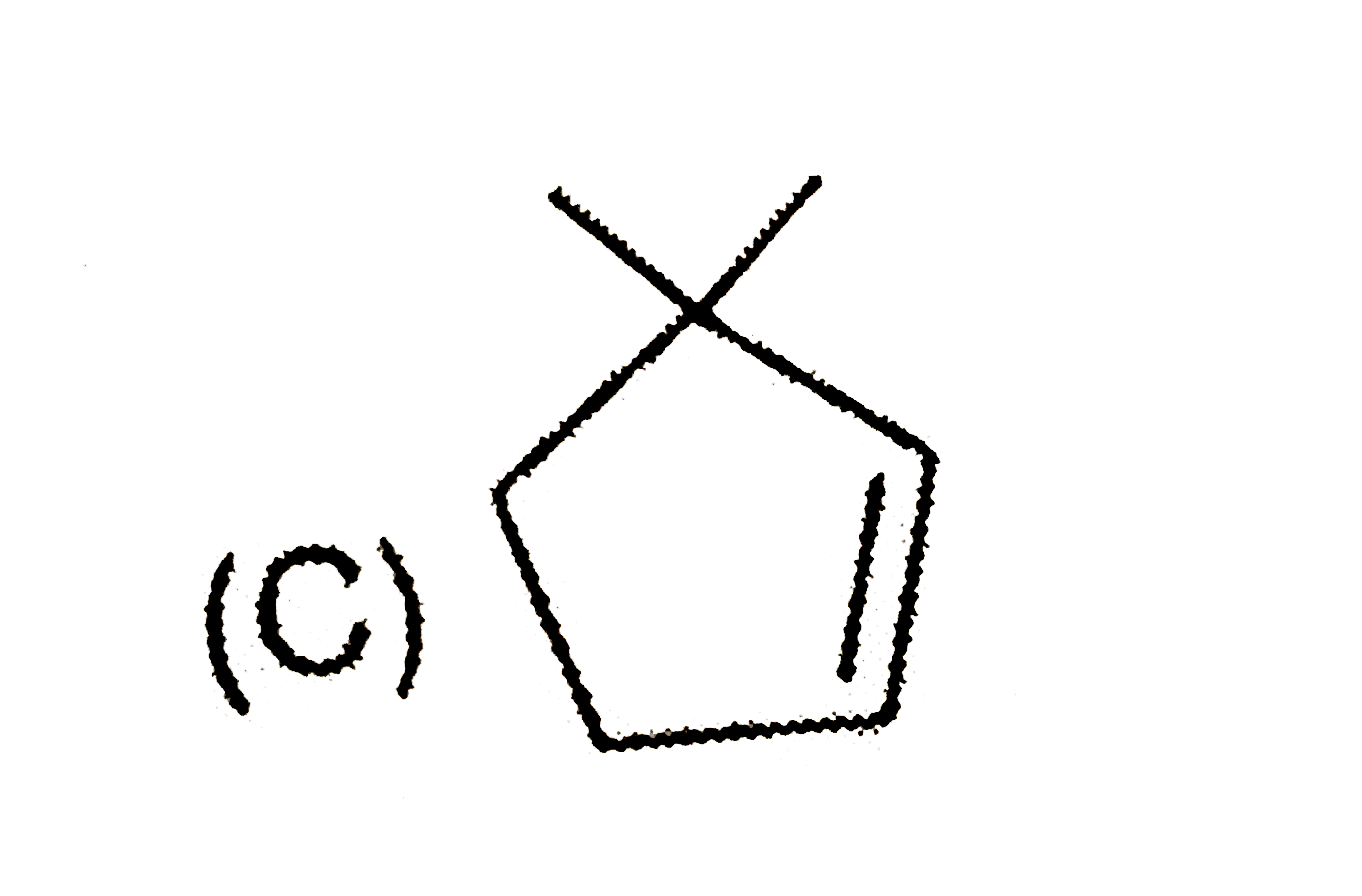Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict which of the following reaction will have appreciable concentration of reactants and products : (a)Cl_(2(g)) hArr 2Cl_((g)) , K_c=5xx10^(-39) (b) Cl_(2(g)) + 2NO_((g)) hArr 2NOCl_((g)) , K_c=3.7xx10^8 (c)Cl_(2(g)) + 2NO_(2(g)) hArr 2NO_2Cl_((g)) , K_c= 1.8 |
|
Answer» Solution :(a)`K_c=5xx10^(-39)` , the value of K is very small, so = products will REMAIN very less at the equilibrium. (B) `K_c = 3.7 XX 10^8`, the value of K is more, so theconcentration of products of reactants will be high than the concentrations of products. (c) `K_c= 1.8`, the value of `K_c`, is in-between `10^(-3)` and `10^(3)` so at the equilibrium the concentration of reactants and products are COUNTABLE. |
|
| 2. |
Predict which of the following hydrides is a gas on a solid (a) HCl (b) NaH. |
|
Answer» Solution :i. At room temperature , HCl is a colourless gas and the solution of HCl in water is called HYDROCHLORIDE acid and it is in liquid state. (II) Sodium hydride NaH is an IONIC COMPOUND and it is made of sodium CATIONS (`Na^+`) and hydride `(H^-)` anions. It has the octahedral crystal structure . It is an alkali metal hydride. |
|
| 3. |
Predict which of the following hydrides is a gas on a solid (a) HCI (b) NaH. Give your reason. |
|
Answer» Solution :At room temperature, HCI is a COLOURLESS gas and the solution of HCl in water is called hydrochloric acid and it is in LIQUID state. (ii) Sodium HYDRIDE NaH is an ionic compound and it is made of sodium cations `(Na^(+))` and hydride `(H^(-))` anions. It has the octahedral crystal STRUCTURE. It is an ALKALI metal hydride. |
|
| 4. |
Predictwhichatom ineach of thefollowingpairshas thegreater firstionizationenthalpy and explain your answer (a) Band C( b) N and O ( c) F and Ne |
|
Answer» (b)Out of N and O, nitrogen has HIGHER first ionisation enthalpy becuase in case of N the electron is to be removed from a HALF filled sub-shell as its ELECTRONIC configuration is `1s^2 2s^2 2p^3` whereas in case of oxygen theelectronic configuration is `1s^2 2s^2 2p^4` so it is easier to remove an electron in comparison to N (C ) Ne has higher ionisation enthalpy as it is a noble gas and it has electronic configuration `1s^2 2s^2 2p^6` , the outermost shell is fully filled so it gets tougher to remove an electron in comparison to F.Solution :(a) Chas higher `Delta_(i)H_1)` than Bbecauseof highernuclear a charge (b) N hashigher `Delta_(i) H_(1)` than Obeacausein case N, theelectronhas to belostfroma morestableexactly half- filledelectronicconfiguration ( c) Ne hashigher`Delta_(i) H_(1)` than Fbacause of stable inertgas configuration |
|
| 5. |
Predict whether zinc and silver react with 1 M suphuric acid to give out hydrogen or not given that h standard potentials of zinc and silver are -0.76 vold and +0.80 volt respectively |
|
Answer» Solution :(a) To predict reaction of zinc with suphuric acid in Zn reacts the following reaction shuld take place `Zn+H_(2)SO_(4)rarrZnSO_(4)+H_(2) i.e Zn+2H^(+)rarrZn^(2+)+H_(2)` By convention the cell MAY be represented as `Zn|zn^(2+)||H^(+)|H_(2)` standart EMF of the cell `E_(cell)^(@)=(H)^(+)/(H_(2))^(E^(@))(Zn^(2+)//Zn)=0-(-0.76)` Thus the EMF of tthe cell comes bout to be positive HENCE the reaction takes place (b)To Predict the reaction of silver with suplhuric acid If Ag rects the following reactionshuld take lace `2Ag+H_(2)SO_(4) RARR Ag_(2)SO_(4)+H_(2) i.e 2Ag+2H^(+) rarr2Ag^(+)+H_(2)` By convention the cell may be represented as `As|Ag^(+)||H^(+)|H_(2)` Thus the EMF of the supposed acell comes out to be negatvie hence this reaction does not take place agent than `H_(2)` and hence can reduce `H^(+)` IONS to evolve `H_(2)` GAS in contrast since `E_(H^(+)//GH_(2)^(@)` =0.0 V therefore Zn is a stronger reducigng that of `E_(H^(+)//H_(2))^(@)=0.0V`therefore Ag is a weaker reducing agent than `H_(2)` and hence cannot reduce `H^(+)` ions ot evolve `H_(2)` gas |
|
| 6. |
Predict which atom in each of the following Period has the highest first ionization energy. |
| Answer» SOLUTION :(a) C, (B) N, (c) NE. | |
| 7. |
Predict whether following reaction will be exothermic or endothermic . Give reaction for your answer H-H(g)+Cl-Cl(g) rarr 2H-Cl(g) |
| Answer» SOLUTION :This REACTION isis accompanied by DECREASE of randomness ,i.e., this factoropposes the process. For the reaction to be spontaneous , the energy FACTOR must favour, i.e., the reaction should be exothermic. | |
| 8. |
Predict whether a precipitate will be formedor not on mixing 20 ml of 0.001 N NaCl solution with 80 ml of 0.01 N AgNO_(3) solution. K_(sp) for AgCl=1.5xx10^(-10). |
|
Answer» 20 ml of `10^(-3) ` M NACL sol `-=` 100 ml of ? M Molarity of NaCl solution in the final solution `=(20xx10^(-3))/(100) = 2xx10^(-4) M or [Cl^(-) ] = 2 xx 10^(-4) M` 80 ml of `10^(-2)` M `AgNO_(3)` sol `-=` 100ml of ? M Molarity of `AgNO_(3)` solution in the final solution `= (80xx10^(-2))/(100) = 8xx10^(-3)` M or `[AG^(+)]=8xx10^(-3)M` `:. ` IONIC product `=[Ag^(+)][Cl^(-)]=(8xx10^(-3))(2xx10^(-4))=16xx10^(-7) = 1.6 xx 10^(-6)` which is greater than the solubility product. Hence, ppt. will be FORMED. |
|
| 9. |
Predict whether a precipitate will be formed or not on mixing 20 mL of 20 mL " of " 0.001 NNaClsolution with 80 mL of 0.01MAgNO_(3)solution. K_(sp) " for " AgCl " is " 1.5 xx 10^(-10). |
| Answer» SOLUTION : YES, PPT will FORMED | |
| 10. |
Predict whether a precipitate of PbI_2 will be formed or not on mixing 20 mL of 3 xx 10^(-3) M Pb(NO_3)_2 solution with 80 mL of 2 xx 10^(-3)M Nal solution. K_(sp) for lead iodide (PBI_2) is 6.0xx10^(-9) . |
| Answer» Solution :`Q_(sp) 1.536xx10^(-9) LT K_(sp)` , No PRECIPITATION | |
| 11. |
Predict the structure of MgO crystal and coordination number of its cation in which cation and anion radii are equal to 65 pm and 140 pm respectively |
|
Answer» |
|
| 12. |
Predict the structure of MgO crystal and coordination number of its cation in which cation and anion radii are equal to 65 pm and 140 om respectively. |
|
Answer» |
|
| 13. |
The state of hybridisation of the central atom of which of the following is sp^3 d^2? |
Answer» SOLUTION :
|
|
| 14. |
Predict the spontaneity of a forward (or) a reverse reaction based on DeltaG of a reversible reaction. |
|
Answer» Solution :The value of `K_(c)` is directly related to the Gibbs free energy change `(DeltaG)` of the reaction. 1. If `DeltaG` = -ve, then the reaction is spontaneous, and proceeds in the forward reaction. 2. If `DeltaG` = +ve, then the reaction is non-spontaneous in the forward direction. The REVERSE reaction will be spontaneous, and thus proceed towards the reactants' SIDE. 3. If `DeltaG` = 0, then the reaction is at equilibrium. No net change takes place. |
|
| 15. |
Predict the sign of entropy change in each of the following: Temperature of perfectly crystalline solid is raised from 0 K to 115 K. |
| Answer» SOLUTION :POSITIVE | |
| 16. |
Predict the sign of entropy change in the following reaction : (i) O_(2)(g) +2SO_(2)(g) rarr2SO_(3)(g) (ii)CaC_(2)O_(4)(s) rarr CaCO_(3)(s)+CO(g) (iii) 2H_(2)(g) +O_(2)(g) rarr 2H_(2)O(g) |
| Answer» Solution :(i) `DELTAS=- IVE` ( because number of decreases ) (II) `DeltaS= + ive`( because reactant is SOLID while one of the productis GASEOUS ) (iii) `DeltaS = -ive` ( because number of molesdecreases ) | |
| 17. |
Predict the sign of q, w ,DeltaU and DeltaH for given processes. Hence, find number process for which at least one of q,omega.DeltaU,DeltaH are zero |
|
Answer» |
|
| 18. |
Predict the sign of q, w andDelta U for given process. Hence , find number of process for which at least one of q,w, DeltaU,DeltaHare zero. |
|
Answer» |
|
| 19. |
Predict the sign of entropy change in each of the following : (i) H_(2) (at 298 K, 1 atm ) rarr H_(2) at 298 K, 10 atm) (ii) H_(2)O(at 298K, 1 atm )rarr H_(2)O ( at 330K, 1atm) (iii) 2NH_(4)NO_(3)(s) rarr 2N_(2)(g)+4H_(2)O(g) +O_(2)(g) (iv)Crystallization of copper sulphatefrom its saturated solution. (v) 2SO_(2)(g) +O_(2)(g) hArr 2SO_(3)(g) |
|
Answer» Solution :Entropy isa measure of randomness or disorder of a system. If randomness increases, entropy increases. (i) GAS at lower pressure has greater randomness than at high pressure ( compressed gas) at the sametemperature. Hence, entropy decreases, i.e., `DELTA S ` is -ve. (ii) Molecules at highertemperaturehave greater randomness at the same pressure . Hence, `Delta S` is `+` ve . (iii) GASEOUS moleculeshave greater randomness than the solid . Hence, `Delta S` is `+ ` ve. (iv) `CuSO_(4)` crystals have ordered ARRANGEMENT whereas ionsin the soluiton have greater randomness. Hence, `Delta S` is `-` ve. (v) There are 3 MOLES of two different gaseous reactants which have greater randomness than 2 moles of the only gaseous product, `SO_(3)` . Hence, randomness decrease, i.e., `Delta S`is `-` ve. |
|
| 20. |
Predict the sign of entropy change in each of the following:AgNO_(3_((g))) rarr AgNO_(3_((aq))) |
| Answer» SOLUTION :POSITIVE | |
| 21. |
Predict the sign of entropy change in each of the following: C (graphite) rarr C (Diamond) |
| Answer» SOLUTION :NEGATIVE | |
| 22. |
Predict the sign of entropy change in each of the following: A liquid crystallises into solid. |
| Answer» SOLUTION :NEGATIVE | |
| 23. |
Predict the sign of DeltaS (positive or negative) for the following changes : (i) HCOOH(l) to H_(2)O(l) + CO_(2)(g) (ii) NH_(3)(g) + HCI(g) to NH_(4)CI(s) (iii) I_(2)(s) to I_(2)(g) (iv) 2O_(3)(g) to 3O_(2)(g) (v) 2H_(2)(g) + O_(2)(g) to 2 H_(2)O(l). |
| Answer» SOLUTION :(i) +ve (II) `-ve` (III)`+ve`(IV)`+ve` (V)`-ve` | |
| 24. |
Predict the sign of DeltaS^(@) for the following reaction : 2H_(2)S (g) + 3O_(2)(g) rarr 2H_(2)O(g) + 2SO_(2)(g) |
|
Answer» <P>`+ve` |
|
| 25. |
Predict the shapes of the following molecules on the basis of hybridization. BCl_(3), CH_(4) ,CO_(2), NH_(3) |
Answer» Solution :In `BCl_(3) `, the GEOMETRY is trigonal PLANAR is due to `sp^(2)` HYBRIDIZATION. `CO_(2)` show linear shape because of sphybridization. The geometry of `NH_(3)` is pyramidal shape andhas `sp^(3)` hybridization 
|
|
| 26. |
Predict the shapes of the following species andthe type of hybrid orbitals on the central atom. a. PbCl_(4) b. SbF_(6)^(-) and c PCl_(3) |
|
Answer» <P> SOLUTION :a. Tetrahedral, HYBRIDISATION of Pb is `sp^(3)`B. Octahedral, HYBRISATION of Sb is `sp^(3)d^(2)`. c. Pyramidal, hybridisation of P is `sp^(3)`. |
|
| 27. |
Predict the shapes and hybridisation of the following molecules CO_(2). |
|
Answer» SOLUTION :Total valence ELECTRONS ` = 4 + 6 xx 2 = 16` ` 16/8 = 2 (Q_(1)) + O (R_(1))` ` :. X = 2` `:. ` Hybridisation in `CO_(2)` is sp. Hence it is Linear in shape. |
|
| 28. |
Predict the shape of ClF_(3) and NH_(3) using VSEPR theory. |
Answer» SOLUTION :`ClF_(3)` consists of 3 bond PAIRS of electrons and 2 lone pairs of electrons, hence its structure must be REPRESENTED as  `NH_(3)` consists of 3 bond pairs of electrons and 1 lone pair of electrons, hence its structure must be represented as 
|
|
| 29. |
Predict the relative energies of the following resonating structures . |
|
Answer» Solution :Structures (A) FOLLOWS all the GUIDELINES as per points (1) to (4) above . Structure (B) violates point (2) because it has TWO formal charges . Structure (C ) violates points (2) and (3) because it has two formal charges and carries a negative charge on carbon atom . RESPECTIVELY . |
|
| 30. |
Predict the products when bromoethane is treated with the following: (i) KNO_2 (ii) AgNO_2 |
|
Answer» SOLUTION :(i) When BROMOETHANE is treated with `KNO_2`, the PRODUCT formed is Ethyl nitrite. `underset("Bromoethane")(CH_3CH_2Br) + underset("Nitrite")underset("Potassium")(KNO_2) to underset("Ethyl nitrite")(CH_3 - CH_2) - O - N = O+ KBR` (ii) When bromoethane is treated with `AgNO_2`,NITROETHANE will be formed as the product. `underset("Bromoethane")(CH_3CH_2Br) + underset("Silver nitrite")(AgNO_2) to underset("Nitroethane")(CH_3CH_2NO_2) + AgBr` |
|
| 31. |
Predict the products of electrolysis in each of the following: (i) An aqueous solution of AgNO_(3) with silver electrodes. (ii) An aqueous solution AgNO_(3) with platinum electrodes. (iii) A dilute solution of H_(2)SO_(4) with platinum electrodes. (iv) An aqueous solution of CuCl_(2) with platinum electrodes. |
|
Answer» Solution :(i) Aqueous solution of `AgNO_(3),Ag_((aq))^(+)andNO_(3(aq))^(-)` ions are formed. `AgNO_(3(aq))toAg_((aq))^(+)+NO_(3(aq))^(-)` During electrolysis `Ag^(+)orH_(2)O` gets reduced at cathode. But reduction potential of `Ag^(+)` is more than that of `H_(2)O`. `Ag_((aq))^(+)+e^(-)toAg_((s))""E^(@)=+0.80"VOLT"` `2H_(2)O_((l))+2e^(-)toH_(2)+2OH_((aq))^(-)" "E^(@)=-0.83"volt"` Therefore, `Ag^(+)` gets reduced at cathode. As same, Ag metal or `H_(2)O` atoms oxidises at anode. But oxidation potential of Ag is more. than `H_(2)O`, therefore Ag gets oxidised at anode. `Ag_((s))toAg_((aq))^(+)+e^(-),""E^(@)=-0.80"volt"` `2H_(2)O_((l))toO_(2(g))+4H_((aq))^(+)+4e^(-)," "E^(@)=-1.23"volt"` (ii) Pt do not gets oxidized therefore at anode water gets reduced and gathered at cathode. (iii) Aqueous solution of `H_(2)SO_(4)` on electrolysis `H^(+)andSO_(4)^(-2)` ions are formed. `H_(2)SO_(4(aq))to2H_((aq))^(+)+SO_(4(aq))^(-2)` During electrolysis `H^(+)orH_(2)O` gets reduced at cathode. But reduction potential of `H^(+)` is more then `H_(2)O`, therefore `H^(+)` is reduced at cathode and `H_(2)` gas is liberated. `2H_((aq))^(+)+2e^(-)toH_(2(g))""E^(@)=-0.0"volt"` `2H_(2)O_((aq))+2e^(-)toH_(2)+2OH_((aq))^(-)" "E^(@)=-0.63"volt"` At anode, `SO_(4)^(-2)orH_(2)O` gets oxidized but for the oxidation of `SO_(4)^(-2)`, more energy is used for the breaking the bond than `H_(2)O`. Therefore, oxidation potential of `SO_(4)^(-2)` is less than `H_(2)O`. At anode, their is oxidation of `H_(2)O` and oxygen gas is released. (IV) Aqueous solution of `CuCl_(2)` gives `Cu^(+2)andCl^(-)` ions. `CuCl_(2(aq))toCu_((aq))^(+2)+2Cl_((aq))^(-)` During electrolysis, `Cu^(+2)` ion of `H_(2)O` is reduced but electrode potential of `Cu^(+2)` is more than that of `H_(2)O`, therefore `Cu^(+2)` is reduced and gathered at cathode. `Cu_((aq))^(+2)+2e^(-2)toCu_((aq))""E^(@)=+0.34"volt"` `H_(2)O_((l))+2e^(-)toH_(2)+2OH^(-)" "E^(@)=-0.83"volt"` Some as, at anode `CL^(-)orH_(2)O` is oxidised. But oxidation potential of `H_(2)O` is more than `Cl^(-)`. `2Cl_((aq))^(-)toCl_(2(g))+2e^(-)""E^(@)=-1.36"volt"` `2H_(2)O_((l))toO_(2(g))+4H_((aq))^(+)+4e^(-)" "E^(@)=-1.23"volt"` For the oxidation of `H_(2)O,O_(2)` gas is released when their is more potential is applied. Therefore, `Cl^(-)` is oxidised at anode and `Cl_(2)` gas is evolved. |
|
| 32. |
Predict the products of electrolysis each of the following : (i) An aqueous solution of AgNO_3 with silver electrodes (ii)An aqueous solution of AgNO_3 with platinum electrodes (iii) An aqueous solution of H_2SO_4 with platinumelectrodes (ii)An aqueous solution of CuCl_2 with platinum electrodes |
|
Answer» Solution :(i) Electrolysis of aqueous solution of `AgNO_3` using SILVER electrodes : `AgNO_3(s)+nH_2O rarr AG^(+)(aq) +NO_3^(-) (aq)` `""H_2O hArr H^(+) +OH^(-)` At cathode : `Ag^+` ions have lower discharge potential than `H^+` ions . Hence `Ag^+` ions will be deposited as silver (in preference to `H^+` ions ) At anode : Since silver electrode is attacked by `NO_3^(-)`ions, Ag anode will dissolve to form `Ag^+` ions in the solution . `Ag rarr Ag^(+) +e^(-)` (II) Electrolysis of aqueous solution of `AgNO_3` using platinum electrodes : (ii) At cathode : same as above At anode : Since silver is not attackable , out of `OH^- and NO_3^(-)` ions, `OH^-` ions have lower discharge potential and hence `OH^-` ions will be discharged in preference to `NO_3^-` . The `OH^-` will DECOMPOSE to give `O_2`. `OH^(-)(aq) rarr OH+e^(-)` `4OH^(-) (aq) rarr 2H_2O(l) +O_2(g)` (iii) Electrolysis of `H_2SO_4` with Pt electrodes : `H_2SO_4(aq) rarr 2H^(+) (aq) +SO_4^(2-) (aq)` `H_2OhArr H^(+) +OH^(-)` At cathode : `H^(+) + e^(-) rarr H` `H+H rarr H_2(g)` At anode : `OH^(-) rarr OH+e^(-)` `4OH rarr 2H_2O+O_2(g)` (iv) Electrolysis of aqueous solution of `CuCl_2` with platinum electrodes : `CuCl_2(s) +(aq) rarr Cu^(2+)(aq) +2Cl^(-) (aq)` `H_2O hArr H^(+) +OH^(-)` At cathode : `Cu^(+)` will be reduced in preference to `H^+` is `Cu^(2+)+2e^(-) rarr Cu` At anode : `Cl^(-)` ions will be oxidised in preference to `OH^-` ions `Cl^(-) rarr Cl+e^(-)` `Cl+Cl rarr Cl_2` Thus copper will be deposited on the cathode and `Cl_2` will be liberated at anode . |
|
| 33. |
Predict the products of electrolysis in each of the following (i) an aqueous solution of AgNO_(3) with silver electrodes. (ii). An aqueous solution of AgNO_(3) with platinum electrodes. (iii). A dilute solution of H_(2)SO_(4) with platinum electrodes. (iv). An aqueous solution of CuCl_(2) with platinum electrodes. |
| Answer» SOLUTION :`H_(2)` at CATHODE, `O_(2)` at ANODE. | |
| 34. |
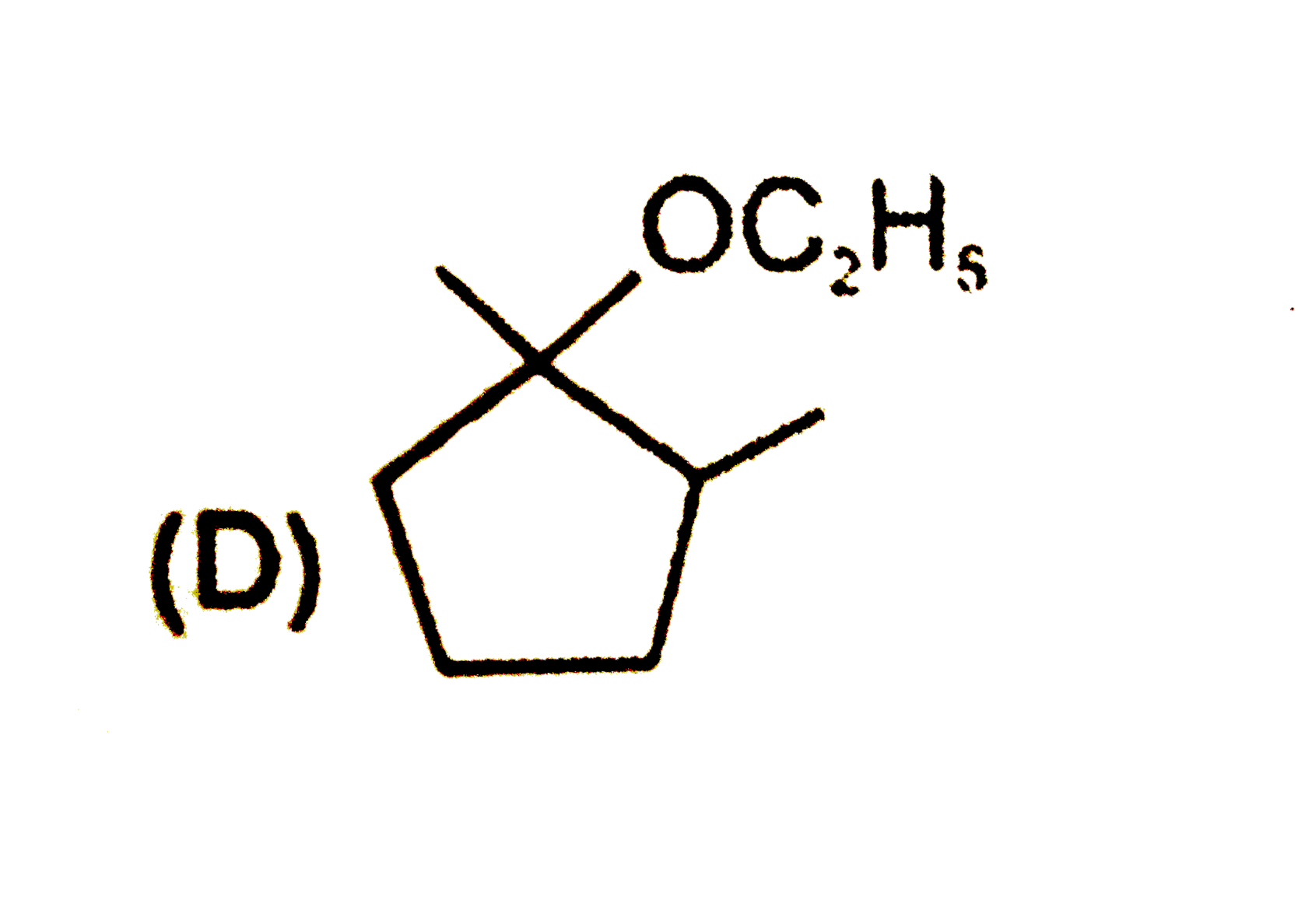
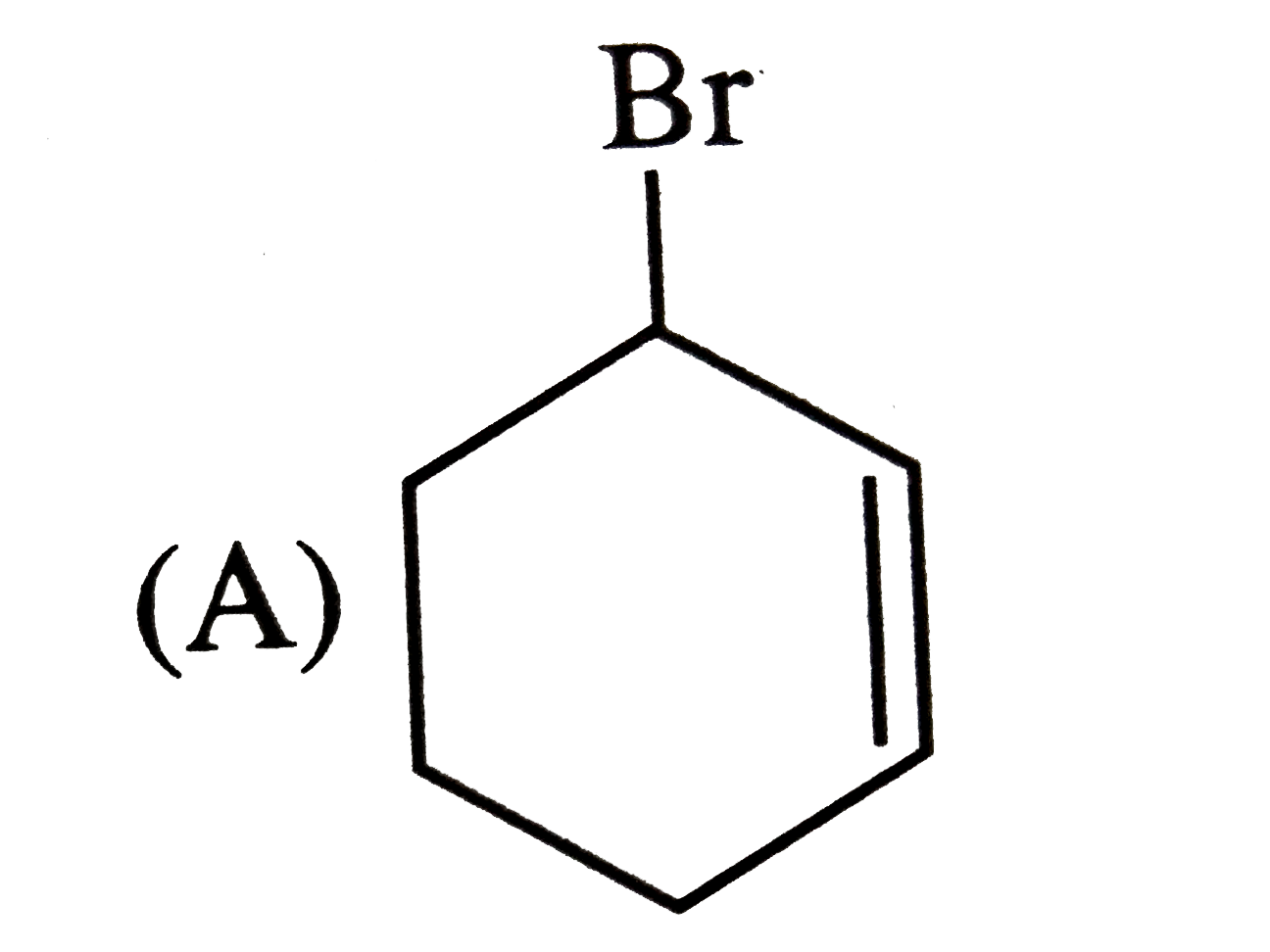
Predict the products expected in the given reaction 2-Bromo-1,1-dimethylcycopentaneunderset(Delta)overset(C_2H_5OH)to |
|
Answer»
|
|
| 35. |
Predict the product (s) and outling the mechanism for the process when benzene is treated with, (i) (CH_(3))_(3)C CI"in presence of "AICI_(3) (ii) (CH_(3))_(2)ChArrCH_(2)"in presence of "H_(2)SO_(4) (iii) (CH_(3))_(2)CHCH_(2)"in presence of "AICI_(3) (iv) (CH_(3))_(3)COH"in presence of "H_(2)SO_(4). |
Answer» Solution :In all the four cases, the same PRODUCT t-butylbenzene is FORMED byelectrophilic substitution, i.e., same electrophile `(CH_(3))_(3)C^(+)` attacks BENZENE in each case. 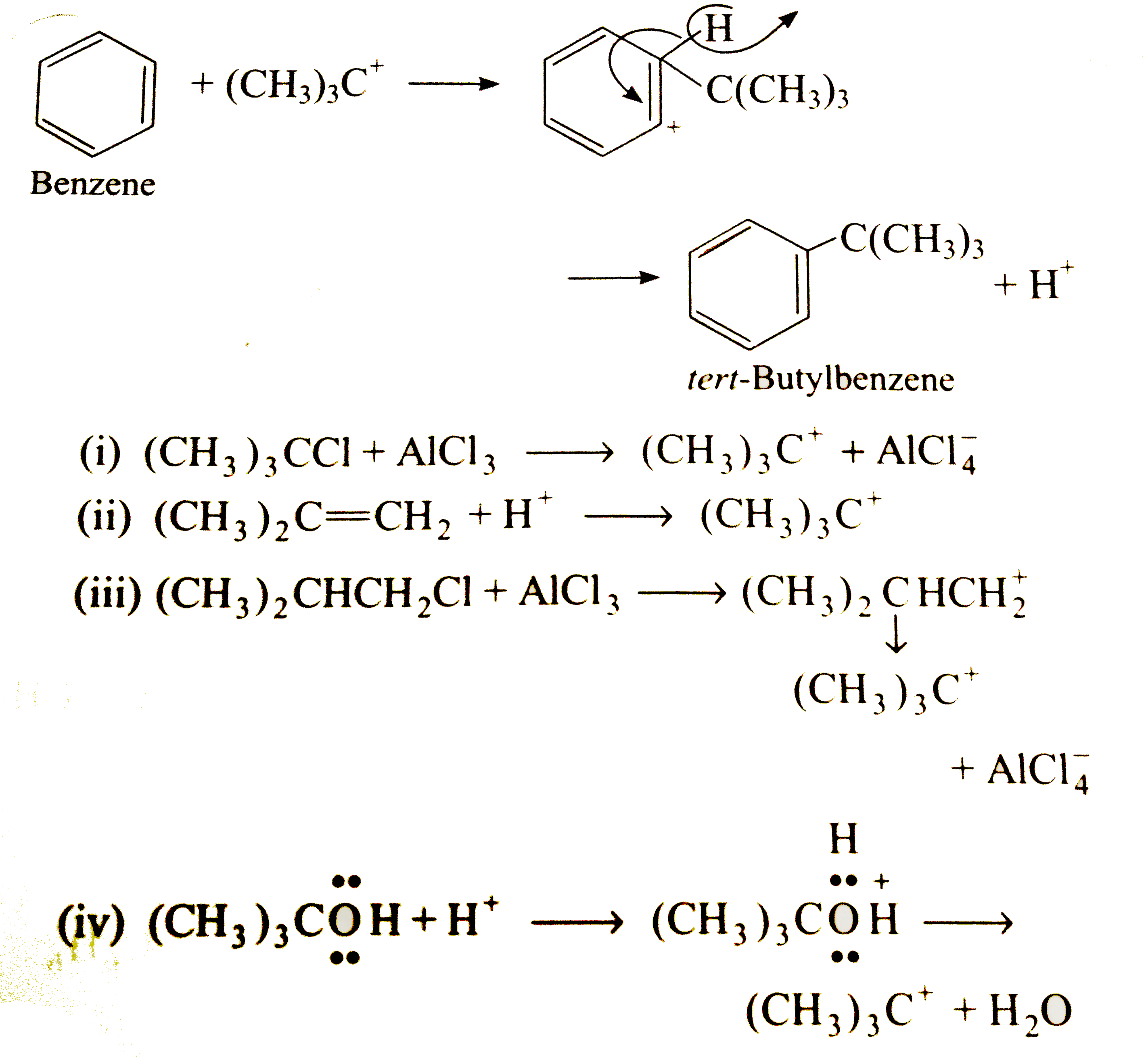
|
|
| 36. |
Predict the product of the reaction C_(6)H_(5)OH underset(NaOH)overset(C Cl_(4))rarr |
|
Answer» o-Hydroxybenzaldehyde 
|
|
| 37. |
Predict the product of the following reaction |
|
Answer» |
|
| 38. |
Predict the product of electolysis in each of the following (i) An aquesous solution of AgNO_(3) with silver electrodes (ii) An Aqueous solution fo silver nitrate with platinum elctrodes (iii) A dilute soution of H_(2)SO_(4) with platinum electrodes (iv) An aqueoius solution of CuCI_(2) with platinum electordes |
|
Answer» Solution :(i) In aqueous solution `AgNO_(3)` ionises ot give `Ag^(+)(Aq)and NO_(3)^(-)(aq)`ions `AgNO_(3)(aq)rarrAg^(+)(aq)+NO_(3_^(-) (aq)` thus when electricity is passed `Ag^(+) (aq)+e^(-)rarrAg(s), E^(@)=+_0.80 v` `2H_(2)O(l)+2e^(-)rarrH_(2)(g)+2O^(-)(aq),e^(@)=-0.83 v` since the elctrode potential i.e reductin potential of `Af^(+)` (aq) ions is higher than that of `H_(2)O` molecules therefore at the CATHODE it is the `Ag^(+)` (aq) ions (RATHER than `H_(2)O` molecules ) which are reduced similary at the naode either ag metal of the anode ofr `H_(2)O` molecules may be oxidised their electrode thus when an queous soluton of `AgNO_(3)` is electrolysed As from As anode dissolved while `Ag^(+)` (aq) INS present in the solution get reduced and get depostied on the cathode (iii) if howver elctrolysis of `AgNO_(3)`is electrolysed Ag from Ag anode dissolves wile `Ag^(+)` ions present in the solutoin get reduced and get depostited on the cathode since the elctron potential of `H^(+)` (aq) ions is higher than that of `H_(2)O` therefore at the cathode it is `H^(+)` (aq) ioons which are reduced to evolve `H_(2)O` gasof `SO_(4)^(2-)` is expected to be much lower than that of `H_(2)O` molecuels therefore at the anode it is `H_(2)O` mleculeswhich are oxidised to evolve `O_(2)` gas from the abvoe discussion it followthat during electrolysis of an aqueous solution of `H_(2)O` only the electrolysis of `H_(2)O` occurs liberating `H_(2)` at the cathode and `O_(2)` at the anode thus as cathode either `Cu^(2+)`(aq) or `H_(2)O` molecuelsare reduced their electrode potential are `Cu^(2+)(aq)+2e^(-)rarrCu(s) , =+ 0.34 V` `H_(2)O(l)+2e^(-)rarrH_(2)(g)+2OH^(-), E^(@)=-0.83V` since the electrode potential of `Cu^(2+)`(aq) ionsis much higher than that of `H_(2)O` therefore at the cathode it is `Cu^(2+)` (aq) ions whihc are reduced and not 1 `H_(2)O` molecules similary at the anode either `CI^(-)` (aQ) ions or `H_(32)O` molecules are oxidised their oxidation potentialsare `2CI^(-)(aq)rarrCI_(2)(g)+2e^(-), E^(@)=-1.36 v` `2H_(2)O(l)rarrO_(2)+4H^(+)(aq)+4e^(-),E^(@)=-1.23 v` Although oxidation potential of `H_(2)O` molecules is higher than of `CI^(-)` ions neverthelss oxidation of `CI^(-)(aq)` ions occurs is prefrence to `H_(2)O` since due to `"overvoltage"^(*)` of `O_(2)` much lower potential than than -1.36 v is NEEDED for the oxidation of `H_(2)O` molecules thus when aqueous solution of `CuCI_(2)` is electrolysed Cu metal is liberated at the cathode while `CI_(2)` gas is evolved at the anode |
|
| 39. |
Predict the product of following reaction Find the value of (x+y). |
Answer» 
|
|
| 41. |
Predict the product formed when cyclo-hexene is treated with N-bromosuccininide. |
Answer» Solution :ALLYLIC substitution takes place. Carbon, ADJACENT to DOUBLE bonded carbon is called allylic carbon. Hydrogen on allylic carbon is substituted by BROMINE. 
|
|
| 42. |
Predict the product for the following . (i) Benzene +H_(2) to ? (ii) Benzene +Cl_(2) to ? (iii) Benzene _O_(2) ? (iv) Benzene +CH_(3)Cl to ? (v) C_(6)H_(5)COONa+NaOHto? |
Answer» Solution :`(i)` `(ii)`  `(iii)`  `(iv)` `underset("Benzene")(C_(6)H_(6))+underset("Chloromethane")(CH_(3)Cl)overset("anhydrous"AlCl_(3))(to)underset("Toluene")(C_(6)H_(5)CH_(3))+HCl` `(V)` `underset("Sodium BENZOATE")(C_(6)H_(5)COONa)+NaOHoverset(CaO)(to)underset("Benzene")(C_(6)H_(6))+Na_(2)CO_(3)` |
|
| 43. |
Predict the product C obtained in the following reaction of butyne - I CH_3 CH_2 - C -= CH + HCl to B overset(HI)to C |
|
Answer» `CH_3CH_2 - OVERSET(I)overset(|)underset(CL)underset(|)C-CH_3` |
|
| 44. |
Predict the product C obtained in the following reaction of butyne-1 CH_3CH_2-C=CH+HCl to B overset"HI"to C |
|
Answer» `CH_3CH_2-undersetunderset(Cl)|oversetoversetI|C-CH_3` |
|
| 46. |
Predict the possible number of alkenes and the main alkene in the following reaction . |
|
Answer»
|
|
| 47. |
Predict the position of the element in periodic table satisfying the electron configuration (n-1)d^(2), ns^(2) where n=5 |
|
Answer» SOLUTION :ELECTRONIC Configuration :(n-1)`d^(2)ns^(2)` For n=5, the electronic configuration is, `1s^22s^2 2p^6 3s^2 3p^64s^2 3d^10 4p^6 4d^2 5s^2` ATOMIC number:40 4 group 5 period (d BLOCK ELEMENT)=Zirconium |
|
| 48. |
Predict the order of reactivity of the following compounds in dehydrohalogenation: (a) CH_(3)CH_(2)CH_(2)CH_(2)Cl, (CH_(3))_(2)CHCH_(2)Cl, (CH_(3))_(2)CH-CH_(2)Br, CH_(3)CH(Br)CH_(2)CH_(3), (CH_(3))_(3)C-Br (b) CH_(3)CH(Br)CH_(3), CH_(3)CH_(2)CH_(2)Br, (CH_(3))_(2)CHCH_(2)Br, (CH_(3))_(2)C CH_(2)Br |
|
Answer» Solution :(a) `(CH_(3))_(3)CBr gt CH_(3)underset(Br)underset(|)CH CH_(2)CH_(3) gt (CH_(3))_(2)CHCH_(2)Br gt (CH_(3))_(2)CHCH_(2)CL gt CH_(3)CH_(2)CH_(2)CH_(2)Cl` (B) `CH_(3)CH(Br)CH_(3) gt (CH_(3))_(3)C CH_(2)Br gt (CH_(3))_(2)CHCH_(2)Br gt CH_(3)CH_(2)CH_(2)Br ` |
|
| 49. |
Predict the nature of the M-effect when 'Cl' and 'NO_(2)' groups are attached to the benzene ring. Write the different resonating structures. |
Answer» SOLUTION :In chlorobenzene, the CHLORINE ATOM has `+M` EFFECT, (electron releasing effect) 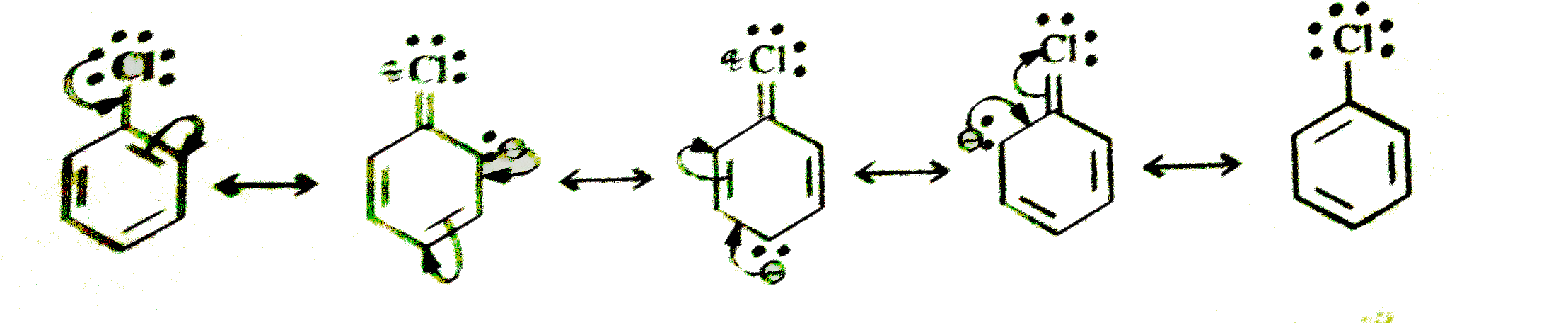 In nitrobenzene, `-NO_(2)` group has `-M` effect, (electron withdrawing effect) 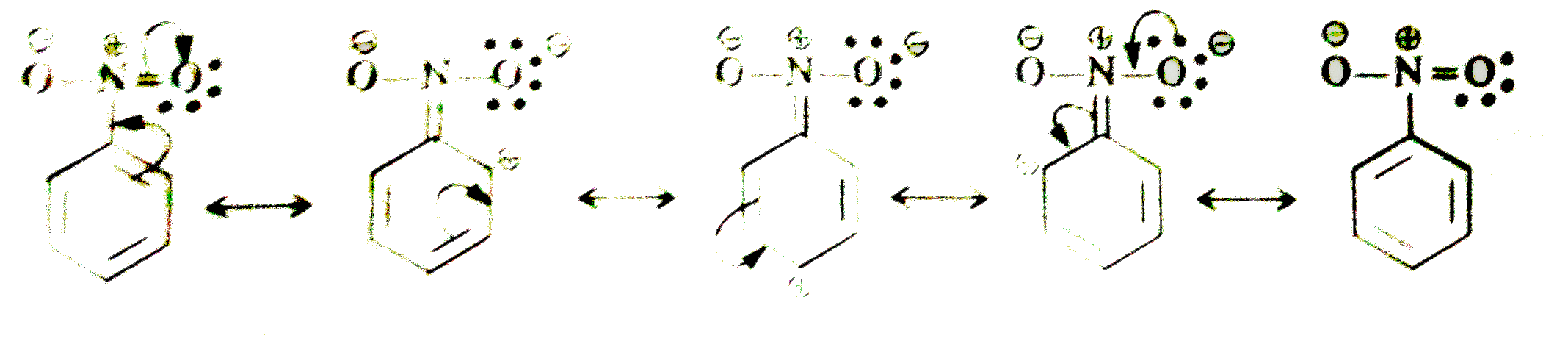
|
|
| 50. |
Predict the major product(s) of the following reactions, and explain their formation. H_(3)C-CH=CH_(2)underset(HBr)overset((Ph-CO-O)_(2))rarr H_(3)C-CH=CH_(2)overset(HBr)rarr |
Answer» Solution :Addition of HBr to PROPENE GIVES 2-Bromopropane. <BR>  (i) In presence of organic PEROXIDE the major product is 1-Bromopropane. `underset("Benzoyl peroxide")(C_(6)H_(5))-overset(O)overset(||)(C)-O-O-overset(O)overset(||)(C)-C_(6)H_(5) overset("Homolysis")rarr 2C_(6)H_(5)-overset(O)overset(||)(C)--2C_(6)H_(5)+2CO_(2)` (iii) `C_(6)H_(5)+H-Broverset("Homolysis")rarr C_(6)H_(5)+Br` (iii)  (iv) `underset(("Major product"))(CH_(3)-CH-CH_(2)Br)+ Hr+H-Br overset("Homolysis")rarr CH_(3)-CH_(2)-CH_(2)+Br+Br` |
|