Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
s-p overlap is defined as the overlapping between __________. |
|
Answer» half filled s-orbital of ONE ATOM and completely electrons with opposite SPINS |
|
| 2. |
s-orbitals have no direction. Comment. |
|
Answer» SOLUTION :For s-orbital, `Psi=f(R)` This solution has only DISTANCE (r), but has no DIRECTION. |
|
| 3. |
S - orbital does not show any preference for direction. Why? |
| Answer» SOLUTION :S - ORBITAL does not show any PREFERENCE for direction DUE to symmetric shape | |
| 4. |
S(+) Mono Sodium Glutamate (MSG) is a flavour enhancer used in many foods. Fast foods often contain substantial amount of MSG and is widely used in Chines food. If one mole of above MSG was placed in 845 ml solution and passed through 200 mm tube, the observed rotation was found to be +9.6^(@). If 33.8g of (+) MSG was put in 338 ml solutionand was mixed with 16.9 g of (-) MSG put in 169 ml solution and the final solution was passed through 400 mm tube. find out observed rotation of these final solution. |
|
Answer» `+1.6^(@)` (-) MSG `rArr` 16.9 gm in 169 ml OPTICAL purity in mixture (+) MSG =16.9 gm in 507 ml solution `C=16.9 /507 gm//ml "" l=4 dm` `theta_("Observed")=[theta^(@)]xxC.l. "" =24xx16.9/507 xx4=+3.2^(@)]` |
|
| 5. |
S^@ of N_2, H_2 and NH_3 respectively are 190, 130& 111 J/K/mol, then DeltaS_("sys") for formation of 8.5 g NH_3 is [in J/K] |
|
Answer» `-179` |
|
| 6. |
S(+) Mono Sodium Glutamate (MSG) is a flavour enhancer used in many foods. Fast foods often contain substantial amount of MSG and is widely used in Chines food. If one mole of above MSG was placed in 845 ml solution and passed through 200 mm tube, the observed rotation was found to be +9.6^(@). Find out the approximate percentage composition of (-) MSG in a mixture containing (+) MSG and (-) MSG whose specific optical rotation is -20^(@) |
|
Answer» 0.833 RM=100 -optical of purity =100-83.33=16.66% (-) MSG TOTAL in MIXTURE `rArr` `{:(83.33%),(UL(+8.33%)),(91.66%):}`] |
|
| 7. |
S(+) Mono Sodium Glutamate (MSG) is a flavour enhancer used in many foods. Fast foods often contain substantial amount of MSG and is widely used in Chines food. If one mole of above MSG was placed in 845 ml solution and passed through 200 mm tube, the observed rotation was found to be +9.6^(@). Find out of the specific rotation of (-) MSG : |
|
Answer» `+24^(@)` `C=(169 gm)/(845 ml)L =2m` `[theta^(@)]=(theta)/(CL)=9.6/(169//845xx2) =[-24^(@)`] |
|
| 8. |
S is a well known pain killer which of the following is 'R'? |
|
Answer» `CH_(3)Coverset(O)OVERSET(||)C-NH_(2)` |
|
| 9. |
S-I: Heavy metal acetylides can be used to purify alkynes S-II: Terminal alkynes form acetylides which are soluble in acids. |
|
Answer» S-I&II are CORRECT, S-II is correct explanaion of S-I |
|
| 10. |
S-I: Both benzene & ethyne give same product on ozonolysis. S-II: Ethyne & benzene possess same emperical formula. |
|
Answer» S-I & S-II are CORRECT, S-II is corret explanation of S-I |
|
| 11. |
S_(H_(2(g)))^(0) = 130.6 JK^(-1) mol^(-1), S_(H_(2)O_((l)))^(0) = 69.9JK^(-1) mol^(-1), S_(O_(2(g)))^(0) = 205 JK^(-1), Then the absolute entropy change of H_(2(g)) + (1)/(2)O_(2(g)) rarr H_(2)O_((l)) is |
|
Answer» `-163.2 J MOL ^(-1) K ^(-1)` |
|
| 12. |
s- andp- Block element arecollectively called ………… elements. |
| Answer» SOLUTION :REPRESENTATIVE ELEMENTS | |
| 13. |
S _(8) + 8O _(2) to 8SO _(2), Delta H =- QKJ.Then Delta H for SO _(2) to S + O _(2) is |
| Answer» ANSWER :C | |
| 14. |
S+(3)/(2) O_(2) to SO_(3) + y " kJ" SO_(2) + (1)/(2) O_(2) to SO_(3) + 2x " kJ" calculate the enthalpy of formation SO_(2). |
|
Answer» `2X+y` |
|
| 15. |
Rutherford'sexperiment , which established the nuclear model of the atom, used a beam of |
|
Answer» `beta`-particles which impinged ona metal foil and got ABSORBED |
|
| 16. |
Rutherford's alpha ray scattering experiment showed for the first time that that atom has |
|
Answer» Nucleus |
|
| 17. |
Rutherford studied the first nuclear reaction [._(7)^(14)N(alpha,p) ._(8)^(17)O] which take place with a change in energy equivalent to 1.193 MeV. Later on varioustypes of nuclear reactions such as artifical radioactivity, artifical transmutatoon , nuclear fission, nuclear fussion, spallation reactions etc. were studied. Which one is correct? |
|
Answer» Nuclear FUSION involves `0.231%` of mass decay |
|
| 18. |
Rutherford proposed the atomic meodel after his most stiking experiment an apha-scattering leading to discoveluy of nucleus . Bohr later on modified the atomci model on the balis of Planck's quantum theory of light and prosed the concept of stateonry cireclar orbits of quanise enrgy(nh) /( 2pi) The colloection of fine lines in line spectrum led Sommerfeld to gae the idea fo elliptical orbits .He successfully explained the existence fo subshells and their number in a shell . The angular momentum of subshells was proposed assqrt ( l(l +120)) . h/( 2 pi) . The emission of a spectral line in atomic spectra was supposed to be due to the jump of elecrtron from one enrgy level to other . The ninumum enrgy is given out when an elecrton jumps form one obit to oter form : |
| Answer» Solution :`E_5 -E-4` is ninimum as ` E_n = E-1 n_^2`. | |
| 19. |
Rutherfordexperimentprove |
|
Answer» Electron orbital`=n^(2)= 16:e^(-) =2 n^(2) = 32` |
|
| 20. |
Russia wanted to ban supersonic jets because |
|
Answer» They travel with high SPEED |
|
| 21. |
Rules for to decide relative stability for different resonance structure |
| Answer» Solution :(i) Among the resonance structures, the one which has more number of covalent BONDS, all the atoms with octet of electrons (except HYDROGEN which has a duplet) are more stable. (ii) Among the resonance structures, less separation of opposite charges, (a negative charge if any on more electronegative atom, a positive charge if any on more ELECTROPOSITIVE atom) and more dispersal of charge, is more stable than others. | |
| 22. |
Rules for Markonikov's and explain with example. |
|
Answer» Solution :Propene reacts with HBr, gives product by 2-Bromo propane has electrophilic ADDITION reaction. Alkene reacts with HYDROGEN halide (HCl, HBr, HI) give product alkyl halide. This reaction is electrophilic addition reaction. (a) "RULE of markonikov" or give the addition reaction of alkene with HBr (HX) : Markovnikov gave rule as 1869. These generalisation in Markovnikov to frame a rule is called Markovnikov rule. The rule states that negative part of the addendum (adding molecule) gets attached to that carbon atom which possesses lesser number of hydrogen atoms. example :(a) Addition reaction of propene to HBr, product (I) and (II) is obtained but as per Markovnikov.s rule, product (I) b is only obtained. Addition of propene to HBr gives product (I) and (II).  (b) As per Markovnikov.s rule, `Br^(-)` is reactant HBr, attaches with LESS number of hdyrogen and forms double donded product. It is a main product.  Information of product formed by Markovnikov.s rule in reaction : In any reaction, product is obtained in major amount. Series of CARBOCATION in `3^(@) gt 2^(@) gt 1^(@) overset(+)(C)H_(3)`. In `pi`-bond, `pi`-electron having -ve charge, `H^(+)` is attarcted and `pi`-bond is broken, formation of `sigma` -bond stable carbocation is formed.  (ii) First `Br^(-)` attaches fast with (y) and forms below product.  Note : In unsaturated alkene C=C, both carbon can have different substitution but has same number of substitution. Reactivity of HX in alkene is `HI gt HBr gt HCl`. Reactivity of carbocation is `3^(@)-C^(+) gt 2^(@) -C^(+) gt 1^(@) - C^(+) gt overset(+)(C)H_(3)`. |
|
| 23. |
Rubies and sapphires are essentially |
|
Answer» `Al_(2)O_(3)+C` |
|
| 24. |
Rubber bands comprise of loosely packed chains of atoms.When stretched, the chain of atoms get neatly lined up in rows and hens the entrops of system decreases whereas when contacted the chains get tangled up in a mess increasing entropy of system. Also on strectching rubber band, it is observed that any substance brought in its contact gets heated up. Based on this information, answer the following questions. Which of the following statement(s) is/are correct? |
|
Answer» A van dar wallls' get at is CRITICAL condition SUBJECTED to adiabatic free expansion experiences cooling effect. |
|
| 25. |
Rubber bands comprise of loosely packed chains of atoms.When stretched, the chain of atoms get neatly lined up in rows and hens the entrops of system decreases whereas when contacted the chains get tangled up in a mess increasing entropy of system. Also on strectching rubber band, it is observed that any substance brought in its contact gets heated up. Based on this information, answer the following questions. Which of the following statement (s) is/are true regarding the change "Rubber band"_("streched") rarr "Rubberband"_("unstreached")? |
|
Answer» The CHANGE is spontaneous. |
|
| 26. |
Rubber bands comprise of loosely packed chains of atoms.When stretched, the chain of atoms get neatly lined up in rows and hens the entrops of system decreases whereas when contacted the chains get tangled up in a mess increasing entropy of system. Also on strectching rubber band, it is observed that any substance brought in its contact gets heated up. Based on this information, answer the following questions. What should happen when a rubber band is heated ? |
|
Answer» It should EXPAND and GET strectched. |
|
| 27. |
Round up the following upto three significant figures : (i) 34.216(ii) 10.4107 (iii) 0.04597(iv) 2808 |
|
Answer» (II) `10.4107-10.4` (III) `0.04597-0.0460` (IV) `2808-2810` |
|
| 28. |
Round up the following upto three significant figures:(i) 34.216 (ii) 10.4107(iii) 0.04597 (iv) 2808 |
| Answer» SOLUTION :(i) 34.2, (II) 10.4, (III) 0.0460, (IV) 2810 | |
| 29. |
Round off the following to report each in three significant figures : (a) 13.821 (b) 4.256 and ( c) 162.5 |
|
Answer» SOLUTION :Rounding each in 3 significant FIGURES, (a) 13.821 is reported as 13.8, (b) 4.356 is reported as 4.36 and ( c) 162.5 is reported as 162 |
|
| 30. |
Rotations around carbon-carbon single bond of ethane is not completely free. Justify the statement. |
| Answer» Solution :Alkanes contain carbon-carbon sigma `(pi)` bonds. Electron distribution of the sigma molecular orbital is symmetrical around the inter nuclear axis of the C-C bond which is not disturbed due to rotatio about axis. This permits free rotation about C-C SINGLE bond. This rotation results into different spatial ARRANGEMENTS of atoms in space which can into ONE another. Such spatial arrangements of atoms which can be converted into one another by rotation around a C-C single bond are called conformation or conformer or rotamers. Alkanes canhave infinite number of conformations by rotations arounf C-C single bonds. How ever, it may be remebered that rotation around a C-C single by a small bond is not completely free. It is HINDERED by a small energy barrier of `1-20 KJ mol^(-1)` due to weak repulsive interaction between the adjacent bonds. Such a type of repulsive interaction is called torsional strain. | |
| 31. |
Rotation around carbon-carbon single bond of ethane is not completely free. Justify the statement. |
|
Answer» Solution :ROTATION about C-C single bond is restricted due to repulsions between the electron clouds of C-H bond on adjacent carbon atoms . As a resultof these repulsions, ethane exists in infinite number of conformations out of which two extreme conformations ,i.e., staggered and eclipsed are important . The staggered conformation is , however , more STABLE than the eclipsed conformation by about 12.55 kJ `mol^(-1)`. This ENERGY difference of `12.55 kJ mol^(-1)` between the staggered and the eclipsed conformation is , in fact, the energy barrier to rotation about C-C single bond in ethane . However, this energy barrier is not large enough to prevent rotation . EVEN at room temperature, collisions of the molecules supply sufficient kinetic energy to overcome this energy barrier. Thus, the two conformations are readily interconvertible . As a result , it is not possible to separate the two confomations of ethane. However, at any given moment, most of the molecules would exist in the staggered conformation due to its MINIMUM energy and maximum stability. |
|
| 32. |
Roshan heard that instructions were given to the laboratory attendant to store a particular chemical, i.e. keep it in the dark room, add some urea in it, and keep it away from dust. This chemical acts as an oxidising as well as a reducing agent in both acidic and alkaline media. This chemical is important for use in the pollution control treatment of domestic and industrial effluents. (i) Write the name of this compound. (ii) Explain why such precautions are taken for storing this chemical. |
|
Answer» Solution :(i) The name of the compound is hydrogen peroxide, `H_2O_2` . It ACTS as an oxidising AGENT as well as reducing agent in both acidic and basic medium. (ii) `H_2O_2` is decomposed by light and dust PARTICIES. Ureas is added as a negative catalyst. i.e. to check its decomposition. `2H_2O_(2(aq)) overset(HV)to 2H_2O_((L)) + O_(2(g))` Because of the oxidising properties `H_2O_2` used to control pollution by oxidation of harmful cyanides and obnoxious smelling sulphides present in domestic and industrial offluects It is also helps in sewage disposal by supplying `O_2` for oxidation of org. matter present in sewage water. |
|
| 33. |
Rosenmund's reduction of an acyl chloride gives |
|
Answer» An aldehyde |
|
| 34. |
Rohan heard that instructions wee given to the laboratory attendant to store a particular chemical i.e, keep it in the dark room, add some urea in it, and keep it away from dust. This chemical acts as an oxidising as well as reducing agent in both acidic and alkaline media. This chemical is important for use in the pollution control treatment of domestic and indusrtial effluents. (i) Write the name of this compound. (ii) Explain why such precautions are taken for storing this chemical. |
|
Answer» Solution :(a) The name of the compound is `H_(2)O_(2)` . It acts as an OXIDISING as well as reducing agent in both acidic basic medium. (b) `H_(2)O_(2)` is decomposed by light and dust particles . Urea is added as a negative catalyst , i.e., to check its decomposition `2H_(2)O_(2)(aq) overset(hv)to 2H_(2)O (l) + O_(2)(G)` Because of the oxidising PROPERTIES, `H_(2)O_(2)` is widely USED to control pollution by oxidation of harmful cyanides and obnoxious smelling SULPHIDES present in domestic adn industrial effluents . It also helps in sewage disposal by supplying `O_(2)` for oxidation of organic matter present in sewage waters. |
|
| 35. |
Rochelle's salt is used in the preparation of Fehling solution. Chemically, the salt is |
|
Answer» POSTASSIUM hydrogen phosphate |
|
| 37. |
Roasting of sulphides gives the gas X as a byproduct . This is a colourless gas with choking smell of burnt sulphur and causes great damage to respiratory organs as a result of acid rain. Its aqueous solution is acidic , acts as a reducing agent and its acid has never been isolated. The gas X is : |
|
Answer» `SO_3` `SO_2+ H_(2)O rArr [H_(2)SO_(3)] LTIMPLIES 2H^(+) + SO_(3)^(2-)` `SO_2` CAUSES acid rain and damage respiratory system. |
|
| 38. |
Roasting of sulphides gives the gas X as a by product. This is a colorless gas with choking smell of burnt sulphur and causes great damage to respiratory organs as it results in acid rain. Its aqueous solution is acidic and acts as a reducing agent, and its acid has never been isolated. The gas X is |
|
Answer» `SO_2` |
|
| 39. |
r_(n,z)= Radius of nth orbit of a single electron species having atomic number 'z' u_(n,z)=Velocity of electron in nth orbit of a single electron specie having atomic number 'z' E_(n,z) = Magnitude of total energy of electron in nth orbit of a single electron species having atomic number 'z'. K_(n,z)= Magnitude of kinetic energy of electron in nth orbit of a single electron species having atomic number 'z' P_(n,z)= Magnitude of potential energy of electron in nth orbit of a single electron species having atomic number 'z' f_(n,z)= Frequency of electron in nth orbit of a single electron species having atomic number 'z'. T_(n,z)= Time period of electron in nth orbit of a single electron species having atomic number 'z' |
|
Answer» |
|
| 40. |
RNC acts only as a nucleophile but not an electrophile. Explain why. |
| Answer» Solution :`R-overset(+)(N)-=C:^(-)`. Although N has a +ve charge but in cannot accept a pair of electrons because it has already 8 electrons in the valence shell and cannot expand its valence shell beyond 8 due to the absence of d-orbitals. Therefore, it cannot act as an electrophile. However, due to the presence of a -ve charge on carbon, it is EASILY donate a pair of electrons and HENCE can act as a nucleophile. | |
| 41. |
R.M.S velocity of ethane molecules when one mole of ethane is present in 10 lt vessel at 10 atm is |
|
Answer» `10^2` cm/sec |
|
| 42. |
RMS velocity of a gas does not vary with________at a given temperature. |
|
Answer» pressure |
|
| 43. |
RMS velocity of a gas at a given temperature is how much factor higher than its most probable velocity |
|
Answer» `3/2` |
|
| 44. |
R.M.S velecity of helium at S.T.Pis x cm/sec. R.M.S velocity of helium at 273 k and 4 atm is |
| Answer» Solution :`(U_1)/(U_2) = 1 ` as T is same. | |
| 45. |
Ring oxidation of mestiylene to mesitol is possible by |
|
Answer» `OSO_(4)` 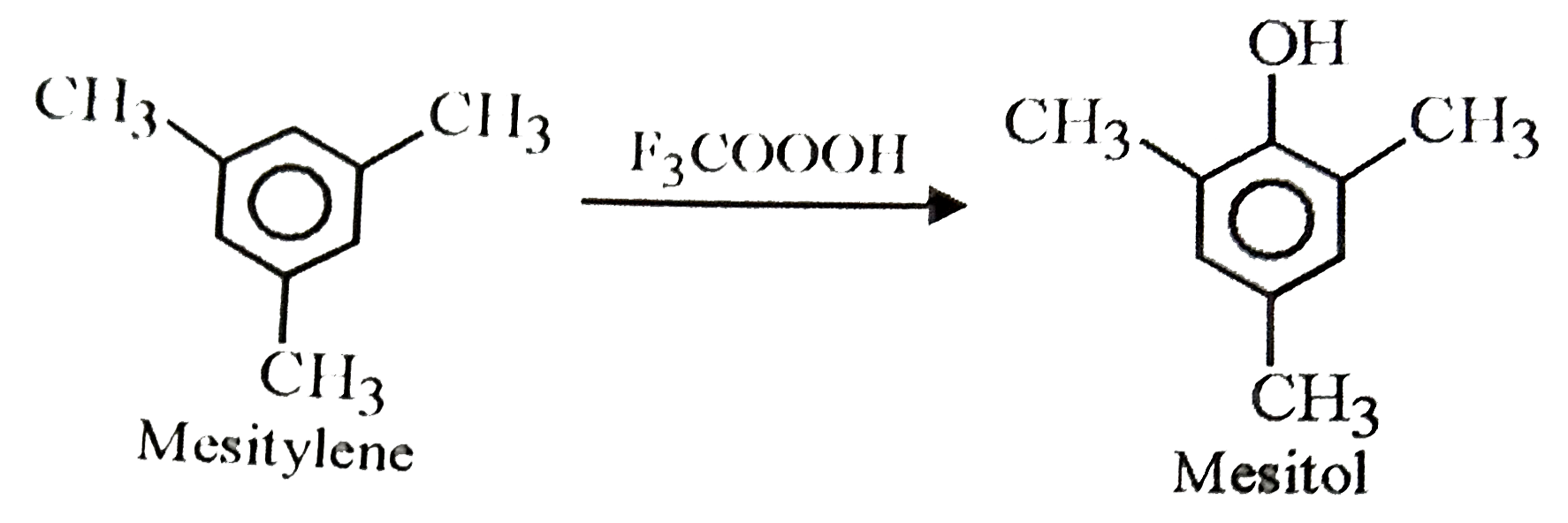
|
|
| 46. |
Ring nitration is more in |
|
Answer» `C_6H_5(CH_3)_3` |
|
| 47. |
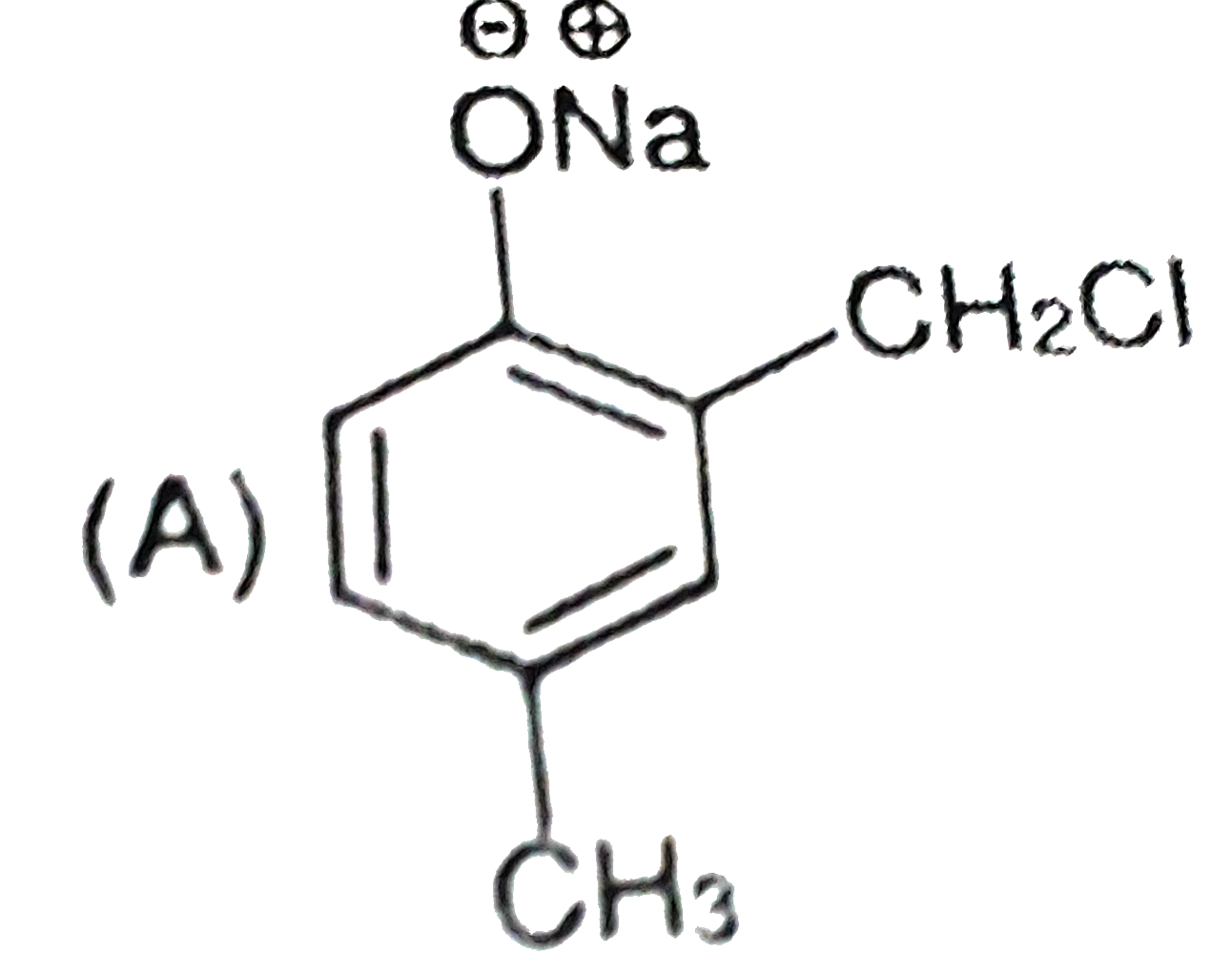
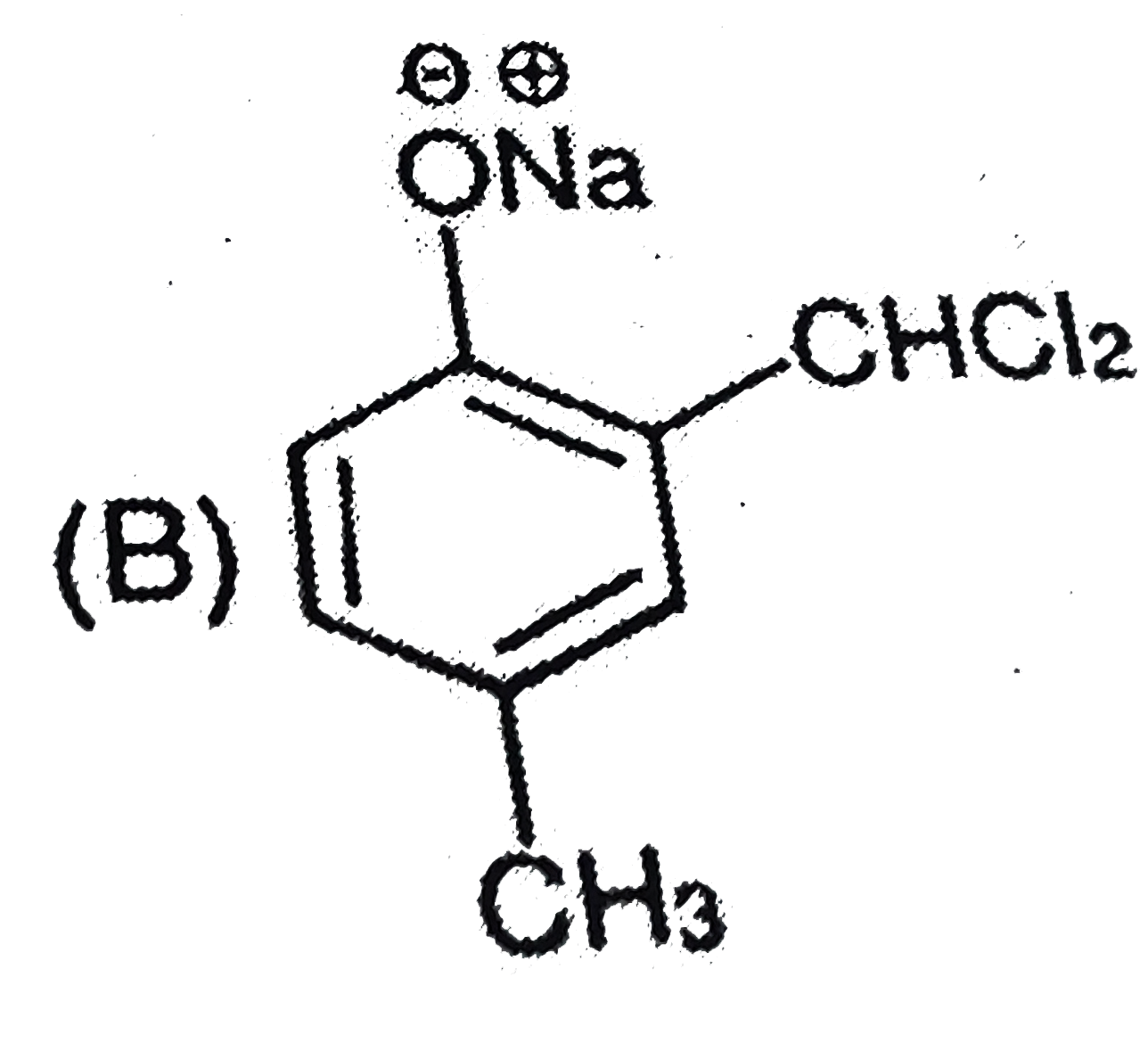
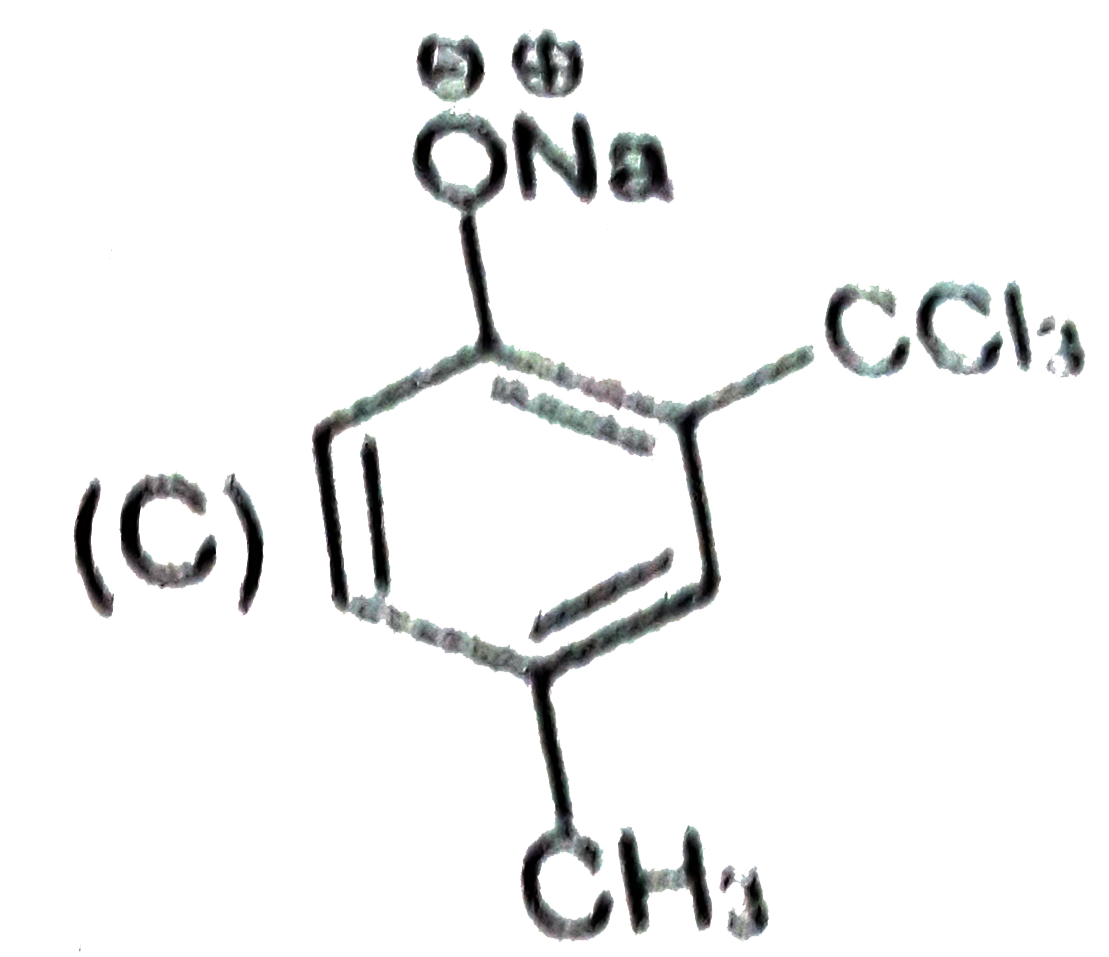
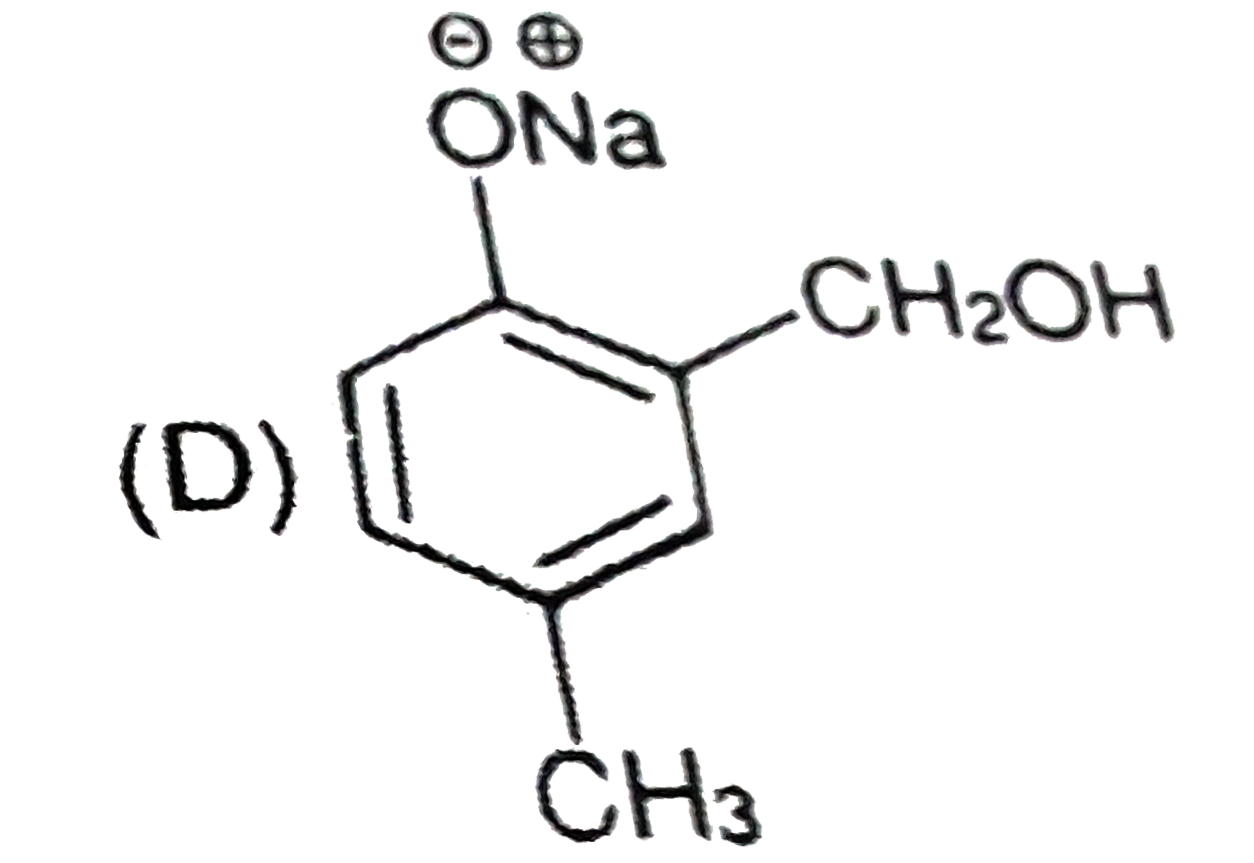
Riemer-Tiemann reaction intorduces an aldehyde group, on to the aromatic ring of phenol, ortho to the hydroxyl group. This reaction involves electrophilic aromatic substitution. This is a general method for the synthesis of substitued salicyladehydes as depicted below. Which one of the following reagents is used in the above reaction? |
|
Answer» aq. NaOH +`CH_(3)Cl` |
|
| 48. |
Riemer-Tiemann reaction intorduces an aldehyde group, on to the aromatic ring of phenol, ortho to the hydroxyl group. This reaction involves electrophilic aromatic substitution. This is a general method for the synthesis of substitued salicyladehydes as depicted below. The structure of the intermediate I is: |
|
Answer»
|
|
| 49. |
Result of ozone hole is |
|
Answer» GREEN house effect |
|
| 50. |
Respresentdiagrammatically the bond moments and the resultant dipole moment in(i) SO_(2) (ii) cis trans forms of C_(2) H_(2) Cl_(2) |
Answer» SOLUTION :(i)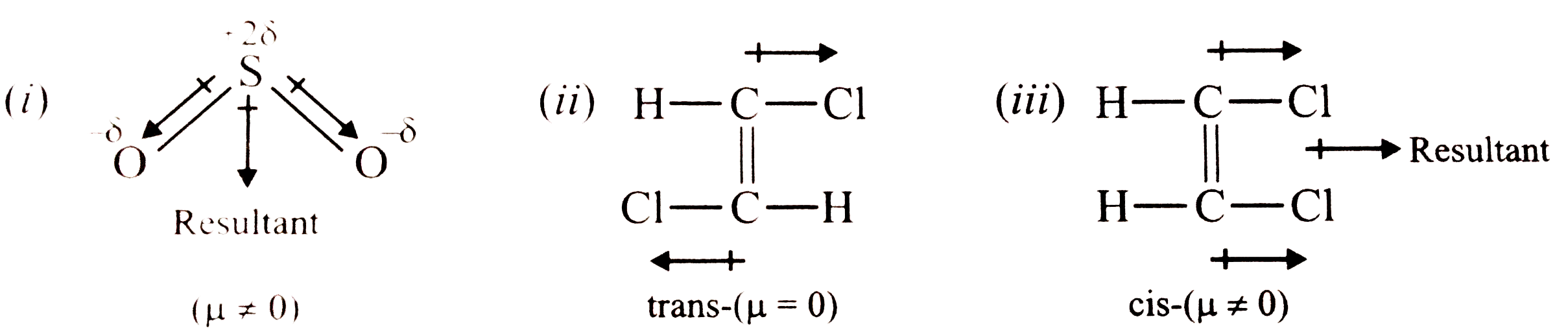
|
|