Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Schottky defect is observed in crystal when ______ |
|
Answer» some CATIONS move from their LATTICE sites to interstitial sites |
|
| 2. |
Schottky defect is observed in crystal when ……… |
|
Answer» some CATIONS move from their lattice sites to interstitial sites. |
|
| 3. |
Schottky defect generally appears in |
| Answer» Solution :Schottky defect is common in highly ionic compounds with high coordination number and SIZE of two ions (cations and anions) are almost same. e.g. NaCl, CsCl, KCl and KBr. | |
| 4. |
Schotten Baumann reaction is |
|
Answer» `"Phenol + BENZOYL CHLORIDE" overset(NaOH)rarr "Phenyl banzoate"` |
|
| 5. |
Schiff's base and Schiff's reagent are |
|
Answer» STRUCTURAL isomers |
|
| 6. |
Schiff's and Piria method is used for the estimation of: |
|
Answer» nitrogen |
|
| 7. |
Scattering of light takes place in |
|
Answer» electrolysis |
|
| 8. |
Saturatedsolutionof crudeNaCl overset("x ") toPureNaCl.In thereaction ,X is___ |
|
Answer» HCIgas |
|
| 9. |
Satement 1 A solution of FeSO_(54) can be stored in a copper vessel Statement 2 2E^(@) "of" Cu "lt" E^(@) of Fe |
|
Answer» Statement 1 is true statement 2 is true , statement 2 is a corrrect EXPLANATION for statement 1 |
|
| 10. |
Sate Gay Lussac's Law. Calculate the pressure exerted by 4 mole of agas occuping a volume of 1.5m^(3) at 100 K. Given R=8.314 J/k/mol. |
|
Answer» Solution :GAY Lussac.s law (PRESSURE-temperature relationship) Gay Lussac.s law states that at constant volume, pressure of a fixed amount of a GAS varies DIRECTLY with the temperature. Given `n=4` moles `V=1.5m^(3),T=100klR=8.314`J/kmol, `P=?` `P=nRTimplies(nRT)/V=(4"moles"xx8.314"Jk mol"xx100k)/(1.5m^(3))=8269Pa` |
|
| 11. |
Saponification of ethyl benzoate with caustic soda as alkalie, gives |
|
Answer» Benzyl alochol, ETHANOIC ACID |
|
| 12. |
Saponification means hydrolysis of an ester with |
| Answer» Solution :dil. NaOH. | |
| 13. |
Saponification of an oil results in the formation of |
| Answer» Solution :Glycerol and soap | |
| 14. |
Sample of water has hardness 77.5ppm Ca^(2+). If this is passed through an ion exchange column where Ca^(2+) is replaced by H^(+), what is the pH of water after it has been so treated |
|
Answer» 1.4 |
|
| 15. |
Sample of different gases are given at different conditions in column-I and column-II consisting of translational kinetic energy of these gases at given conditions. {:(,"Column-I",,"Column-II"),("(P)",2mole SO_(2)(g)at 700K,,"(1)Maximum K.E. per gram"),("(Q)",1 mole SO_(3)(g)at 400K,,"(2)Maximum total K.E."),("(R)",4 mole CH_(4)(g)at 300K,,"(3)Maximum K.E. per gram"),("(S)",2.5 mole He(g)at 450 K,,"(4)Minimum K.E. per molecule"):} |
|
Answer» `{:("PQRS"),("4312"):}` |
|
| 16. |
Same volume of two monoatomic gases A and B is mixed at same temperature. ........ .. Is (C_p)/( C_v) = …....... |
|
Answer» `0.83` `therefore (C_p)/( C_v) = (5)/(3) = 1.67` |
|
| 17. |
Same reasons are given reagarding the limited use of H_(2) as fuel (I) Its calorific value is low (II) It availability in free state is less (III) It is difficult to store (IV) Its transportation is easy The correct statement are |
| Answer» Answer :D | |
| 18. |
Same mass of diamond and graphite ( bothbeing carbon) are burnt in oxygen. Will be heat produced be same or different ? Why ? |
| Answer» Solution :Heat EVOLVED will be difficult . This is because they have DIFFERENT crystal STRUCTURE. | |
| 19. |
Same functional group containing different organic compounds possesses………chemical reaction |
|
Answer» not equal |
|
| 20. |
Same amount of metal combines with 0.1g of oxygen and lg of a halogen. Hence the equivalent mass of halogen is : |
|
Answer» 9 `8 G O_(2) rarr ? = 80` The amount of the substance which combines with the 8gm of oxygen is CALLED its eqwt. |
|
| 21. |
Samarium-62 is the member of..... |
|
Answer» s-block element |
|
| 22. |
samall quantities of solutionof componds TX ,TYand TZ are put in to separate test tubes cntaining x y and z solutoin Tx does not react with any of these TY reacts oth with x and z tz rectswithx th drcasing order of ase of oxidation of the anons X^(-) Y^(-0) and Z^(-1) id |
|
Answer» `Y^(-),Z^(-),X^(-)` `Y^(-) +XrarrX^(-)+Y` and `Y^(-)+ZrarrZ^(-)+Y` THEREFORE `Y^(-)` is mosteasily oxidizedsince tZ reacts with ony X i.r `Z^(-)`is more easily oxidized than `X^(-)` combining all the result the decreasing order of OXIDATION of anions follows the order `Y^(-)GTZ^(-)gtX^(-)` |
|
| 23. |
Salts given in column-I reacts with the excess of reagents given in column-II and form white/coloured precipitates. Select the correct options for the salts given in column-I with the reagent(s) given in the column-II. |
|
Answer» `CU^(2+)` is soluble in `NH_(3)` solution (excess). |
|
| 24. |
Salt (W) gives golden yellow colouration in Bunsen flame. When (W) is oxidised by acidified K_(2)Cr_(2)O_(7), a colourless gas (X) is evolved which turns lime water milky and the milkiness disappears on passing excess of gas (X). However gas (X) does not decolourise pink solution of acidified KMnO_(4). when aqueous solution of salt (W) is added slowly to hydrated MnO_(2), red coloured solution is obtained due to formation of soluble complex (Y) along with evolution of gas (X). The red coloured complex (Y) is: |
|
Answer» `[Mn(C_(2)O_(4))_(3)]^(2-)` `7C_(2)O_(4)^(2-)+2MnO(OH)_(2)darr +8H^(+) rarr 2[Mn(C_(2)O_(4))_(3)]^(3-)+2CO_(2)uarr +6H_(2)O` |
|
| 25. |
Salt (W) gives golden yellow colouration in Bunsen flame. When (W) is oxidised by acidified K_(2)Cr_(2)O_(7), a colourless gas (X) is evolved which turns lime water milky and the milkiness disappears on passing excess of gas (X). However gas (X) does not decolourise pink solution of acidified KMnO_(4). when aqueous solution of salt (W) is added slowly to hydrated MnO_(2), red coloured solution is obtained due to formation of soluble complex (Y) along with evolution of gas (X). The salt (W) is: |
|
Answer» `HCOONa` `7C_(2)O_(4)^(2-)+2MnO(OH)_(2)darr +8H^(+) rarr 2[Mn(C_(2)O_(4))_(3)]^(3-)+2CO_(2)uarr +6H_(2)O` |
|
| 26. |
Salt (W) gives golden yellow colouration in Bunsen flame. When (W) is oxidised by acidified K_(2)Cr_(2)O_(7), a colourless gas (X) is evolved which turns lime water milky and the milkiness disappears on passing excess of gas (X). However gas (X) does not decolourise pink solution of acidified KMnO_(4). when aqueous solution of salt (W) is added slowly to hydrated MnO_(2), red coloured solution is obtained due to formation of soluble complex (Y) along with evolution of gas (X). Gas (X) is: |
|
Answer» `CO_(2)` `7C_(2)O_(4)^(2-)+2MnO(OH)_(2)DARR +8H^(+) rarr 2[Mn(C_(2)O_(4))_(3)]^(3-)+2CO_(2)uarr +6H_(2)O` |
|
| 27. |
Salt solution of 'A' + dil. HCI rarr only 'B' gas{:(("suffocating odour of burning sulphur")),(""darrBaCI_(2)),('C'["white ppt"]rarr"Soluble in dilHCI and gives 'B' gas"),(""darr"Reagent M"),("White ppt.of"[D]("insoluble in dilHCI")):} Find the total number of reagents 'M': (conc. HNO_(3)),O_(2),H_(2)O_(2),N_(2),NH_(3),(Br_(2)+H_(2)O) |
|
Answer» <BR> Solution :`HNO_(3),O_(2),H_(2)O_(2),(Br_(2)+H_(2)O)` |
|
| 28. |
Salt is a mixed form of acidic and basic radical. The confirmatory test of basic and acidic radicals by using chemical reagent is called qualitative analysis. Precipitation reaction is used for the confirmatory test of radicals. Which of the following is an example of IIA group basic radical sulphide? |
| Answer» Answer :C | |
| 29. |
Salt is a mixed form of acidic and basic radical. The confirmatory test of basic and acidic radicals by using chemical reagent is called qualitative analysis. Precipitation reaction is used for the confirmatory test of radicals. Which of the following iodide has scarlet red colour? |
|
Answer» `PbI_(2)` |
|
| 30. |
Salt bridge maintains ……………………in the solution of two half cells |
|
Answer» |
|
| 31. |
Salt (A) on heating gives a colourless neutral gas which supports combustion. From the aforesaid, flow diagram, answer the following questions. Which of the following statement is correct? |
|
Answer» Salt (A) gives yellow PRECIPITATE with chloroplantinic acid as well as with SODIUM cobaltinitrite. `(B) rarr NH_(3) (C) [Fe(H_(2)O)_(5)NO] SO_(4)` or `FeSO_(4).NO` `(A) rarr NH_(4)NO_(3)` |
|
| 32. |
Salt (A) on heating gives a colourless neutral gas which supports combustion. From the aforesaid, flow diagram, answer the following questions. The compound (A) contains the following acidic radical: |
|
Answer» `NO_(2)^(-)`<BR>`NO_(3)^(-)` `(B) rarr NH_(3) (C) [Fe(H_(2)O)_(5)NO] SO_(4)` or `FeSO_(4).NO` `(A) rarr NH_(4)NO_(3)` |
|
| 33. |
Salt (A) on heating gives a colourless neutral gas which supports combustion. From the aforesaid, flow diagram, answer the following questions. The basic radical of salt (A) and gas (B) both gives brown precipitate with Nessler's reagent. the composition of the brown precipitate is: |
|
Answer» `(NH_(4))_(2)[HgI_(4)]` `(B) rarr NH_(3) (C) [FE(H_(2)O)_(5)NO] SO_(4)` or `FeSO_(4).NO` `(A) rarr NH_(4)NO_(3)` |
|
| 34. |
Salol is prepared from |
|
Answer» SALICYLIC ACID and phenol 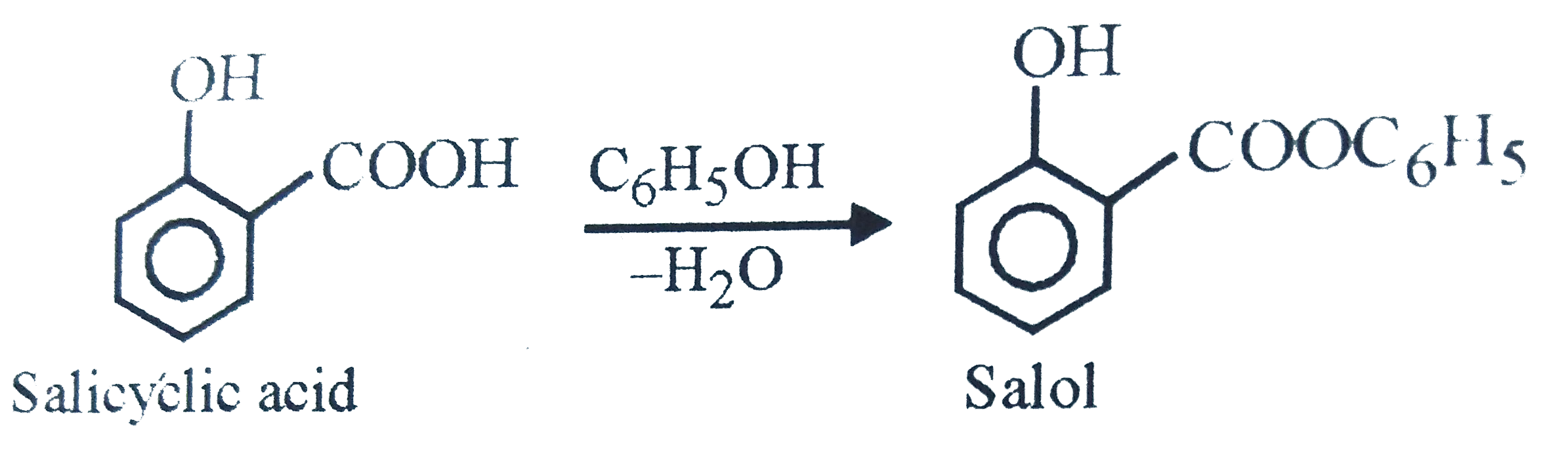
|
|
| 35. |
Salol is |
|
Answer» ACETYL SALICYLIC acid |
|
| 36. |
Salol can be used as |
|
Answer» Anseptic |
|
| 37. |
Saline hydrides are known to react violently with water producing fire, Can CO_(2), a well known extinguisher, be used in this case? Explain. |
|
Answer» SOLUTION :Whenever a saline hydride `(NaH or CaH_(2))` reacts with water, the reaction is so HIGHLY exothermic that the hydrogen evolved catched fire. For example, `NaH(s)+H_(2)O(aq) to NaOH(aq)+H_(2)(g)+"heat"` `CaH_(2)(s)+2H_(2)O(aq) to Ca(OH)_(2)(aq)+2H_(2)(g)+"heat"` `CO_(2)` which is normally used as fire EXTINGUISHER cannot be used in this case beacuse it will react with the hydroxide formed in the reaction to form a carbonate. This will INCREASE the rate of the forward reaction in which heat is evolved. `2NaOH(aq)+CO_(2)(g) to Na_(2)CO_(3)(aq)+H_(2)(aq).` |
|
| 38. |
Saline hydrides are known to react with water violently producing fire. Can CO_2 a well known fire extinguisher be used inthis case? Explain. |
| Answer» Solution :No.The FIRE produced by the action of water on SALINE hydrides cannot be put out by `CO_2`because the HOT METAL hydride reduces `CO_2` readily. `NaH+CO_2rarrHCOONa`(spadium formate) | |
| 39. |
Saline hydrides are known to react with water violently poducing fire. Can CO_(2) , a well known fire entingiusher, be used in this case? Explain . |
|
Answer» Solution :Saline hydrides (such as NaH, `CaH_(2)`, etc.) , react with water violently to form the corresponding metal hydroides with the evolution of dihyrogen . (`NaH(s) + H_(2)O(l) to NaOH(aq) + H_(2)(g)` `CaH_(2)(s) + 2H_(2)O(l) to Ca(OH)_(2) (aq) +2H_(2)(g)` These REACTIONS are so much exothermic that the evolved `H_(2)` catches fire. The fire so produced cannot be extinguished by`CO_(2)` because it gets reduced by the HOT metal hydride to form sodiym FORMATE. `NaH+ CO_(2) to HCOONa` `However, sand is useful since it is a highly STABLE solid |
|
| 40. |
Saline hydrides are known to react with water violently producing fire can CO_2, a well known fire extinguisher be used in this case ? Explain. |
|
Answer» Solution :Saline hydrides react violently with WATER PRODUCING DIHYDROGEN gas `NaH_((s)) + H_2O_((aq)) to NaOH_((aq)) + H_(2(g))` But A well known fire extinguisher `CO_2` is used then FOLLOWING red will be obtained `NaH+CO_2 to HCOONa` |
|
| 41. |
Salient features of Molecules Orbital Theory(MOT) |
|
Answer» Solution :1. The electrons in an atom are found in atomic orbtials, the electrons, in a MOLECULE are found in molecuar orbitals. 2. The molecule orbitals are formed by the COMBINATION of atomic orbitals of comparable energies ande proper symmetry. 3. The BMOhas LOWER energyand hence GREATER STABILITY than the corresponding b ABMO. 4 The molecular orbitals are filled by electrons in accordance with Aufau principle, Pauli.s exclustion principle and the Hung.s rule. |
|
| 42. |
Salicylic acid is prepared from phenol by |
|
Answer» REIMER Teimann reaction |
|
| 43. |
Salicylic acid is a stronger acid than p-hydroxybenzoic acid due to |
|
Answer» STERIC hindracne |
|
| 44. |
Salicyladehyde can be prepared from |
|
Answer» PHENOL and CHLOROFORM 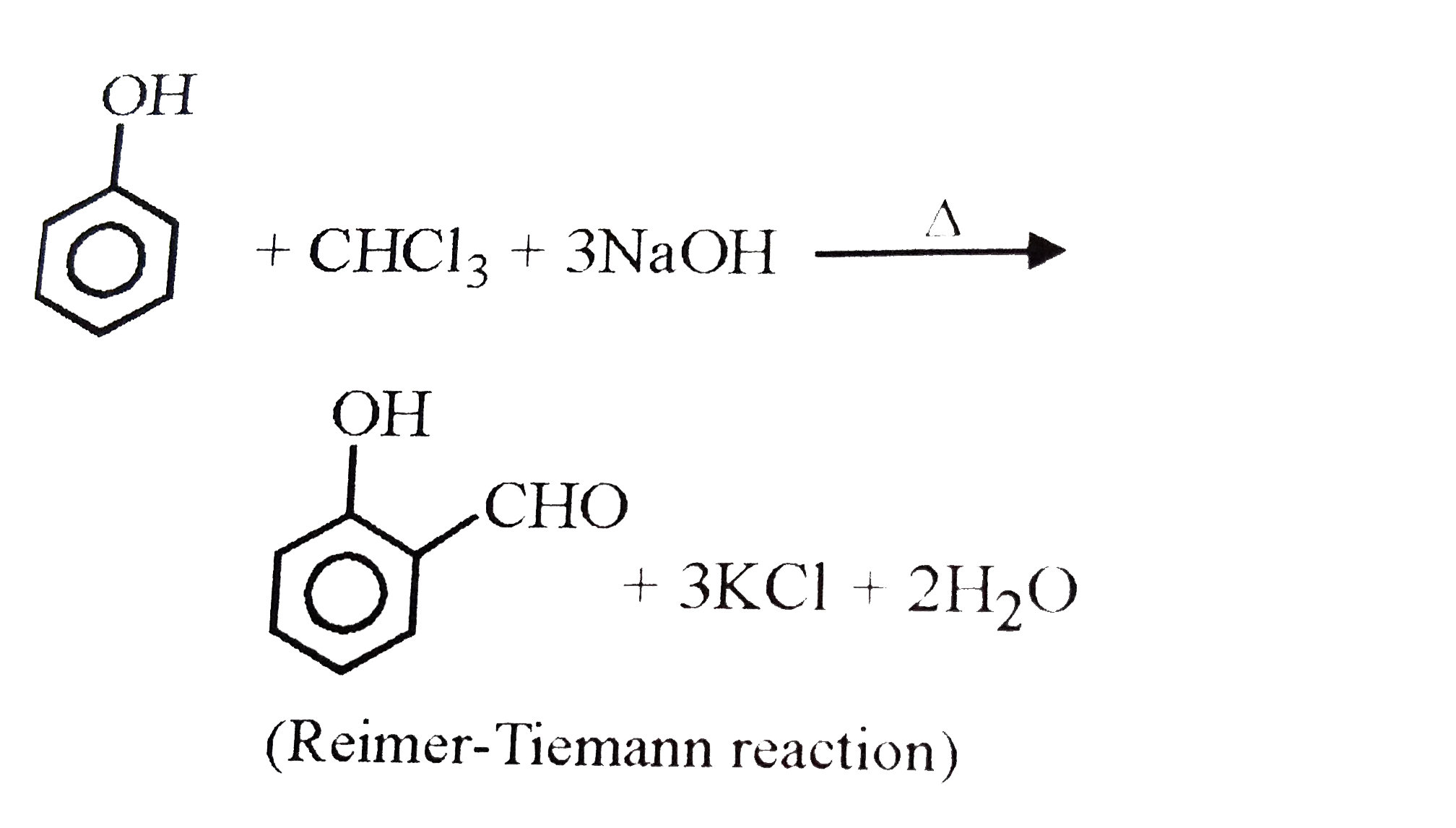
|
|
| 45. |
Salicyaldehyde involves________hydrogen bonding . |
|
Answer» |
|
| 46. |
Sacchrin an artificial sweetener, is manufactured from : |
|
Answer» CELLULOSE |
|
| 47. |
Saccharin, an artificial sweetener, is manufacturered from : |
|
Answer» cellulose |
|
| 49. |
S_(4)N_(x) in vapour state is subjected to decompoition totally to get octaatomic sulphur vapour and nitrogen gas . Under similar conditions the volume of products is 2.5 times to the reactants. Deduce the molecular formula of the nitride of sulphur. Calculate the weight percentage of nitrogen. |
|
Answer» Solution :The decompoition is given as `UNDERSET((G))(S_(4)N_(x))to underset((g))((1)/(2))S_(8) +underset((g))((x)/(8))N_(2)` By analysing the given data , 1+x=5:x=4 MOLECULAR formula =`S_(4)N_(4)` Weight percentage of NITROGEN `(4xx14)/(4(32+14))xx10=30.43%` |
|
| 50. |
S^(2-) and SO_(3)^(2-) ions can be distinguished by using |
|
Answer» `(CH_(3)COO)_(2) Pb` `Pb(CH_(3)COO)_(2)` gives a BLACK ppt with `S^(2-)` ions (e.g., `H_(2)S`) while no precipitate is formed with `SO_(3)^(2-)` ions. `Pb(CH_(3)COO)_(2) + H_(2)S rarr PBS rarr + underset(("Black ppt"))(2CH_(3) COOH)` `Na_(2) [Fe(CN)_(5)NO]` gives a purple colouration with `S^(2-)` ions `(Na_(2)S)` while no precipitate is formed with `SO_(3)^(2-)` ions. `Na_(2) [Fe(CN)_(5) NO] + Na_(2)S rarr Na_(4) [Fe(CN)_(5)NOS]` |
|