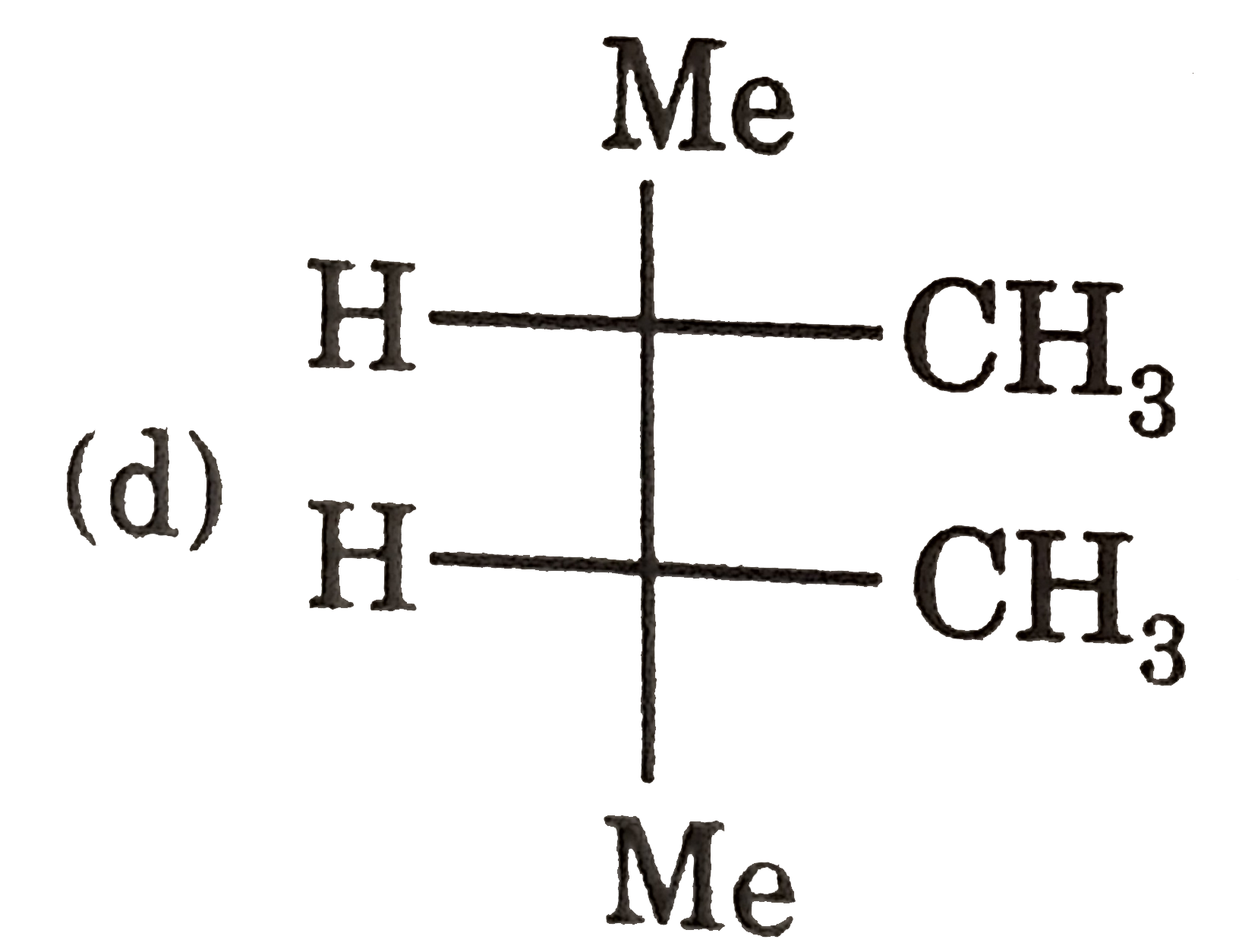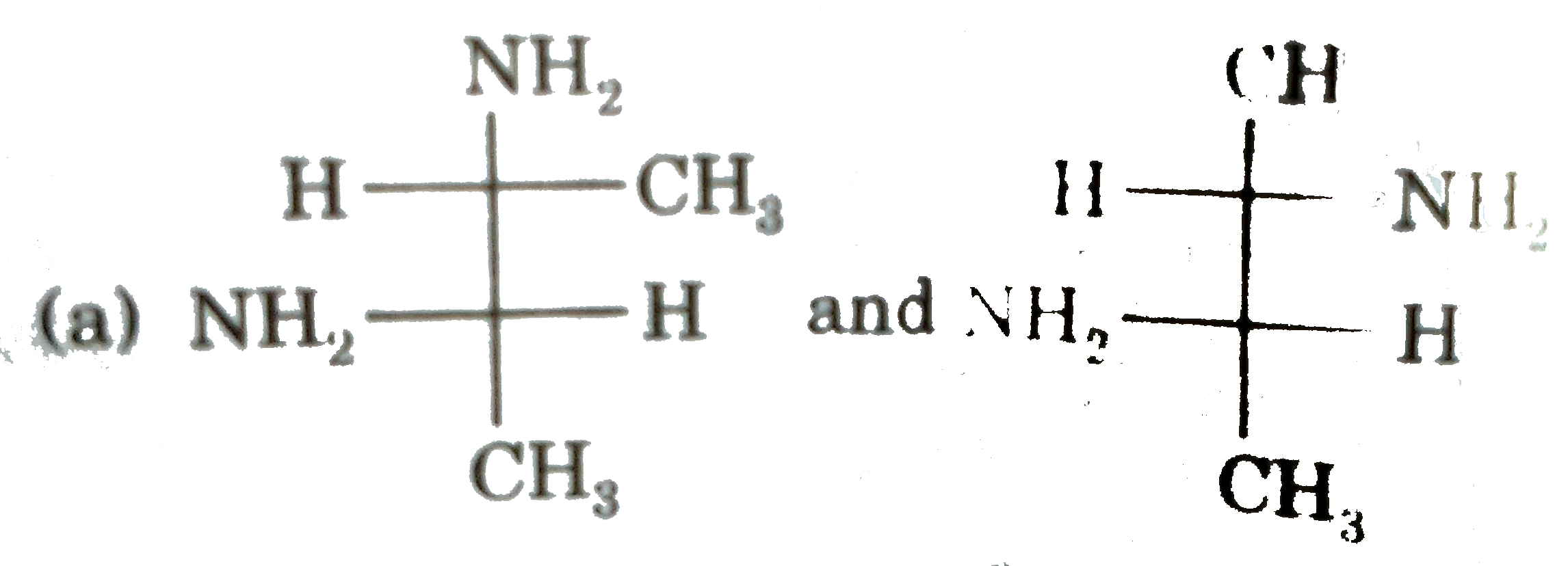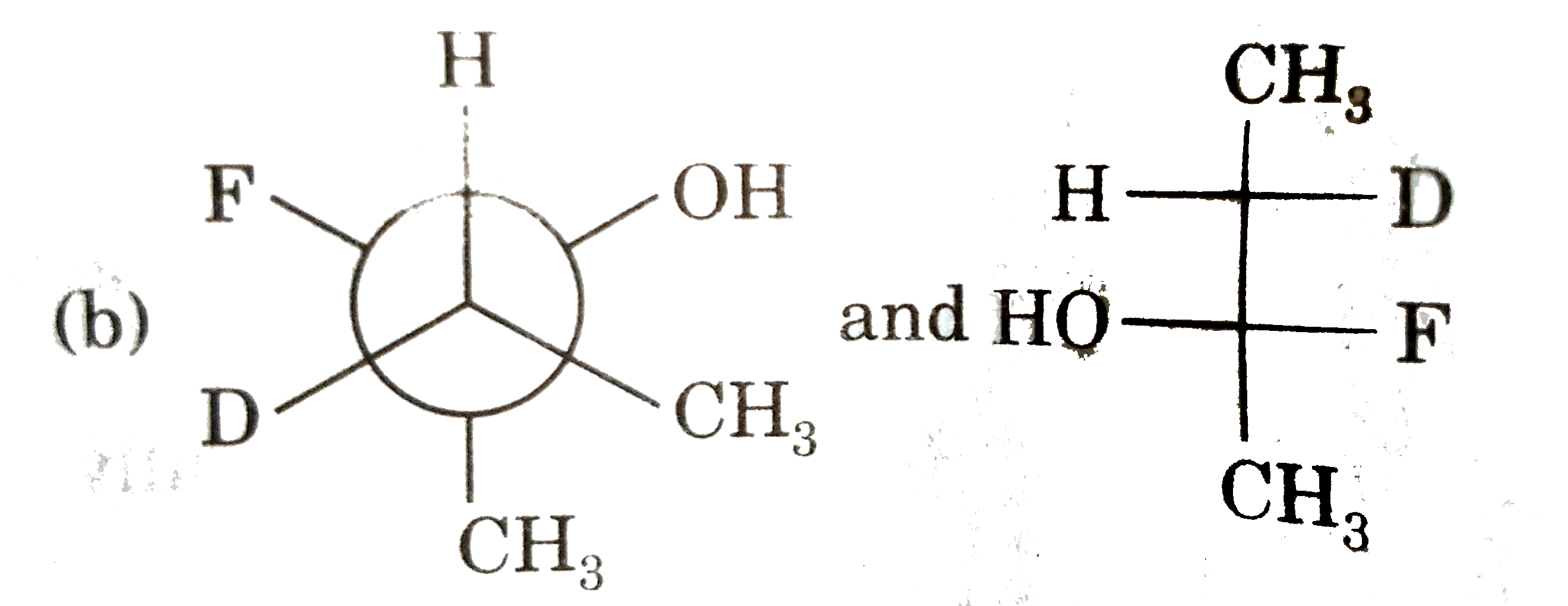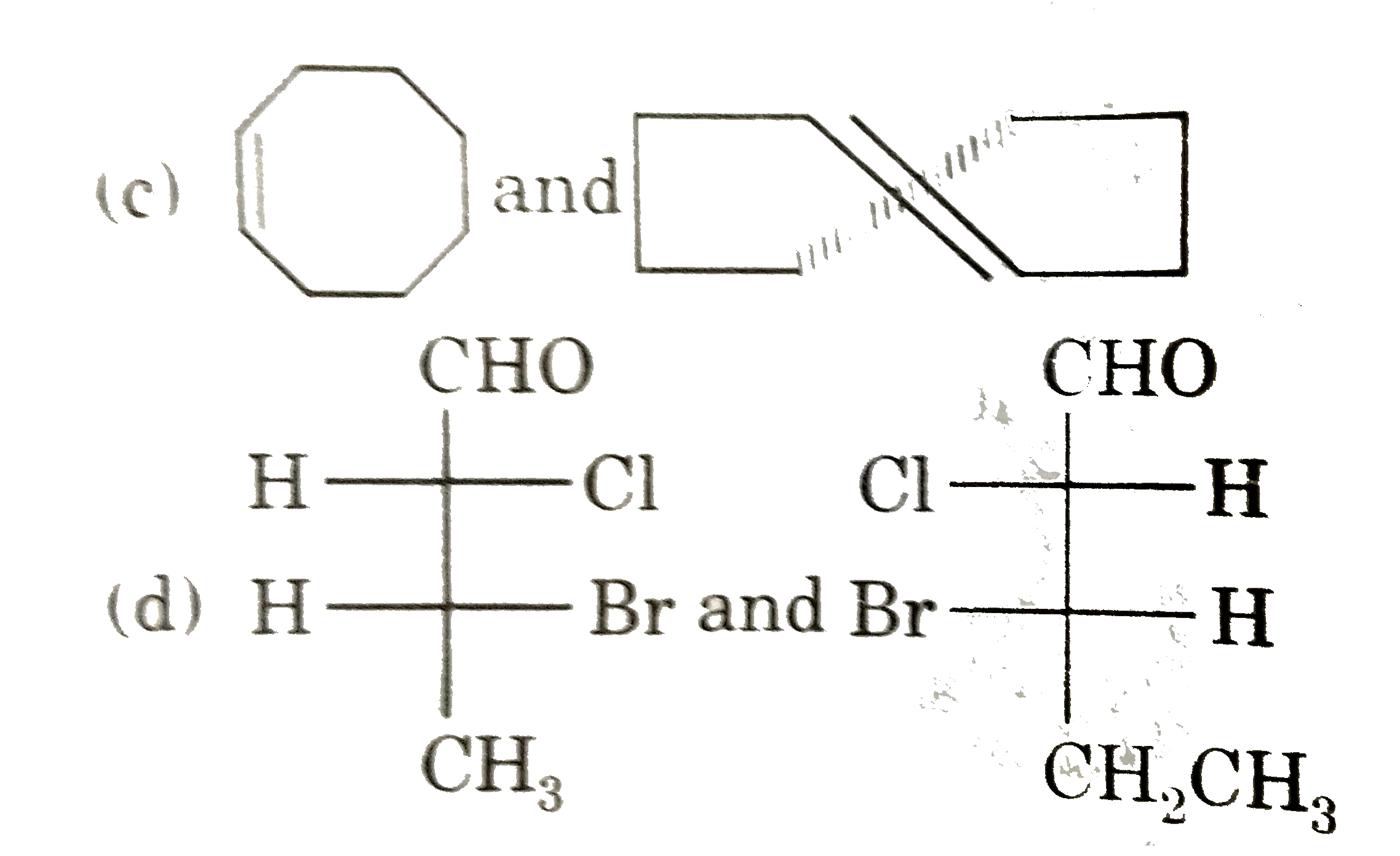Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the chain propogation steps in the free-radical chlorination of methane 1) Cl_2 to 2Cl^(*) 2) Cl^(*)+ CH_4 to CH_3Cl + H^(*) 3) Cl^(*)+CH_4 to CH_3^(*) + HCl 4)H^(*) + Cl_2 to HCl + Cl^(*) 5) CH_3^(*) + Cl_2 to CH_3Cl + Cl^(*) |
|
Answer» 2,3,5 |
|
| 2. |
Select the basic strength order of following molecules ? |
|
Answer» `III GT II gt I` |
|
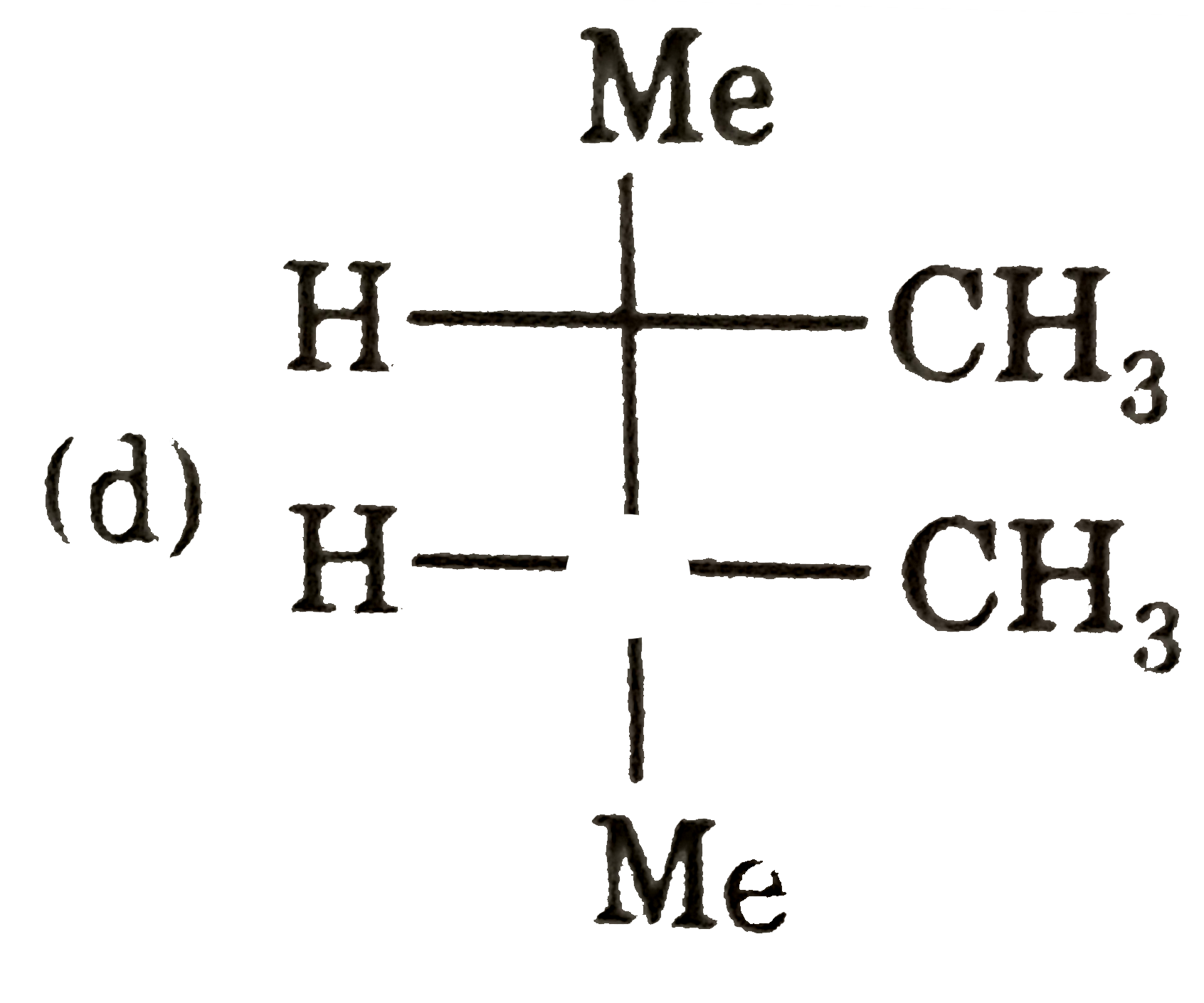
| 3. |
Select (T) statement(s). |
|
Answer» I and II are ENANTIOMERS of each other |
|
| 4. |
Select set of quantum numbers which is possible for maximum numberof electrons inan atom: |
|
Answer» (B)thisset representsmaximum two electronspresentin5 d orbital (c ) thissetrepesentsone 3s one 3p and3delectron . Somaximum3 such electronsare possibel (d) 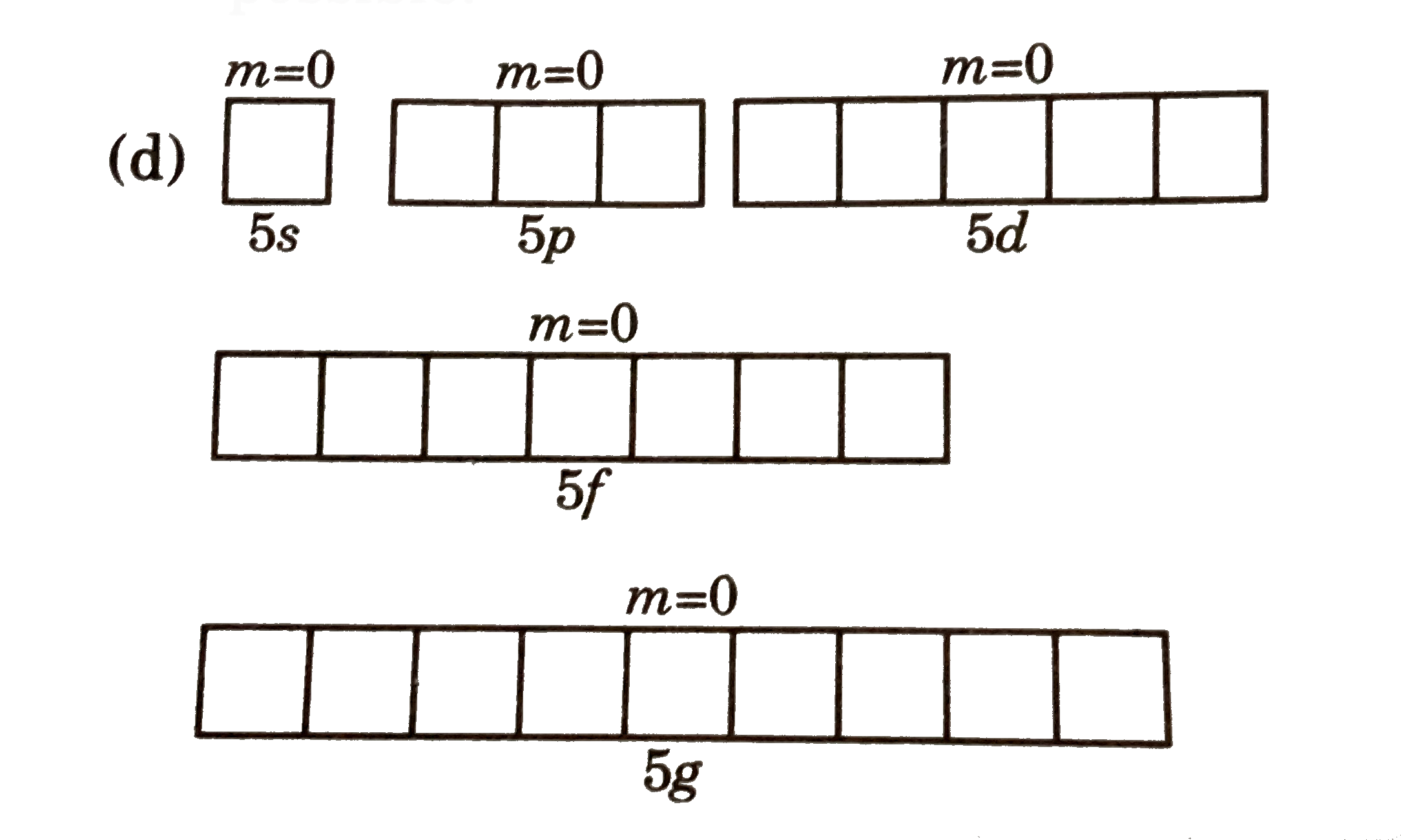 thisset represnts one 5 s one 5pone 5done 5 fandone 5g electrons Somaximum 5 electrons are POSSIBLE HENCE (d) iscorrect . |
|
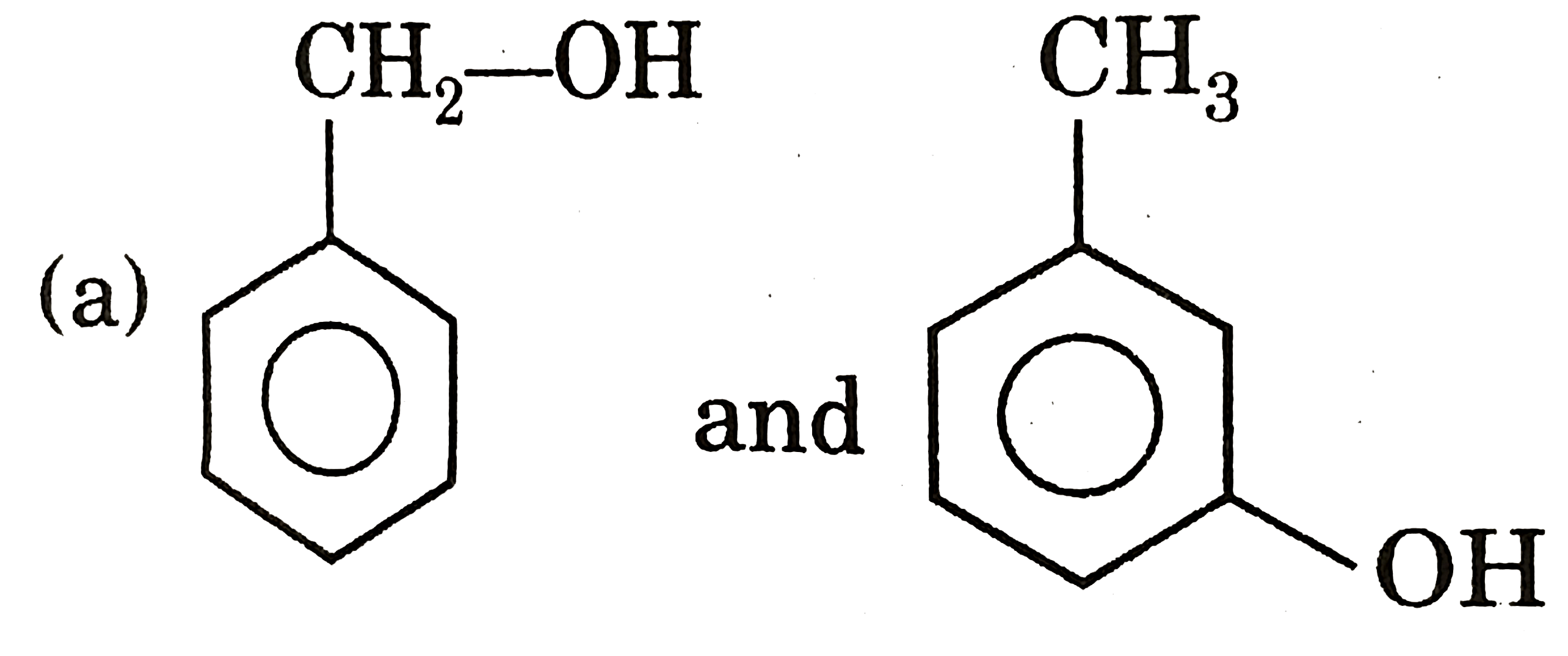
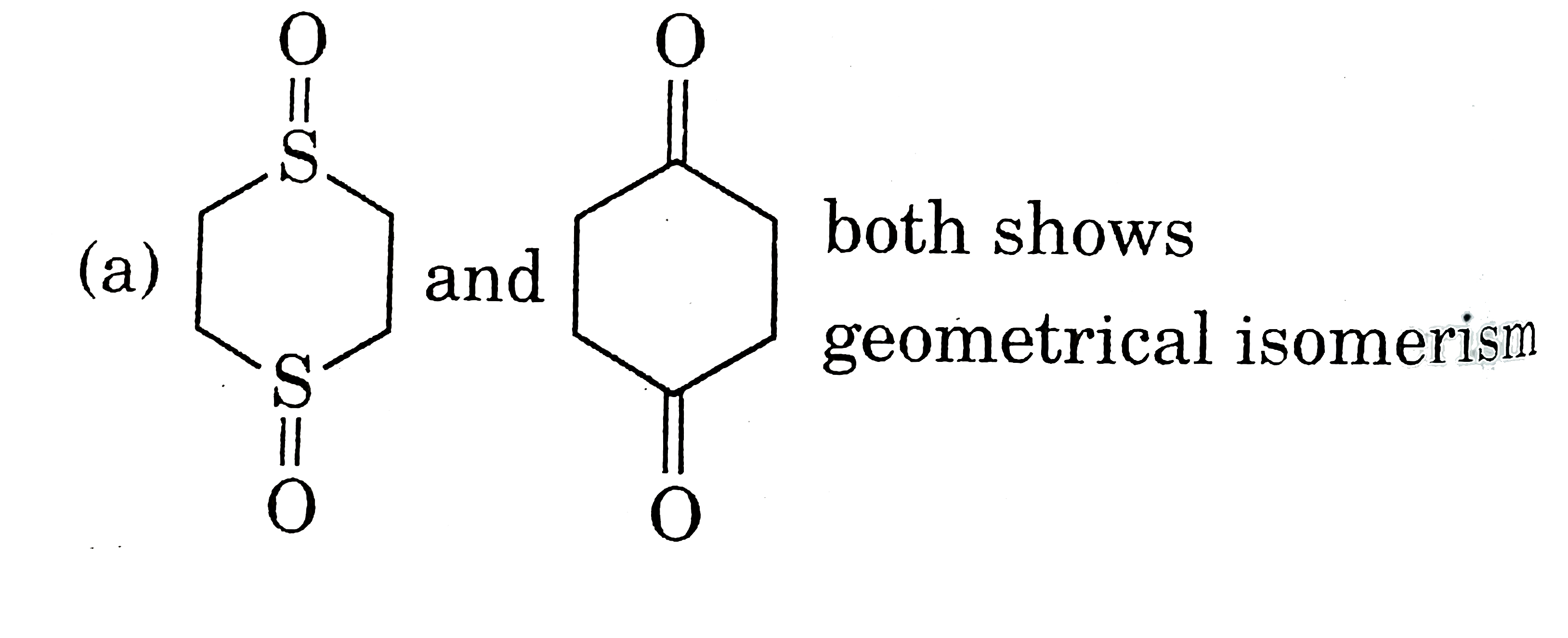
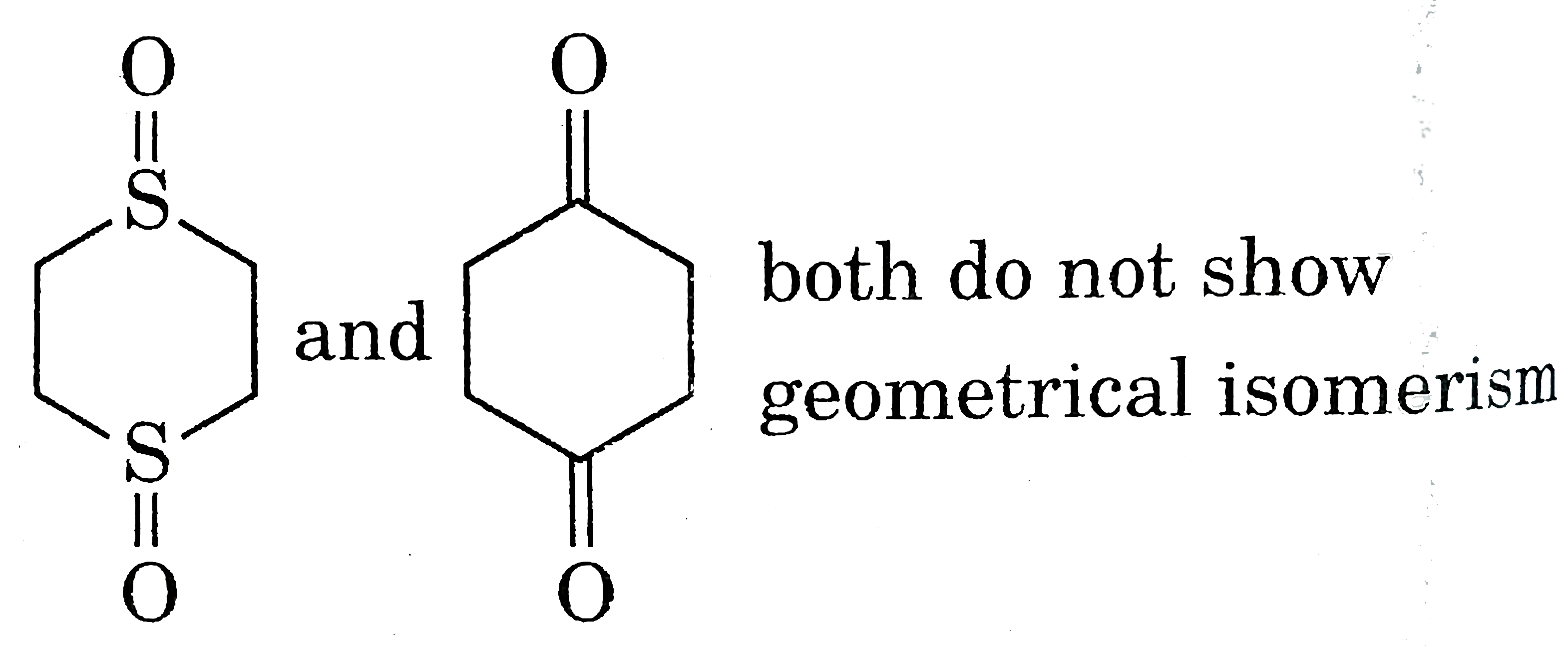
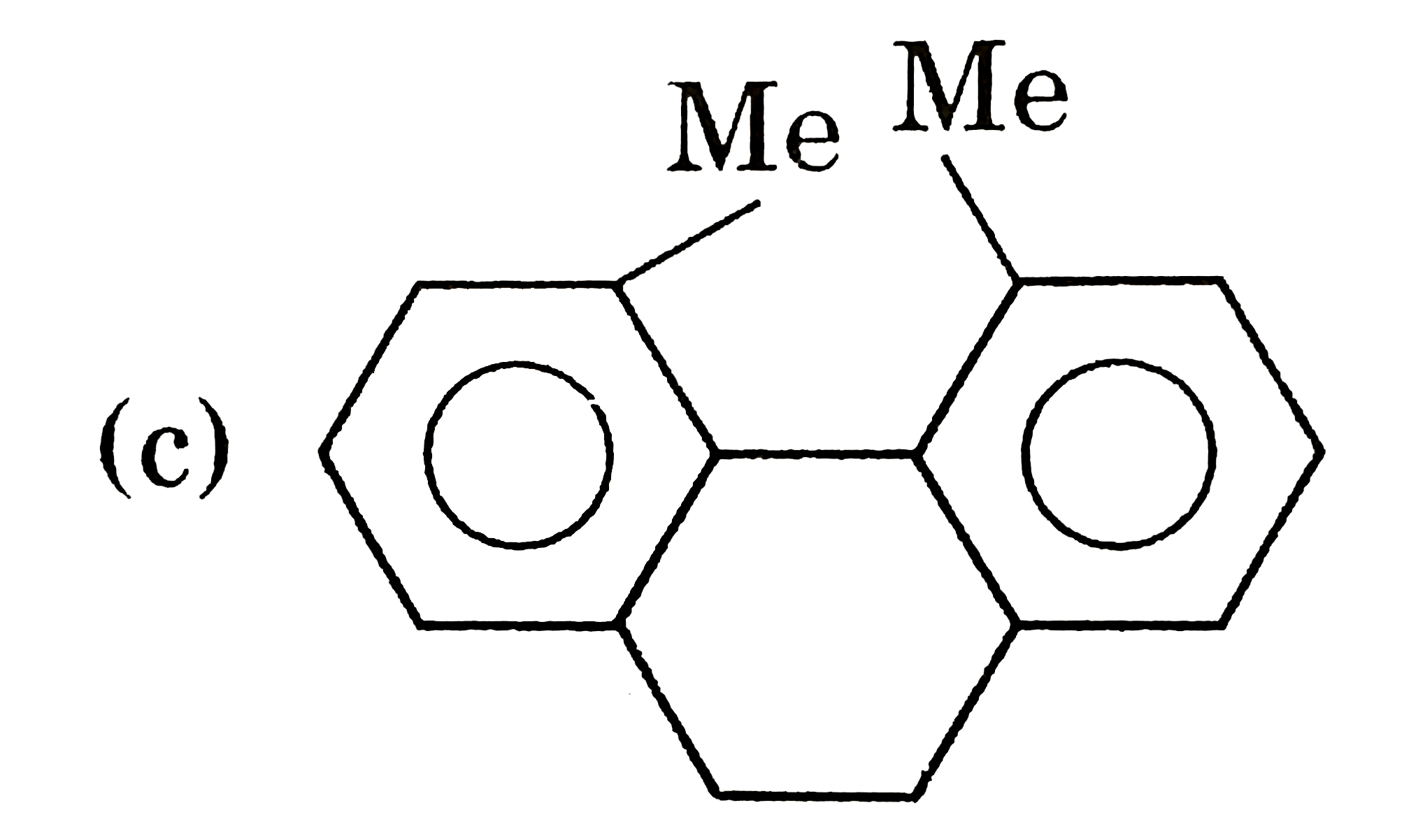
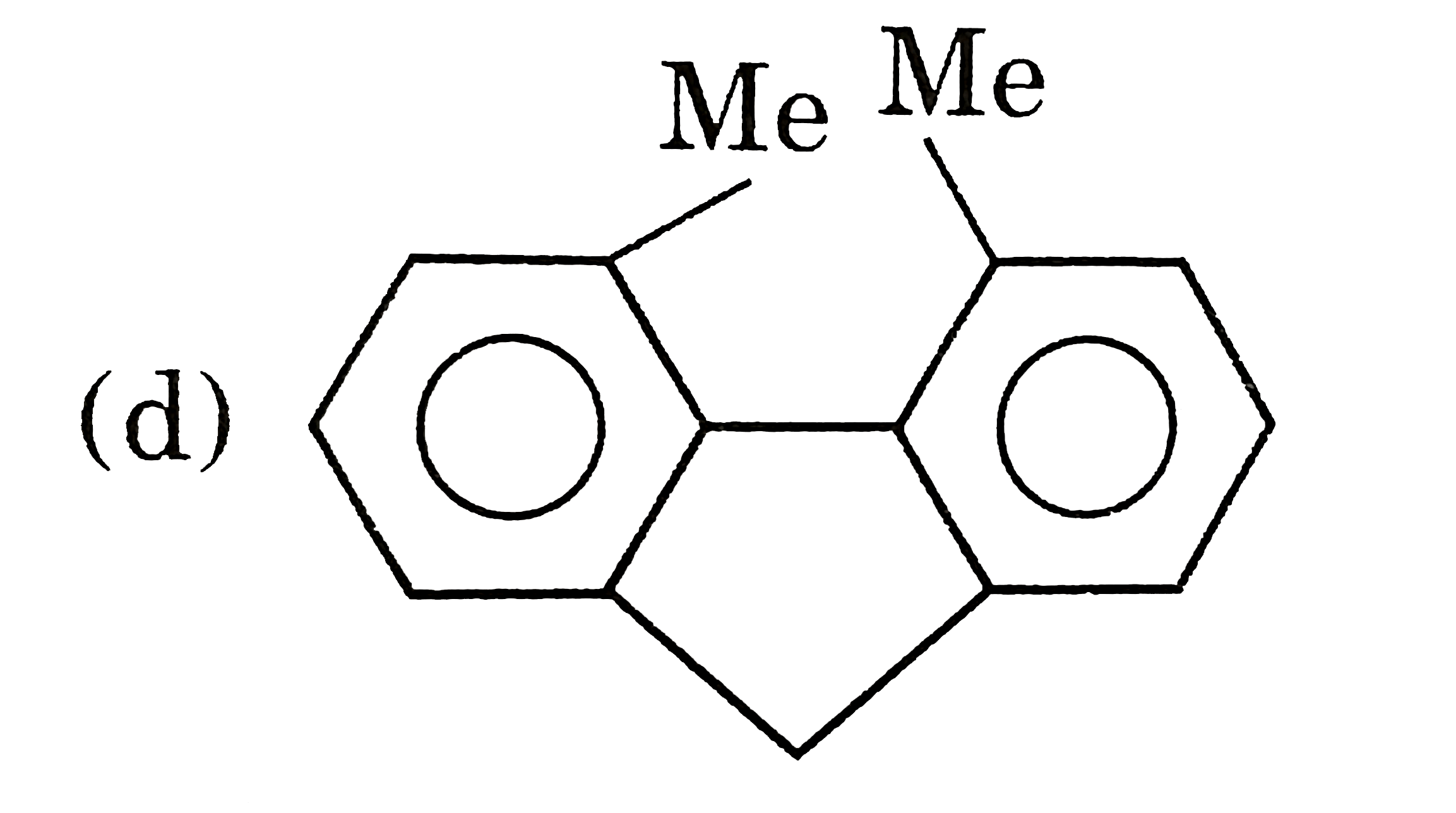
| 5. |
Select resolvable sompounds. |
|
Answer»
(B) No POS and COS so resolvable 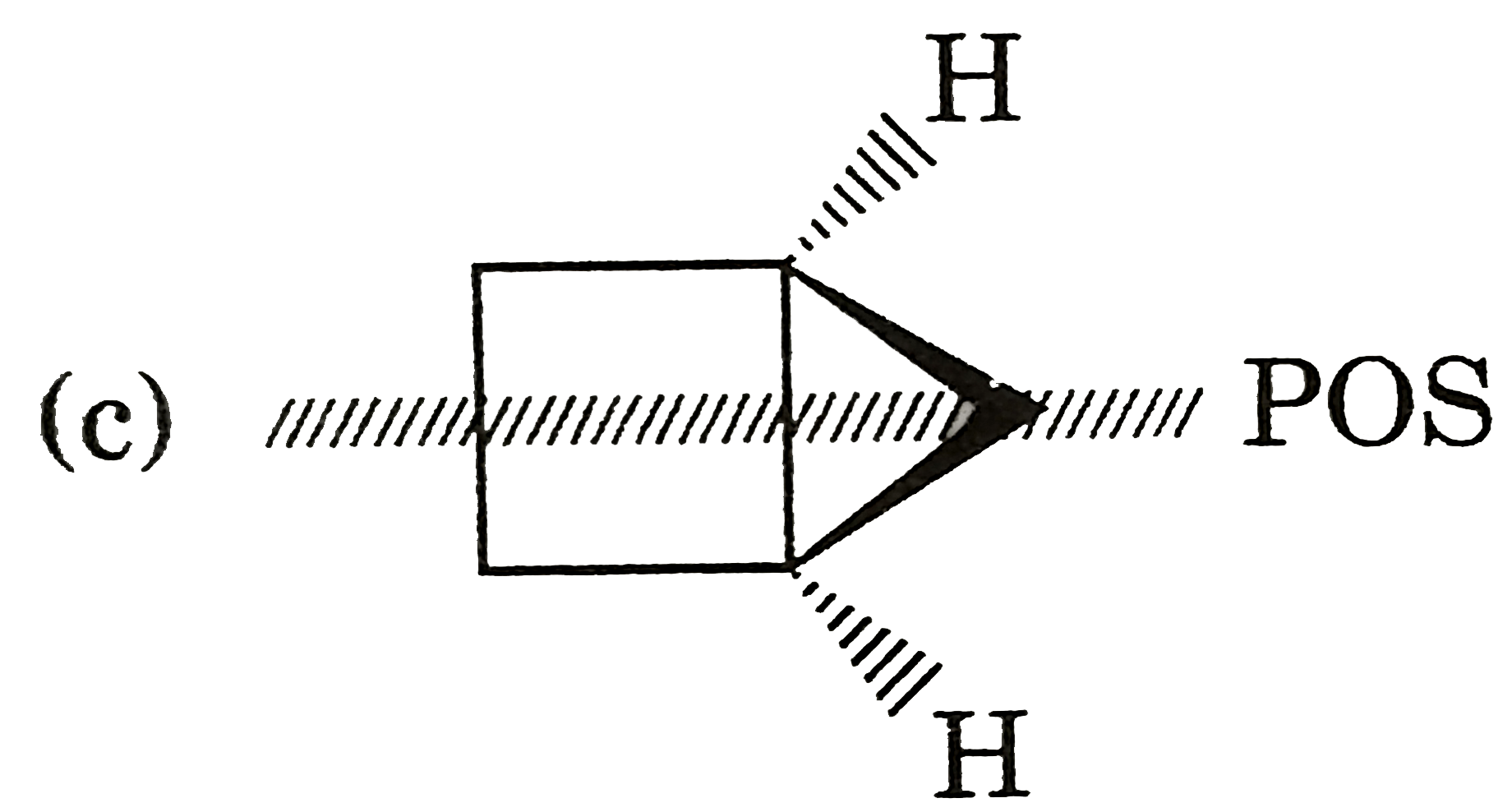 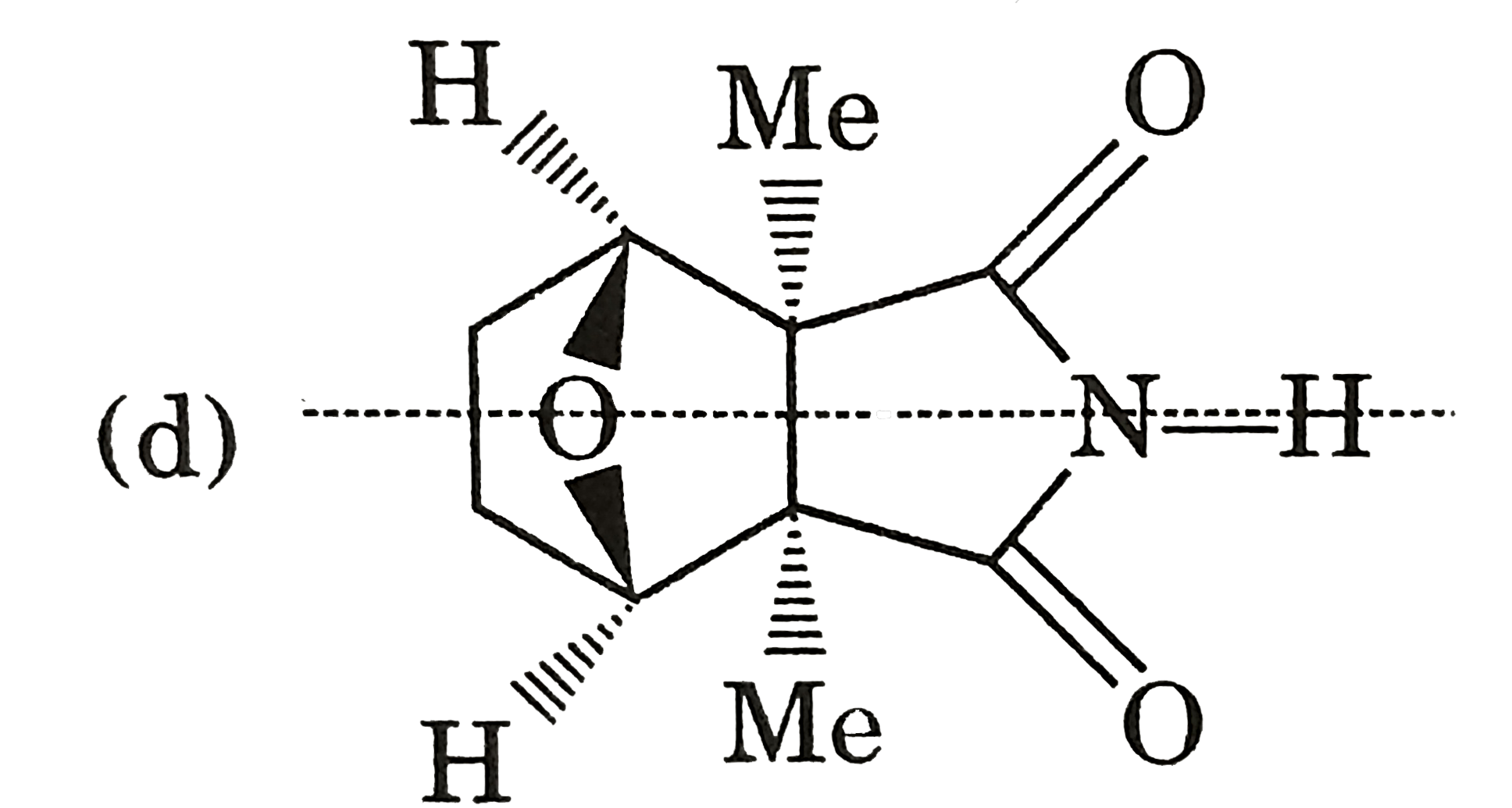
|
|
| 7. |
Select incorrect statements |
|
Answer» `(PV)/T = K`, the value of K is independent of amount of gas `IMPLIES K` is dependent of amount b) R does not depend on anything C) `R/(N_0)` is Boltzmann.s constant . d) KE depneds only on T. |
|
| 8. |
Select incorrect statements(s) |
|
Answer» At very low pressure REAL gases show minimum deviation from IDEAL behaviour |
|
| 9. |
Select incorrect statement (s). |
|
Answer» Conjugate acid of pyrrole is anti-aromatic 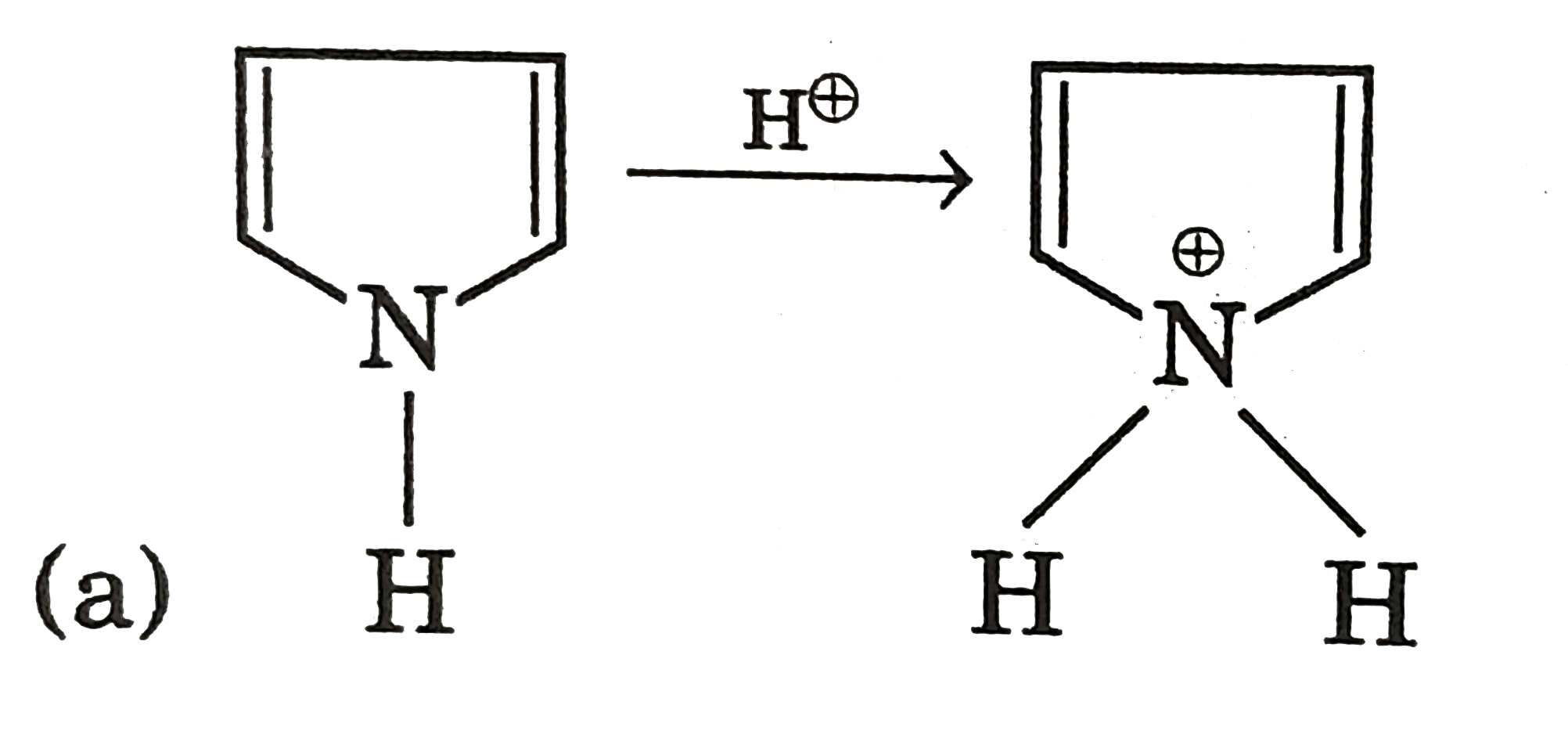 (b) `Me_(3)C-underset(O)underset(||)C-ND_(2)iffMe_(3)C-overset(O)overset(|)C=ND` ( c) `CH_(3)S^(-)` is more stable than `CH_(3)-O^(-)` due to larger size of SULPHUR atom 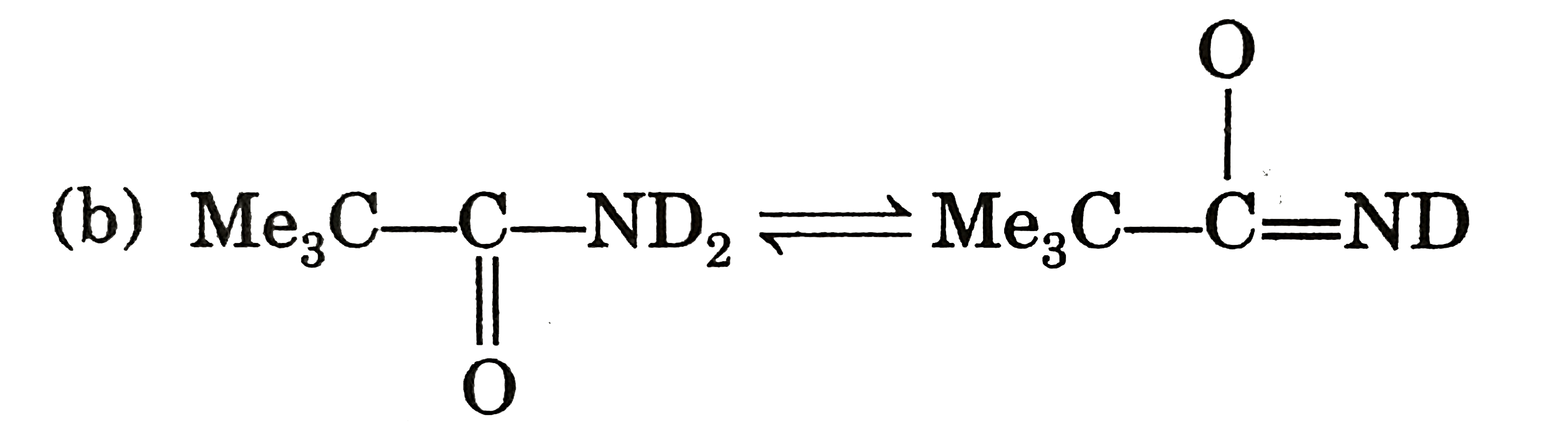 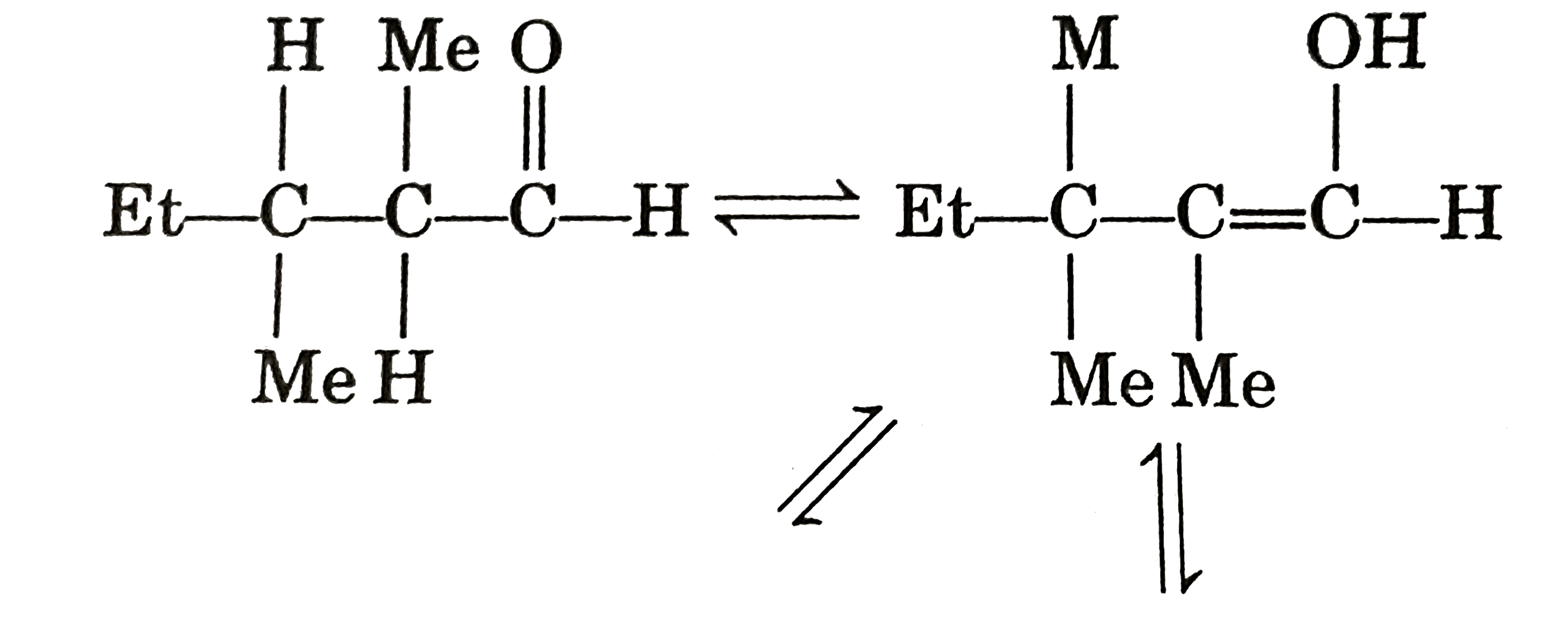 
|
|
| 10. |
Select incorrect statement |
|
Answer» NaOH is STRONG enough to remove the proton from phenol and carboxylic acids |
|
| 11. |
Select incorrect option. |
|
Answer»
|
|
| 12. |
Select he catalyst which can help in conversion of ethene into acetaldehyde? |
|
Answer» `Pd//Al_(2) O_(3)` |
|
| 13. |
Select from the following lists, the elemens belonging to same group |
|
Answer» Z = 12, 38, 4, 88 |
|
| 14. |
Select from each of the following sets those elements which belong to the same groupof the periodic table ? |
|
Answer» At. Nos. `12,4,88` |
|
| 15. |
Select from each of the following gropus, the one which has the largest radius (i) Co,Co^(2+),Co^(3+) (ii) S^(2-), Ar, K^(o+) (iii) Li,Na,Rb (iv) C,N,O (v) Ne,Na,Mg (vi) La,Lu (vii) Cu, Ag, Au (viii) Ba, H_(r) (ix) Mg, na Na^(o+) , Mg^(+2) ,Al (b) IE_(1) of C is 11.2 eV What would be the value of iE_(1) of Si to be greater or less than this amount ? (c) IE_(1) of Li and K are 5.4 and 4.3 eV respectively What would be the value of IE_(1) of P ? (d) IE_(1) of Na? . (e) The IE' s of Li, Be and C are 5.4, 9.3 and 11.3 eV What would be the value of IE s of B and N ? (f) Which of these elements have the lowest IE_(1) Sr, As, Xe, S,F ? (g) Select from each of the following group the element which has the largest IE (i) Na,P,CI (ii) He,Ne,Ar (iii) O,F,Na (h) Arrange the species in each group in order of decreasing IE' s and in each case explain the reason for the sequence (i) K,Rb,Cs (ii) Be,B,C (iii) Cu,Ag,Au ? (iv) C,N,O (v) N,O,F (vi) K, Ca, Sc (vii) Na,Mg,AI (viii) Fe,Fe^(+2),Fe^(+3) (ix) K^(o+),Ar,CI^(Θ) (i) Explain why IE_(1) of Cu is higher than that of K whereas IE_(2) are in reverse order (j) Account for the difference in IE (i) between K^(o+) and Ca^(o+) (ii) between Cu^(o+) and Zn^(o+) (k) Ionisation potential is an old term for ionisation energy. Explain why yhe two are synonymous ? . |
|
Answer» Solution :Co (The others have the same nuclear charge, but less electrons) (ii) `S^(2-)` (All have the same electronic configuration, but the `S` nucleus has the smallest positive charge) (iii) Rb (It is in the LARGEST period) (iv) `C` (It is the farthest left in the periodic table) (v) na(It is the first element of a new period) (vi) La(Lu is smaller because of lanthaniod contraction) (vii) Au (Ag is almost the same size, because of the effect of the lanthanoid contraction on Au) (viii) Ba(`HF` is much smaller because of the effect of the lanthanoid contraction) (ix) na(The size decreases along the period `(rarr)` and also as electrons are removed) (b) Less (`IE` decreases down the group `(darr)` due to increasing atomic size, but other factors also play an important role) The observed value is 8.1 EV (c) It is difficult to remove `e^(-)` from `P` than from `S` because of the added stability of the half- filled p-subshell The observed value is `10.9 eV` (d) The `IE` of Na should be intermediate between that of `Li` and `K` The `IE` of Na should be close to the arithmatic average of the two, or `4.9 eV` The observed is `5.1 eV` (e) Generally `IE` increases along the period `(rarr)` However there is a larger increase in `IE` from `Li(Z=3)` to Be `(Z=4)` than from Be to `C(Z=6)` `IE` of `B` actually is less than that of Be due to penetration effect in `B` The observed `IE` of `B` is `8.3 eV` N has a half -filled `2p` subshell and should have extra stability for this reason The increase in going from `N(Z=5)` to `(Z=6)` is `3.0 eV` and the additional increase should be greater than this, thus the `IE` of `N` should exceed `14.3 eV` The experimental value is `14.5 eV` (f) Sr (It is a metal) (g) (i) `CI` (All of them belong to 3rd period and `CI` is farthest to the right of the three elements) (ii) He (iii) Ne (h) (i) `K gt Rb gt CS` (Generally `IE` decreases down the group `(darr)`) (ii) `C gt Be gtB` (The `2se^(-)` is more difficult to remove than 2p (penetration effect) (iii) `Au gt Cu gtAg` (The lanthanoid contraction makes Ag and Au about equal in size but Au has a much greater nuclear charge) (iv) `N gt O gt C` (Due to stability of half- filledorbitals) (v) `F gtN gt O` (The half- filed p-subshell of `N` imparts enough extra stability to make its `IE gt IE` of O) (vi) `Sc gt Ca gt K` (In each of these cases, a `4s e^(-)` is being removed and the order is as shown) (vii) `Mg gtAI gt Na` (It is more difficult to remove an `e^(-)` from Mg because the `e^(-)` being removed is a 3s `e^(-)` from a filled subshell The `3pe^(-)` of AI is easily removed It is easiest to remove an `e^(-)` from na because of its large size) (viii) `Fe^(3+) gt Fe^(2+) gt Fe` (All have same nuclear charge and the number of `e^(-')s` increases in the order listed) (ix) `K^(o+) gt Ar gt CI^(Θ)` (All have the same electronic configuration and the nuclear chared decreases in the order listed) (i) `Cu(Z =29) implies 3d^(10) 4s^(1) , Cu^(o+) = 3d^(10) 4s^(@)` `Cu^(+2) = 3d^(9) 4s^(@)` (ii) `K(Z =19) implies 4s^(1), K^(o+) = 4s^(@), k^(+2) = 3p^(5)` Cu has 10 more protons and more `e^(-')s` than does `K` but due to imperfect SCREENING effect of `de^(-')s, IE_(1)` of `Cu gt IE_(1)` of `K` and `IE_(2)` for `K` involves the removal of an `e^(-)` from an octet (`3s^(2) 3p^(6))` whereas that of `Cu` involves the more easily ionised `d^(10)` configuration Hence `IE_(2)` of `K gt IE_(2)` of `Cu` (j) `K^(o+)` loses an `e^(-)` from its `3p^(6)` subhell, `Ca^(o+)` from its 4s subshhell, which requires less energey Hence `IE(2)` of `K^(o+) gt IE_(2)` of `Ca^(o+)` (ii) `Zn (Z =30) implies 3d^(10) 4s^(2) , Zn^(o+) implies 3d^(10) 4s^(1)` `Cu (Z =29) implies 3d^(10) 4s^(1) ,Cu^(o+) implies 3d^(10) 4s^(@)` `Zn^(o+)` loses an `e^(-)` from `4s^(1)` more easily than `Cu^(o+)` loses an `e^(-)` from `3d^(10)` Hence `IE_(2)` of `Cu^(o+) gt IE_(2)` of `Zn^(o+)` (k) The `IE` of an `e^(-)` in `eV` is numerically equal to `IP` in volts Note If a particle being accelerated by a potential has a charge equal in magnitude to the charge on an `e^(-)` the number of `eV` of energy is numerically equal to the potential in valts `(V)` . |
|
| 16. |
Select from eachgroupthe specieswhichhas thesmallestradius statingappropriate reason (a) O, O^(-) , O^(2-)(b) K^(+), Sr^(2+) , Ar ( C) Si, P,Cl |
|
Answer» (b)Although `Sr^(2+)` has Kr gasconfiguration(i.e., has four shells ) but `K^(+)` has Ar gasconfiguration (i.e., has three shells), nevertheless IONIC sizeof `Sr^(2+)` is smallerthan thatof `K^(+)` obviously due tohigher nuclear charge whichoutweighsthe effect of anadditional shell. ( c) Si P and CI and belongto 3RD period.Sinceatomicsize decrease as thenuclear chargeincrease ThereforeCI withthe HIGHEST nuclear charge hs the smallest radius. |
|
| 17. |
Select electrophiles out of the following : H^(+), Na^(+), Cl^(-), C_(2)H_(5)OH, AlCl_(3), SO_(3), CN^(-), CH_(3)CH_(2)^(+), : C Cl_(2), R-X |
|
Answer» Solution :Electrophiles are : `H^(+), AlCl_(3), SO_(3), CH_(3)CH_(2)^(+), :C Cl_(2), R-X`. In `SO_(3), O = underset(+)overset(O)overset(||)(S)-O^(-)`, S atom carries a positive charge and hence acts as an electrophile. In `AlCl_(3)`, Al atom has SIX and in : `C Cl_(2), C` atom has six electrons in the valence shell and hence each one of these needs TWO more electrons to complete their respective octets. As a result, both `AlCl_(3)` and `:C Cl_(2)` act as electrophiles. `overset(delta+)(R)-overset(delta-)(X)`, due to GREATER electronegativity of X, R carries a PARTIAL positive charge and hence acts as an electrophile. `Na^(+)` ion does not act as an electrophile even though it has +ve charge. The reason being that it has already an octet of electrons in the valence shell. Further, `Na^(+)` cannot expand its valence shell beyond 8 electrons since it does not have d-orbitals. |
|
| 18. |
Select correct statements |
|
Answer» BOND length of `overset(+)NO gt NO` |
|
| 19. |
Select correct statement(s) |
|
Answer» OXIDES and peroxides of alkali METAL are diamagentic and colourless. |
|
| 20. |
Select correct statement(s). |
|
Answer»
|
|
| 21. |
Select correct statements: |
|
Answer» Low PRESSURE is favourable for evaporation of `H_2O(I)`. |
|
| 22. |
Select correct statement for BrF_(5). |
|
Answer» All fluorine ATOMS are in same plane<BR>Four fluorine atoms and Br atom is in same plane. |
|
| 23. |
Select correct statement for AB_(n)L_(2) : [A = central atom, L = lone pair of electron A, n = number of monovalent atom B ] |
|
Answer» Molecule will be planar and NON POLAR when N = 4 |
|
| 24. |
Select correct statement |
|
Answer» `-NO_(2)" and "-COOH` GROUP DEACTIVATES benzene nucleus for ATTACK of E+ at o- and p- sites. B) `-NH_2 and -OMe` group activates benzene nucleous for attack of ET at o- and p-sites. |
|
| 25. |
Select correct statement- |
|
Answer» Methane cannot be prepared by catalytic hydrogenation of alkene (B). `CH_(3)-CH_(2)-CH_(2)-CH_(2)-CH=CH_(2)UNDERSET(NI)overset(H_(2))toCH_(3)-CH_(2)-CH_(2)-CH_(2)-CH_(2)-CH_(3)` `CH_(3)-underset(CH_(3))underset(|)(C)H-CH_(2)-CH=CH_(2)underset(Ni)overset(H_(2))toCH_(3)-CH_(3)underset(CH_(3))underset(|)(C)H-CH_(2)-CH_(2)-CH_(3)` `CH_(3)-CH_(2)-underset(CH_(3))underset(|)(C)H-CH=CH_(2)underset(Ni)overset(H_(2))toCH_(3)-CH_(2)-underset(CH_(3))underset(|)(C)H-CH_(2)-CH_(3)` `CH_(3)-underset(CH_(3))underset(|)(C)=underset(CH_(3))underset(|)(C)-CH_(3)underset(Ni)overset(H_(2))toCH_(3)-underset(CH_(3))underset(|)(C)H-underset(CH_(3))underset(|)(C)H-CH_(3)` `CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=CH_(2)underset(Ni)overset(H_(2))toCH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH_(2)-CH_(3)` (C). Neopentane can not be prepared. (D). 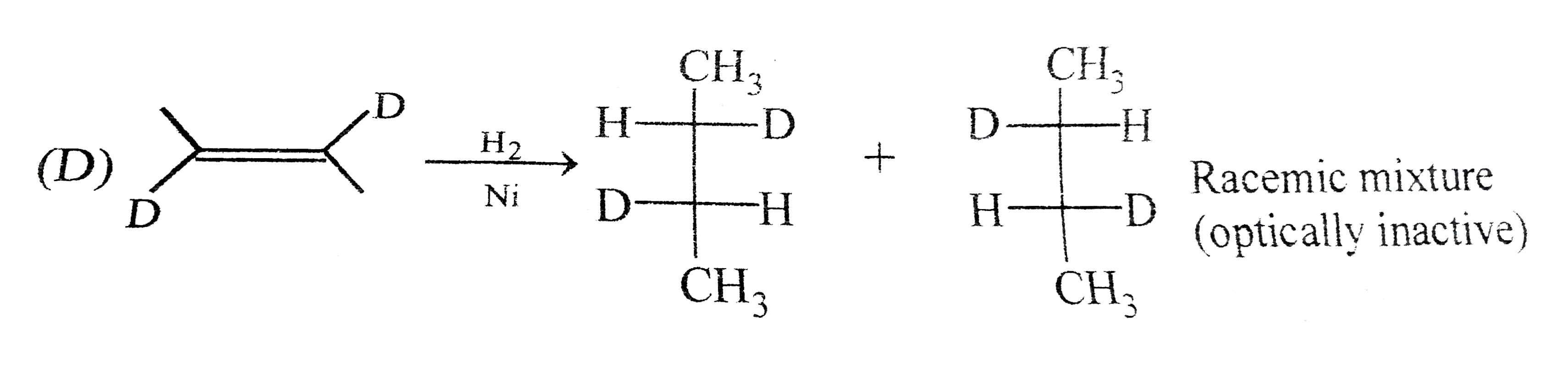
|
|
| 26. |
Select correct statememnt : |
|
Answer»
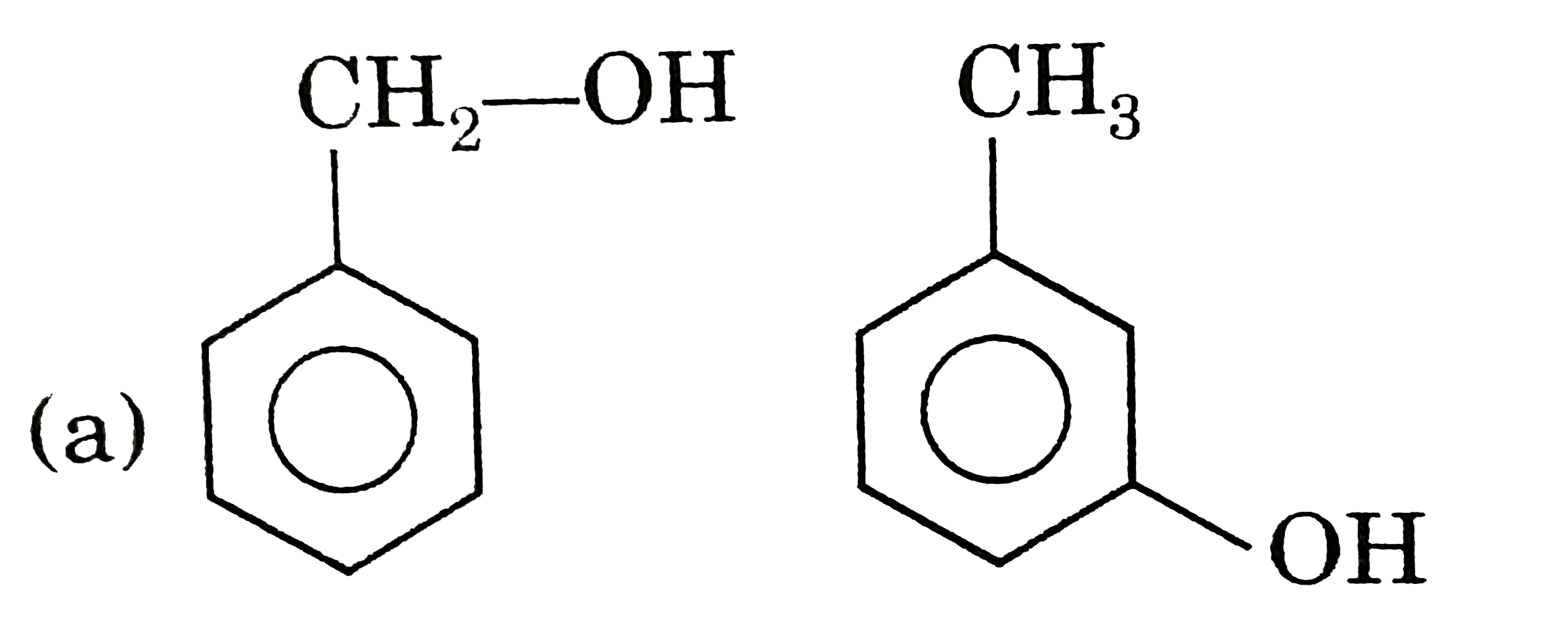 Alcojol and phenols are functioned group isomer of each other. 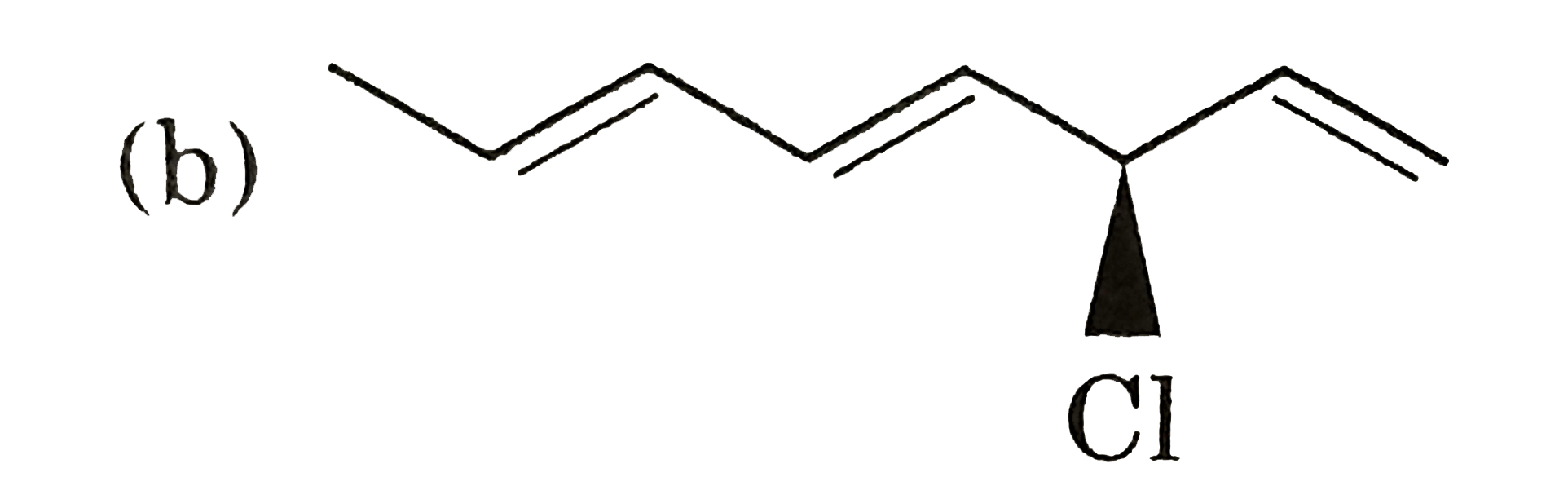 3 stereoganic AREA so, 8 steroisomers. ( c) `Me-underset(CL)underset(|)CH-underset(Cl)underset(|)CH-underset(Cl)underset(|)CH-Me` `RARR` 2 opticaacetic = 2 meso  No chiral CARBON so can t be meso. No chiral CARBON so can t be meso.
|
|
| 27. |
Select correct relation. |
|
Answer»
 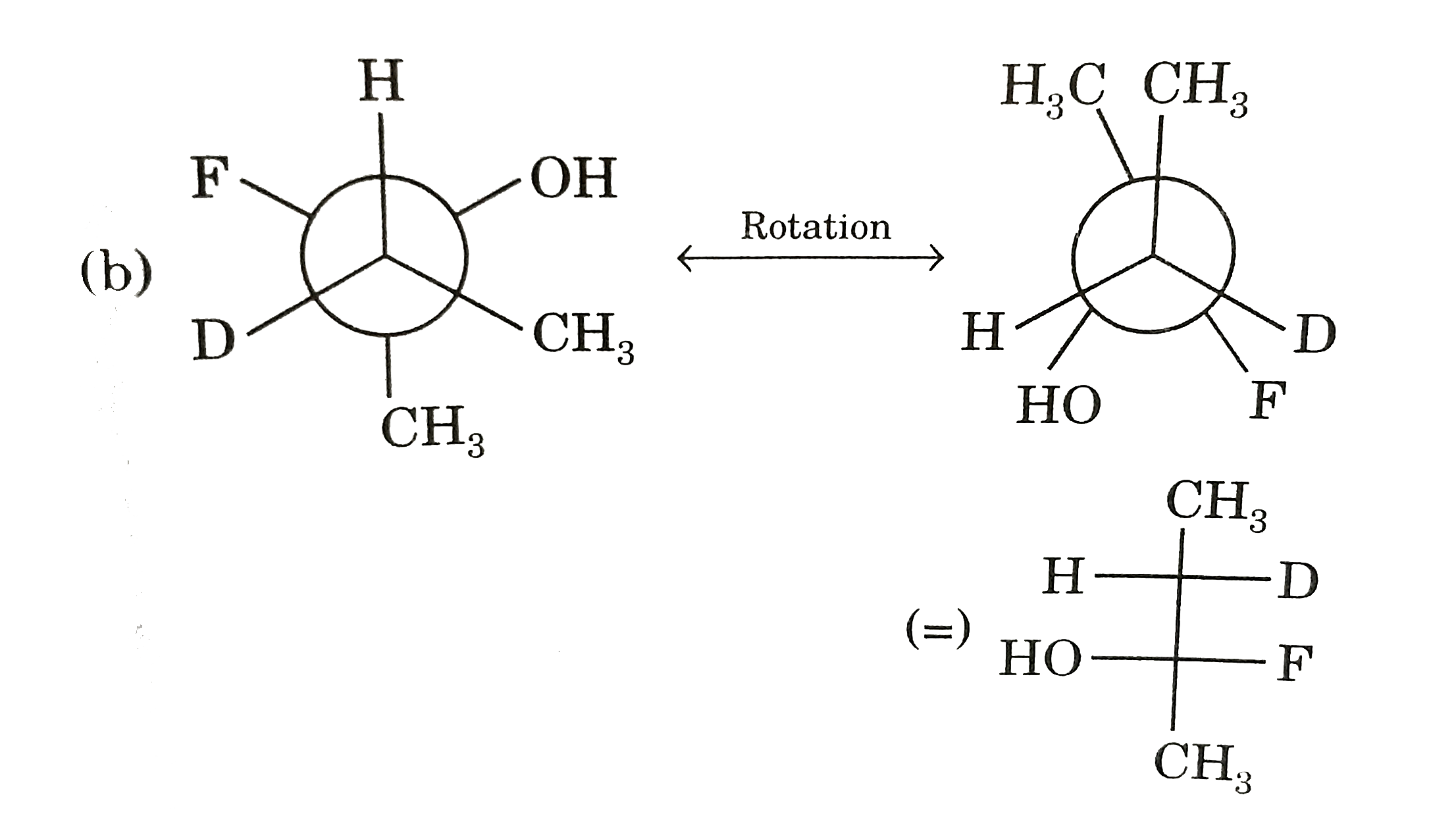 Since they differ in molecuar FORMULA by `-CH_(2)-` `therefore` They are HOMOLOGOUS. 
|
|
| 28. |
Select correct order of stabillity. |
|
Answer» 1,2-dimethycyl cylohexane`""CIS GT TRANS` |
|
| 29. |
Select correct options, for identical pairs |
| Answer» Solution :`Ph-underset(O)underset(||)(C)-NH_(2) overset(H_(2)O^(o+))rarr underset((A))(PhCOOH) overset(LiAH_(4))rarr underset((B))(Ph-CH_(2)CH)overset(KMnO_(4))rarr underset((C))(PhCOOH)` | |
| 30. |
Select correct option for true statement using symbol T and false statement using symbol F for following statements. (1) Epsom salt is chief minerals of Magnesium. (2) Gypsum is a name of minerals of Magnesium. (3) Apatite are phosphate compound. (4) Witherite is a sulphate salts of Barium. |
|
Answer» F T T F |
|
| 31. |
Select correct option for correct statement T and for false statement F for following statement. (1) Accepting the electronegativity of Lithium atom as unity, with relative other elements electronegativity is considered. (2) In fluorine compound oxidation number is + ve " and " -veboth is possible. (3) In group 14, it gives oxide of MO_(2)types (4) By increasing atomic number in periodic table always metallic character increase. |
|
Answer» TFTF |
|
| 32. |
Select correct option : |
|
Answer»
|
|
| 34. |
Select chiral molecule. |
|
Answer» MeNHEt |
|
| 35. |
Selec the correct statement(s) : |
|
Answer» Borax is made up of two triangular `BO_(3)` units and twotetrahedral `BO_(4)` units |
|
| 36. |
Segregated the following as open , closed or isolated systems. (i) Tiger (ii) The earth (iii) Tea in a thermos flask (iv) Tin in a carbonated drink (v) Hellium filled balloon. (vi) Ice cube tray filled with water. |
|
Answer» Solution :OPEN system: Tiger , the earth , ICE cube tray filled with WATER . Closesystem :HELLIUM filled in balloon, Tin of carbonated drink Isolated system : Tea in a thermos flask. |
|
| 37. |
Secondary butyl group is |
|
Answer» `CH_(3)-CH_(2)-CH_(2)-CH_(2)-` |
|
| 38. |
Second ionisation potential of oxygen is |
|
Answer» Equal to that of fluorine |
|
| 39. |
Second ioniation energy is higher than first ionisation energy for an element. This is because |
|
Answer» Nuclear charge is HIGH in CATION |
|
| 40. |
Sea water can't be used in boiler. Explain given chemical equations. |
|
Answer» Solution :Sea water contains SOLUBLE salts of calcium and magnesium, which on boiling change to insoluble salts and get deposited as a crust on the inner of a boiler. This crust acts as a bad conductor of heat and PREVENT the flow of heat to water. A large QUANTITY of fuel GETS WASTED. Chemical Reaction. `CA(HCO_(3))_(2)overset(Delta)toCaCO_(3)+CO_(2)+H_(2)O` `Mg(HCO_(3))_(2)overset(Delta)toMgCO_(3)+CO_(2)+H_(2)O` |
|
| 41. |
Screening effect is the effect produced by intervening electron between nucleus and valence electrons. They shield the nucleus from valence electron and effective nuclear charge decreases if there is less shielding effect, the effective nuclear charge decreases. Balance electrons are attracted by nucleus and repelled by other electrons. Net attractive force on electron under consideration =Z-sigma = (Nuclear charge screening effect) which is Slater's formula for screening constant. If one electron is present in outermost orbit, there will be no screening in that orbital. Each electron contributes 0.35 (total electrons minus 1) present in outermost shell. In penultimate energy level, electrons contribute 0.85. A contribution of 1.0 is from remaining electrons (present in last but one energy level). The number of lobes in most of the f-orbitals are |
|
Answer» 6 |
|
| 42. |
Screening effect is the effect produced by intervening electron between nucleus and valence electrons. They shield the nucleus from valence electron and effective nuclear charge decreases if there is less shielding effect, the effective nuclear charge decreases. Balance electrons are attracted by nucleus and repelled by other electrons. Net attractive force on electron under consideration =Z-sigma = (Nuclear charge screening effect) which is Slater's formula for screening constant. If one electron is present in outermost orbit, there will be no screening in that orbital. Each electron contributes 0.35 (total electrons minus 1) present in outermost shell. In penultimate energy level, electrons contribute 0.85. A contribution of 1.0 is from remaining electrons (present in last but one energy level). The effective nuclear charge for 4s electrons of Zn will be |
|
Answer» 26.85 |
|
| 43. |
Screeningeffect is notobserved in |
|
Answer» `He^(+)` `(1s^(1))` haveno ELECTRONS INTHE innershellsand hencescreeningeffectis notobserved. |
|
| 44. |
Screening effect is not common for the elements of the period |
|
Answer» 7 |
|
| 46. |
Screening constant depends upon A) Number of inner shells B) Number of electrons in inner shells |
|
Answer» A only |
|
| 48. |
Scientists believe that all moving objects travel in the form of wave. Then why a car moving on the road does not travel in the form of a wave ? |
| Answer» Solution :The WAVELENGTH of the WAVE is inversely PROPORTIONAL to the MASS of the moving OBJECT `(lamda = h//mv)`. As car has large mass, wavelength associated with it is negligible. | |
| 49. |
Scientific study of fermentation was first made by |
|
Answer» Buchner |
|

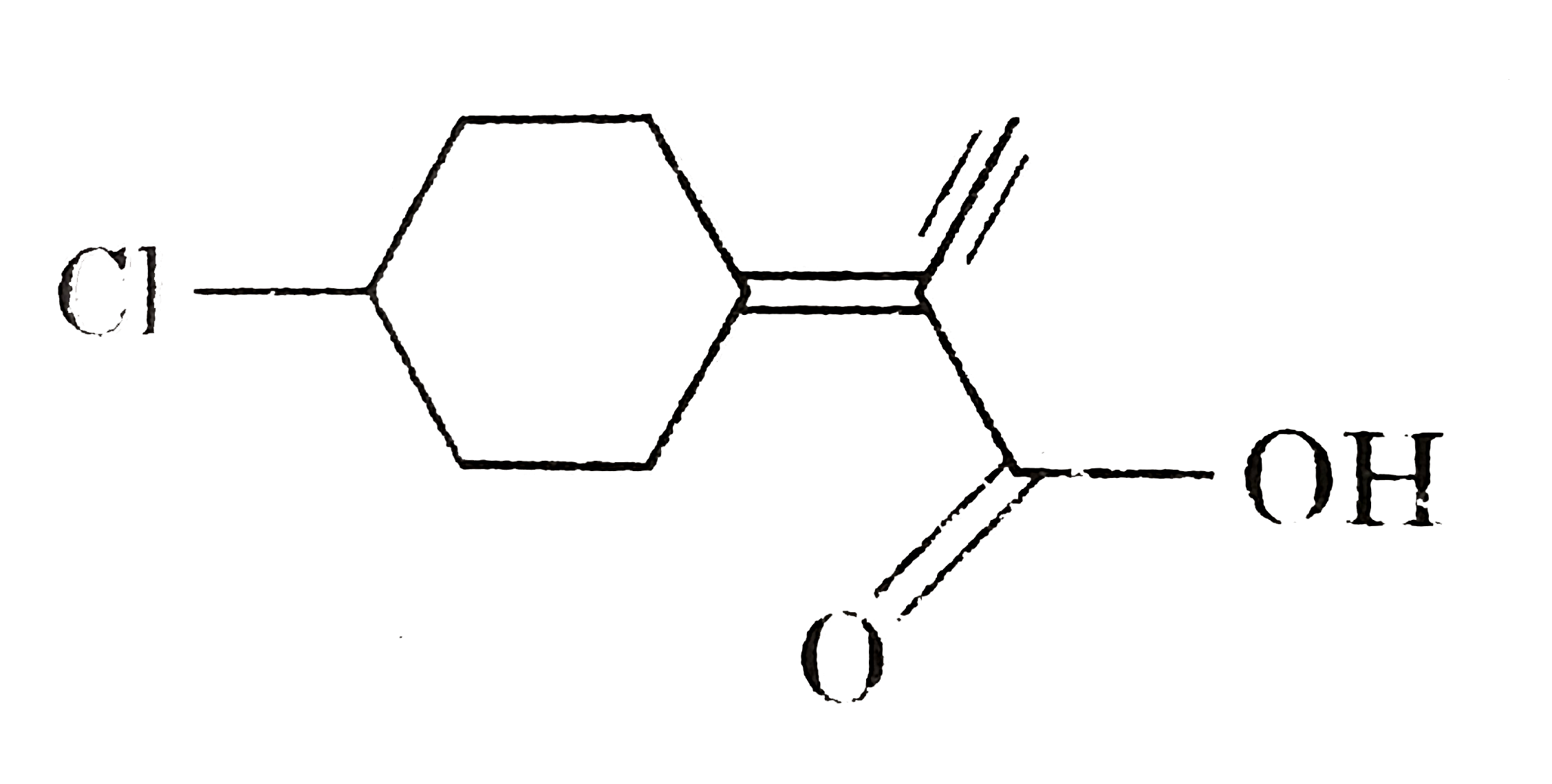
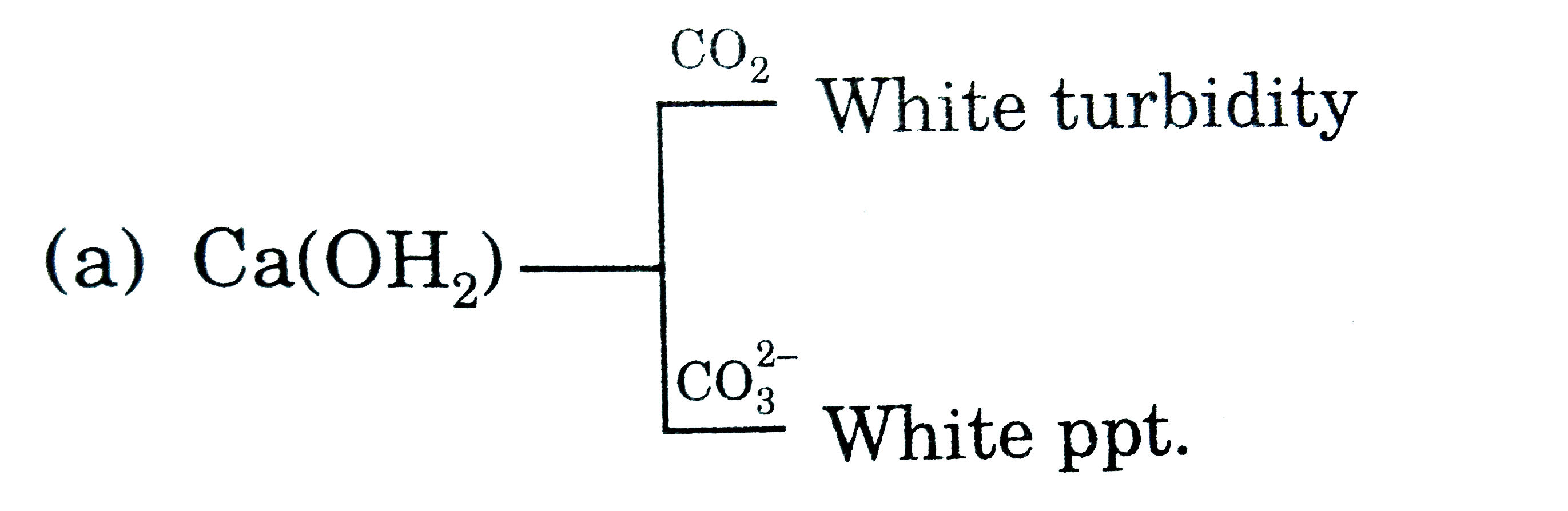
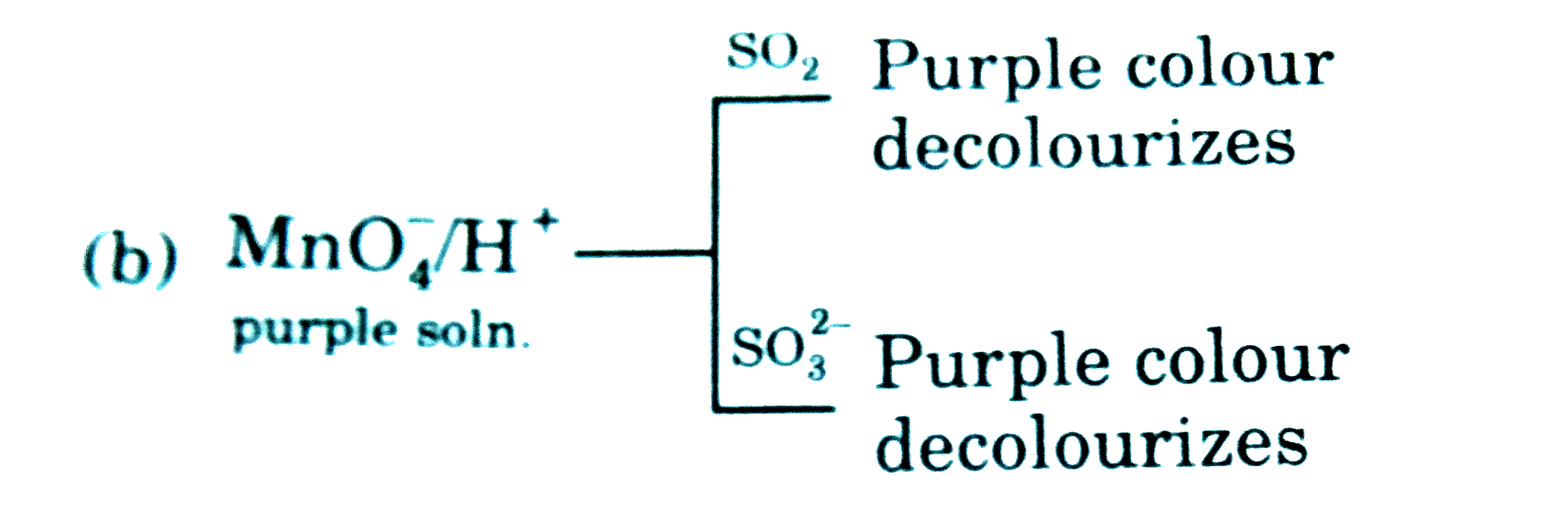


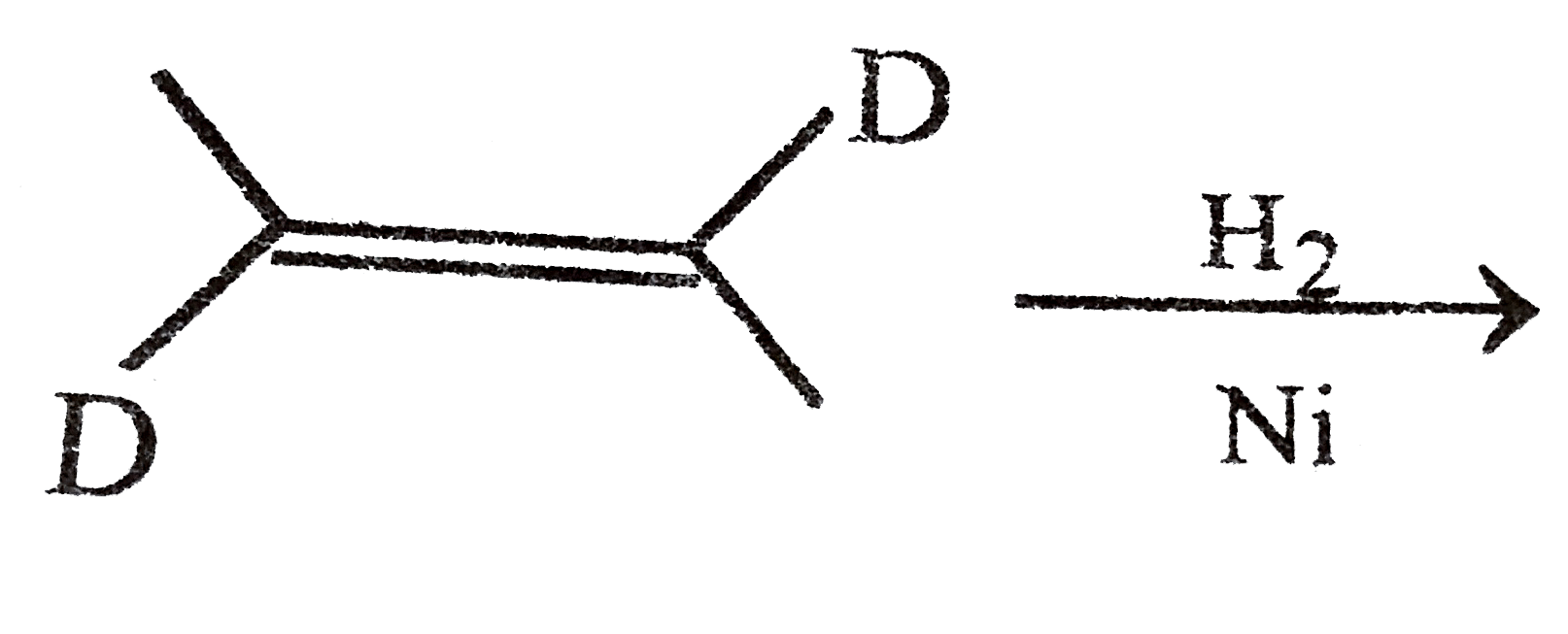 product
product