Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The answer to each of the following questions is a single digit integer from 0 to 9. If the correct answer to the question number A,B, C and D say 3,0,8,5 respectively, then the correct darkening of bubbles should be as follow. Amonst the following, the ores which are roasted to convert them into their corresponding metal oxides are: Alumina, zinc, blende, iron pyrites, copper pyrites, galena, chalcorite |
|
Answer» |
|
| 2. |
The answer to each of the following question is a single interger, ranging from 0 to 9 If the correct answers to questions, numbers A,B,Cand D say are 4,0,9 and 2, then the correct darkening of bubbles should be as shown on the side (B) In haemoglobin , CO and not O_(2) links Fe (if both CO and O_(2) are present) beacuse CO is a stronger ligand than O_(2). The number of groups which Fe is coordinated other than vacant site for CO in haemoglobin is |
|
Answer» |
|
| 3. |
The answer to each of the following question is a single interger, ranging from 0 to 9 If the correct answers to questions, numbers A,B,Cand D say are 4,0,9 and 2, then the correct darkening of bubbles should be as shown on the side (A) CO is a pollutant produced due to incomplete combindaation of butane. one mole of butane requires 6.5 moles of O_(2) for complete combustion .If 6 moles of oxygen are available , the number of CO produced will be |
|
Answer» |
|
| 4. |
The answer to each of the following question is a single interger, ranging from 0 to 9 If the correct answers to questions, numbers A,B,Cand D say are 4,0,9 and 2, then the correct darkening of bubbles should be as shown on the side (C )In measurement of BOD_(n) ,n is generally taken as |
|
Answer» |
|
| 5. |
The answer to each of the following questions is a single digit integer from 0 to 9. If the correct answer to the question number A,B, C and D say 3,0,8,5 respectively, then the correct darkening of bubbles should be as follow. How many of the following metals can be refined by the vapour phase refining? Zr, Zn, Cd, Hg, Ni, Co, Pt, Fe, Ti |
|
Answer» |
|
| 6. |
The answer of the calculation (2.568 xx 5.8)/(4.168) in significant figures will be |
|
Answer» 3.57 THUS, INSIGNIFICANT figures = 3.6 |
|
| 7. |
The anolic form of ethyl acetoacetate as shown below has |
|
Answer» 9 SIGMA BONDS and 2 pi-bonds 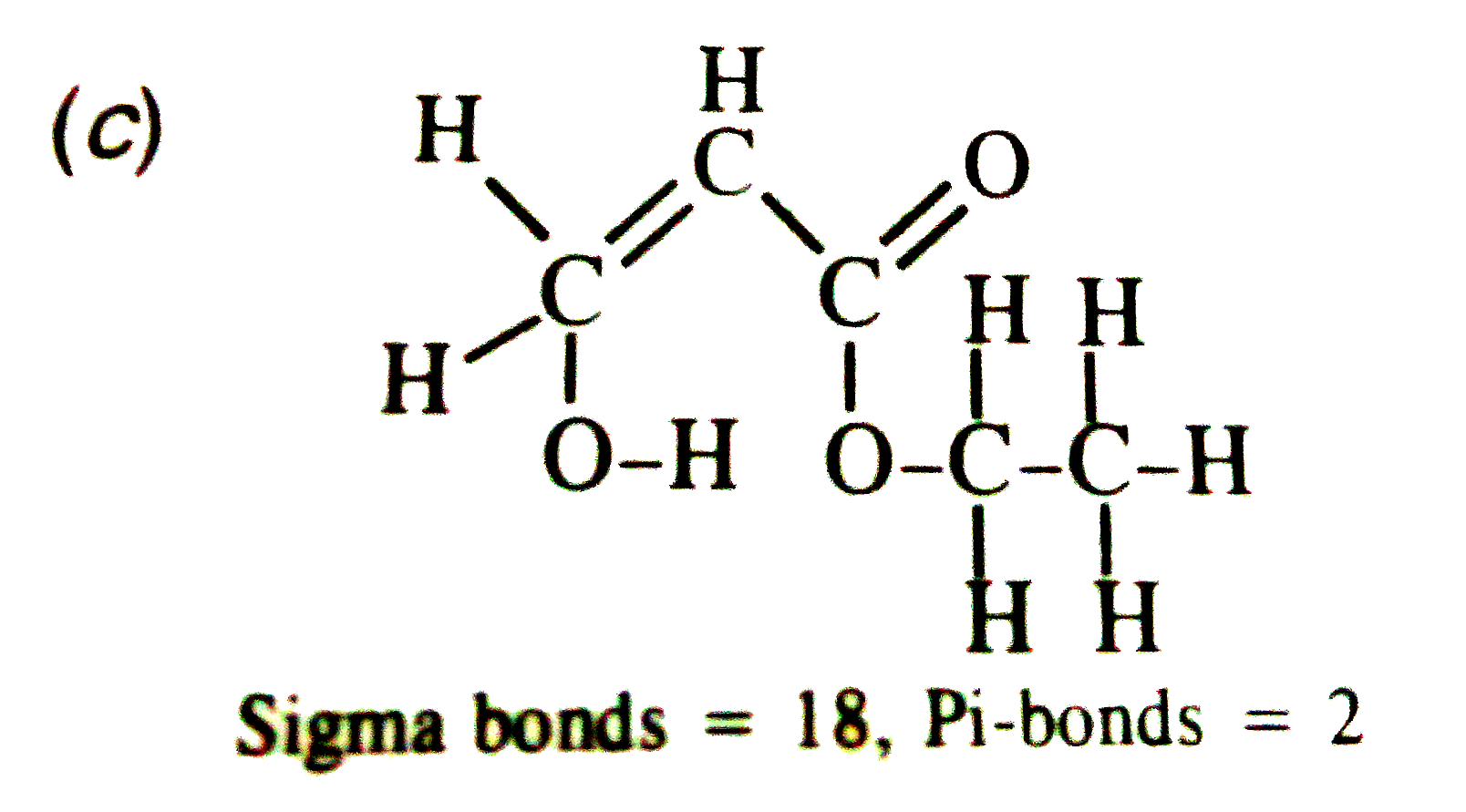
|
|
| 8. |
The anolic form of acetone contains : |
|
Answer» 9 sigma bonds, 1 pi bonds and two LONE pairs `pi`-Bonds = 1 Lonepairs = 2 |
|
| 9. |
The anion nitrate is converted into ammonium ion. The equivalent mass of nitrate ion in the reactionwould be |
|
Answer» `6.20` `E=M/8,(62)/8=7.75` |
|
| 12. |
The angular shape of ozone molecule (O_(3)) consists of : |
|
Answer» I sigma and 2 PI bonds The SHAPE of ozone molecule is In it we FIND 2 `sigma and 1 pi` bond, i.e., option (D) is correct. 
|
|
| 13. |
The angular momentum of the electron in the 4th energy shell in the hydrogen atom is ________. |
| Answer» SOLUTION :`(2H)/(PI)` | |
| 14. |
The angular momentum of electron in 'd' orbials is equal to: |
|
Answer» `sqrt2 H` Orbital angularmoment= `sqrt(l(l +1)) h` dorbitals have ` l= 2` ` :.` orbital angular MOMENTUM `= sqrt92(2 +1)h0= sqrt(6) h` |
|
| 15. |
The angular momentum of electron in 'd' orbital is equal to |
|
Answer» `2 SQRT 3 H` For d-orbital, l = 2 `:.` Angular momentum `= sqrt(2(2 + 1)) h = sqrt6 h` |
|
| 16. |
The angular momentum of an electron present in the excited state of hydrogen is 1.5h//pi. The electron is present in |
|
Answer» THIRD orbit |
|
| 17. |
The angular momentum of an electron in Bohr's orbit of hydrogen atom is 4.22 xx 10^(-34) kg m^(2) s^(-1). Calculate the wavelength of the spectral line when the electron falls from this level to the next lower level. |
|
Answer» Solution :Angular momentum `(mvr) = n (h)/(2PI) = 4.22 xx 10^(-34) kg m^(2) s^(-1)` `:. n = 4.22 xx 10^(-34) xx (2pi)/(h) = (2 xx 4.22 xx 10^(-34) xx 3.14)/(6.626 xx 10^(-34)) = 4` (Given) When the electron jumps from n = 4 to n = 3, the wavelength of the SPECTRAL line can be calculated as FOLLOWS: `(1)/(lamda) = R_(H) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) = 109, 677 cm^(-1) ((1)/(3^(2)) - (1)/(4^(2))) = 109677 xx ((1)/(9) - (1)/(16)) = 109677 xx (7)/(144) cm^(-1)` or `lamda = (144)/(109677 xx 7) cm = 1.88 xx 10^(-4) cm` |
|
| 18. |
The angular momentum of an electron is zero. In which orbital may ti be present ? |
|
Answer» 2s |
|
| 19. |
The angular momentum of a revolving electron in an orbit is equal to |
|
Answer» `(nh)/(2pi)` |
|
| 20. |
The angular m om entum of electron in ‘d ’ orbital is equal to |
|
Answer» 0h |
|
| 22. |
The angle between two covalent bonds is minimum in |
| Answer» Answer :A | |
| 23. |
The angle between the planes of H_(2)O_(2) molecule in gaseous phase is |
|
Answer» `101.5^(@)` |
|
| 24. |
The amphoteric oxide from among the following is |
|
Answer» `B_(2)O_(3)` |
|
| 25. |
The amount of zinc required to produce 224 ml of H_(2) at STP on treatment with dilute H_(2)SO_(4) will be (Zn = 65) |
| Answer» ANSWER :B | |
| 26. |
The amount of zinc required to produce 224 ml of H_(2) at NTP on treatement with dilute H_(2)SO_(4) solution will be |
|
Answer» `0.65 g` |
|
| 27. |
The amount of oxygen used by the suitable microorganisms present in water during five days at 20^(@)C is called as BOD. For pure water BOD is about 1 ppm fairly pure water 3 ppm and doubtful purity 5 ppm the municipal sewage has BOD values 100-4000 ppm. BOD= Number of moles of O_2 required / Number of liters of water sample If water is polluted its BOD value is |
| Answer» ANSWER :D | |
| 28. |
The amount of oxygen used by the suitable microorganisms present in water during five days at 20^(@)C is called as BOD. For pure water BOD is about 1 ppm fairly pure water 3 ppm and doubtful purity 5 ppm the municipal sewage has BOD values 100-4000 ppm. BOD= Number of moles of O_2 required / Number of liters of water sample BOD is normally cxpressed for a time period of |
|
Answer» 5 hours |
|
| 29. |
The amount of oxygen used by the suitable microorganisms present in water during five days at 20^(@)C is called as BOD. For pure water BOD is about 1 ppm fairly pure water 3 ppm and doubtful purity 5 ppm the municipal sewage has BOD values 100-4000 ppm. BOD= Number of moles of O_2 required / Number of liters of water sample The BOD value of municipal sewage is |
| Answer» Answer :D | |
| 30. |
The amount of oxygen used by the suitable microorganisms present in water during five days at 20^(@)C is called as BOD. For pure water BOD is about 1 ppm fairly pure water 3 ppm and doubtful purity 5 ppm the municipal sewage has BOD values 100-4000 ppm. BOD= Number of moles of O_2 required / Number of liters of water sample The BOD values of three water sample x,y and z are 5 mg/litre, 16 mg/Litre and 400 mg/Litre respectively the more polluted sample of water is |
|
Answer» x |
|
| 31. |
The amount of oxygen used by micro organisms present in water for five days at 20^@Cis called |
|
Answer» COD |
|
| 32. |
The amount of oxygen used by micro organisms present in water for five days at 20^@C is called |
| Answer» Solution :BOD | |
| 33. |
The amount of oxygen required to oxidise organic substance present in water is called |
|
Answer» DO |
|
| 34. |
The amount of oxygen consumed by pollutants in water found by titration with acidified K_2Cr_2O_7 is called _____ of water. |
|
Answer» |
|
| 35. |
The amount of oxygen required for healthy growth of plants and animals in water is |
|
Answer» 1-2 mg/mI |
|
| 36. |
The amount of oxygen (in grams) present dissolved in 10^(x) gm of water is called dissolved oxygen. Then the value of x is ____. |
|
Answer» |
|
| 37. |
The amount of (NH_(4))_(2)SO_(4) in grams which must be added to 500 ml of 0.2 M NH_(2) to yield a solution og p^(H) = 9.35 (K_(b) for NH_(3) = 1.78 xx 10^(-5)) will be |
|
Answer» `7.248 G` |
|
| 38. |
The amount of Mg in gms. to be dissolved in dilute H_(2)SO_(4) to liberate H_(2) which is just sufficient to reduce 160g of ferric oxide is |
|
Answer» 24 |
|
| 39. |
The amount of Na_(2)S_(2)O_(3) to required for reducing iodine produced by the reaction of 1 mol of KI with H_(2)O_(2) in acid medium is- |
|
Answer» 0.5 |
|
| 40. |
The amount of heat released, when 20 ml of 0.5 M NaOH is mixed with 100 mL of 0.1M HCl, is x k cal. The heat of neutralisation is |
|
Answer» `10x "K Cal mole"^(-1)` 10 m.eq `rarr` - x kJ 1 eq `rarr` ? kJ `(-x)/(10 xx 10^(-3)) = - 100x kJ`/mole |
|
| 41. |
The amount of heat exchanged with the surrounding at constant quantity ______ |
|
Answer» `DELTAE` |
|
| 42. |
The amount of heat evolved when 500cm^(3) of0.1 MHCl is mixed with 200 cm^(3) of 0.2 M NaOH is "….................." |
|
Answer» 2.292 Kj `=50` millimole`=0.050`mole `200 cm^(3)` of0.2 M NaOH `= 200 xx0.2` millimole `=4` millimole `=40`millimole `=0.040 ` mole Thus, 0.04 mole of NaOH neturalizes 0.04 mole of HCl. Heat evolved from 1 mole of HCl`= 57.3 kJ` `:.` Heat evolved from0.04mole of HCl `= 57.3xx 0.04 kJ=2.292kJ` |
|
| 43. |
The amount of heat evolved on combustion of 10 grams of benzoic acid is 10 K.Cals. The heat of combustion of benzoic acid is |
|
Answer» `-122 K.Cals` |
|
| 44. |
The amount of gas adsorbed (x/m) is the function of temperatureand pressure, which can be shown as under (x)/(m)= f(p,T) The variation x/m and p, at constant temperature is known as adsorption isotherm i.e., x/m=f(p) at constant temperature Seven type of physisorption isotherms are known. Some of them are shown below Physisorption Type I. Where there is formation of monolayer on the surface of adsorbent, this typeof curve is obtained. Physisorption Type II. The transition point 'X' of this physisorption isotherm indicates, that the formation of mono layer is complete and that of multilayer is started. Physisorption Type III. This isotherm has no transition point. In this type of curve, the multimolecular starts before the completion of monomolecular layer. On the basis of this comprehension, answer teh following question from the physisorption isotherm of a gas on a solid shown as under. The point Z of the physisorption correctly represents |
|
Answer» <P>Just beginning of formation of MULTILAYER |
|
| 45. |
The amount of gas adsorbed (x/m) is the function of temperatureand pressure, which can be shown as under (x)/(m)= f(p,T) The variation x/m and p, at constant temperature is known as adsorption isotherm i.e., x/m=f(p) at constant temperature Seven type of physisorption isotherms are known. Some of them are shown below Physisorption Type I. Where there is formation of monolayer on the surface of adsorbent, this typeof curve is obtained. Physisorption Type II. The transition point 'X' of this physisorption isotherm indicates, that the formation of mono layer is complete and that of multilayer is started. Physisorption Type III. This isotherm has no transition point. In this type of curve, the multimolecular starts before the completion of monomolecular layer. On the basis of this comprehension, answer teh following question from the physisorption isotherm of a gas on a solid shown as under. The point Y of physisorption isotherm correctly represents |
|
Answer» FORMATION of MONOLAYER |
|
| 46. |
The amount of gas adsorbed (x/m) is the function of temperatureand pressure, which can be shown as under (x)/(m)= f(p,T) The variation x/m and p, at constant temperature is known as adsorption isotherm i.e., x/m=f(p) at constant temperature Seven type of physisorption isotherms are known. Some of them are shown below Physisorption Type I. Where there is formation of monolayer on the surface of adsorbent, this typeof curve is obtained. Physisorption Type II. The transition point 'X' of this physisorption isotherm indicates, that the formation of mono layer is complete and that of multilayer is started. Physisorption Type III. This isotherm has no transition point. In this type of curve, the multimolecular starts before the completion of monomolecular layer. On the basis of this comprehension, answer teh following question from the physisorption isotherm of a gas on a solid shown as under. The portion XY of the curve correctly represents |
|
Answer» <P>x/m is DIRECTLY PROPORTIONAL to `p^(n)`, where n is greater than 1 |
|
| 47. |
The amount of gas adsorbed (x/m) is the function of temperatureand pressure, which can be shown as under (x)/(m)= f(p,T) The variation x/m and p, at constant temperature is known as adsorption isotherm i.e., x/m=f(p) at constant temperature Seven type of physisorption isotherms are known. Some of them are shown below Physisorption Type I. Where there is formation of monolayer on the surface of adsorbent, this typeof curve is obtained. Physisorption Type II. The transition point 'X' of this physisorption isotherm indicates, that the formation of mono layer is complete and that of multilayer is started. Physisorption Type III. This isotherm has no transition point. In this type of curve, the multimolecular starts before the completion of monomolecular layer. On the basis of this comprehension, answer teh following question from the physisorption isotherm of a gas on a solid shown as under. The portion ''OX'' of the physisorption isotherm represents |
|
Answer» x/m is directly proportional to p |
|
| 48. |
The amount of energy required to remove the electron from a Li^(+2) ion in its ground state is x times that of the amount of energy required to remove the electron from an H-atom in its ground state find .x. |
|
Answer» |
|
| 49. |
The amount of energy released when an electron is added to an isolated gaseous atom to produce a monovalent anion is called electron affinity or first electron affinity or electron gain enthalpy. The first electron is given a negative sign as the addition of an electron to a neutral atom is an exoergic process. The addition of electron to A^(-1) requires energy to overcome the force of repulsion. Thus, the second electron affinity is an endoergic process. The magnitude of electron affinity depends on a number of factors such as (i) atomic size (ii) effective nuclear charge (iii) screening effect (iv) half and fully filled orbitals and (v) shape of orbital. In general, electron affinity increases as the atomic radii decrease in a period. However, there are exceptions when the atoms have stable configurations. In a group, electron affinity decreases as the size increases. However, the members of 3rd period have somewhat higher values than the members in the 2nd period of the same subgroups. Which of the following species has the highest electron affinity? |
| Answer» ANSWER :D | |
| 50. |
The amount of energyreleasedwhenone millionatomsof iodine are completely converted into I^(-) ionsin thevapour stateaccordingto theequationI(g)+ e^(-)to I^(-) ( g) " is" 4.9 xx 10^(-13) J Calculatethe electronaffinity ofiodine in(i) Kj//mol and (ii) in eVper atom. |
|
Answer» Electronaffinity `=6.023 xx 10^(23) xx 4.9 xx 10^(-13) xx 10^(-6)` `=29 .5 xx 10^(4) xx 10^(-3) KJ =295 kJ mol^(-1)` `= 295// 96.49 eV //atom =3.06 eV //atom` |
|