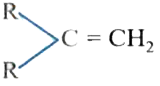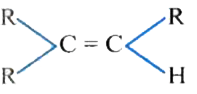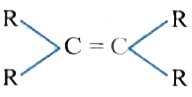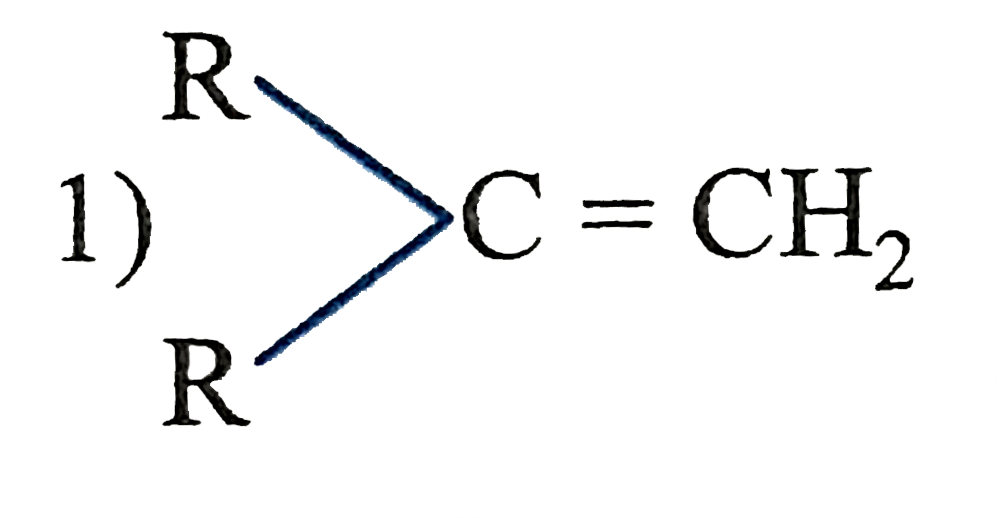Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The charge on colloidal particles is due to the ___________. |
|
Answer» adsorption of CHARGED species by disperse phase |
|
| 3. |
The charge on a cation 'M' is +2 and anion 'A' is -3. The compound formed has the formula |
|
Answer» `MA_(2)` |
|
| 4. |
The charge on cation M is +2 and anion A is -3. The compound formed has the formula |
|
Answer» `MA_(2)` |
|
| 5. |
The charge of an electron is 1.6xx10^(-19) coulombs. What will be the value of charge on Na^(+) ion |
|
Answer» `1.6xx10^(-19)C` |
|
| 6. |
the chargeof anelectronis -1.6 xx 10^(19) C.the valueof freechargeon Li^(+) ion will be |
|
Answer» `3.6 XX 10^(19) C` |
|
| 7. |
The charge balance equation of species in 0.100 M acetic acid solution is given by |
|
Answer» `[H^(+)]=[OH^(-)]` |
|
| 8. |
The charge and mass of the electron are....coulombs and kg respectively |
| Answer» SOLUTION :`1.60 XX 10^(-19), 9.11 xx 10^(-31)` | |
| 9. |
The characteristic temperature of a ferromagnetic substance above which it shows no ferromagnetism is known as ______ |
|
Answer» |
|
| 10. |
The characteristic properties of transition elements are due to |
|
Answer» UNPAIRED electron in d-subshell |
|
| 11. |
The characteristic not related to alkali metals is |
|
Answer» their ions are isoelectronic with noble GASES |
|
| 12. |
The characteristic group of secondary alcohol is : |
|
Answer» `-CH_(2)OH` |
|
| 13. |
The characteristic feature of orderly arrangement of molecules belongs to |
| Answer» Answer :D | |
| 14. |
The change of energy on freezing 1.00 kg of liquidwater at 0^(@)C and1 atmis |
|
Answer» `236 .7 kJ KG^(-1)` `:.`Enthalpy of freezing of water `= - 6.03 kJmol^(-1) `at`0^(@)C` i.e.,.Enthalpy change on freezing of 18 g ofwater at `0^(@)C =- 6.03 kJ` `:.`ENTHALPYCHANGE on freezing1000 g ofwter. `= - ( 6.03)/( 18) xx 1000 =- 335 kJ ` |
|
| 15. |
The change in velocity when electron jumps from the first orbit to the second orbit is |
|
Answer» Half its ORIGINAL VELOCITY |
|
| 16. |
The change in Gibbs free energy of the system along provides a criterion for the spontaneity of a process at constant temperature and pressure. A change in the free energy of a sytem at constant temperature and pressure will be: DeltaG_("system") = DeltaH_("system") - T DeltaS_("system") The free energy for a reaction having Delta H= 31400 cal, DeltaS= 32 cal K^(-1) mol^(-1) at 1000^(@)C is |
|
Answer» `-9336` CAL `= 31400 - 32 (1273) = - 9336` cal |
|
| 17. |
For a system in equilibrium Delta G= 0 under conditions of constant: |
|
Answer» temperature and pressure |
|
| 18. |
The change in free energy during working of pencil cell is -1372.58 cal. Calculate the equilibrium constant of the cell under standard condition (R=2 "cal mol"^(-1) K^(-1) ) |
|
Answer» `1.00 XX 10^(5)` |
|
| 19. |
The change in enthalpy when one mole ofC_("diamond") " to " C _("graphite")is called ……….. |
|
Answer» MOLAR heat of VAPORISATION |
|
| 20. |
The change in enthalpy of NaOH+HClrarrNaCl+H_(2)Ois called ……… |
|
Answer» Heat of REACTION |
|
| 21. |
The chair form is ___________ stabole than boat form by potential energy _______kJ//mol. |
|
Answer» More, `44 kJ//mol` |
|
| 22. |
The CGS unit of dipolemoment is …………. Whereas its SIunit is ………….. |
| Answer» SOLUTION :DEBYE, COULOMB METRE (C m) | |
| 23. |
The centre of the Sun consists of gases whose average molecular weight is 2 . If the density of the gases is 2.73 xx 10^(3) kg/ m^(3) at a pressure of 1.12 xx 10^(9) atm , the temperature at the centre of the Sun is (assuming ideal behaviour) |
| Answer» Answer :D | |
| 24. |
The central carbon atom in dichlorocarbene contains_______valence electrons. |
|
Answer» 6 |
|
| 25. |
The central C-atom of a carbanion possesses |
|
Answer» sectet of electrons |
|
| 27. |
The cell constant of a given cell is 0.47 cm^(-1) . The resistance of a solution placed in this cell is measured to be 3.16 ohm . The conductivity of the solution (inA cm^(-1) where S has usual meaning ) is |
|
Answer» 0.15 |
|
| 28. |
The C—C bond length is maximum in :(a)C_(60)(b)diamond(c)C_(70)(d)All of these |
| Answer» Solution :diamond | |
| 29. |
The cause of permanent hardness of water is due to |
|
Answer» `Ca(HCO_(3))_(2)` |
|
| 30. |
The cause of cis-trans isomerism is |
|
Answer» strength of the double bond |
|
| 31. |
The cause of Brownian movement is |
|
Answer» heat changes in liquid state |
|
| 32. |
The cation which gives a yellow precipitate with potassium chromate is |
|
Answer» `NH_(4)^(+)` `K_(2)CrO_(4) +Ba^(2+) to BaCrO_(4) +2K^(+)` |
|
| 34. |
The cathode in middle compartment of Castner-Kellner process is |
|
Answer» Graphite |
|
| 35. |
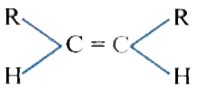
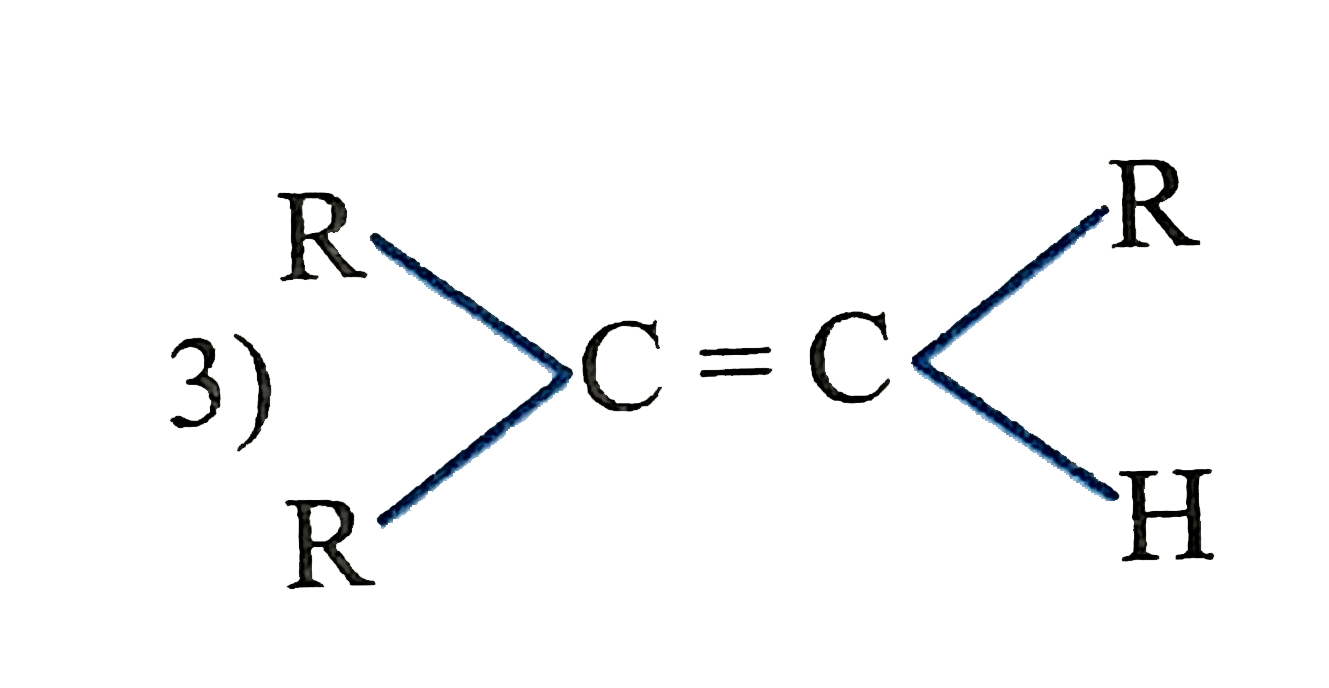
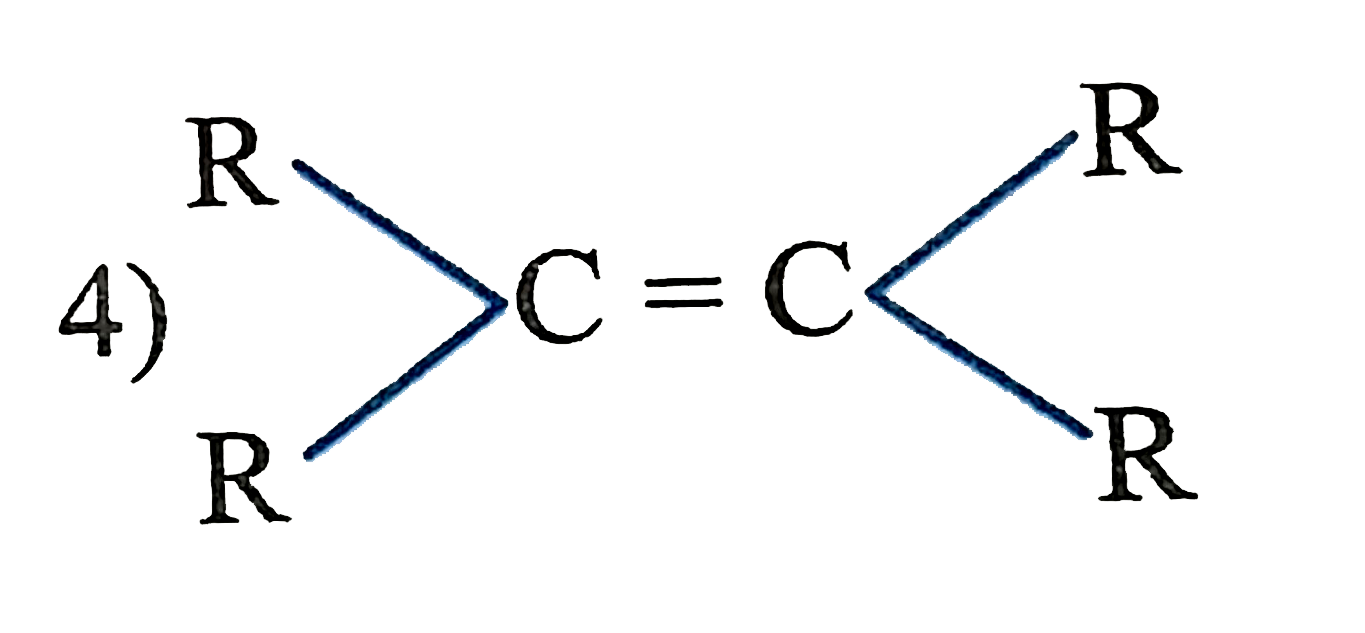
The catalytic hydrogentaion more easier in case of which alkene? |
|
Answer»
|
|
| 37. |
The catalyst used in the preparation of C Cl_2F_2 from C Cl_4 and HF is ……………….. . |
| Answer» SOLUTION :`SbCl_5` | |
| 38. |
The catalyst used in the preparation of chlorobenzene from benzene is …………….. . |
| Answer» SOLUTION :`FeCl_3` | |
| 39. |
The catalyst used in the manufacture of methanol from water gas is:- |
| Answer» Solution :`[CO+H_(2)]+H_(2)overset(ZnO+Cr_(2)O_(3))toCH_(3)OH`. | |
| 42. |
The catalyst used for obtaining carbonyl compound from alkyne during hydration is ….. |
| Answer» Answer :B | |
| 43. |
The carboxyl functional group (-COOH) is present in |
|
Answer» PICRIC acid |
|
| 44. |
The carbonate which decomposes on heating is |
|
Answer» `Na_(2)CO_(3)` |
|
| 45. |
The carbonate that will not decompose on heating is |
|
Answer» `Na_(2)CO_(3)` |
|
| 46. |
The carbonate having high decomposition temperature among the following is |
|
Answer» `MgCO_(3)` |
|
| 47. |
The carbon-hydrogen bonds in benzene are |
|
Answer» `SIGMA SP^2 - s` |
|
| 49. |
The carbon-carbon bonds in benzene are |
|
Answer» `SIGMA SP^3 - sp` |
|