Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number of unpaired electrons in Fe^3+ (Z=26) are |
|
Answer» 0 
|
|
| 2. |
The number of 3 centered, 2 electron bonds in doborane is |
|
Answer» 2 
|
|
| 3. |
The number of 2p electrons having spin quantum number s = -1//2 are |
|
Answer» 6 |
|
| 4. |
The number of three centred, 2 electron bonds in diborane is |
|
Answer» 2 |
|
| 5. |
The number of 2-centre-2-electron and 3-centre-2-electron bonds in B_2H_6, respectively, are..... |
Answer» SOLUTION :
|
|
| 7. |
The number and types of bonds between two carbon atoms in calcium carbide are |
|
Answer» one SIGMA , one pi |
|
| 8. |
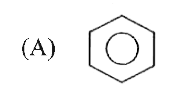
The number and type of bonds between two carbon atoms in CaC_(2) are |
|
Answer» one `SIGMA` and one `PI` BOND  one `sigma` and two `pi` bonds. one `sigma` and two `pi` bonds.
|
|
| 9. |
The num ber of protons in nucleus of an elem ent having maximum unpaired electrons in 2p sub-shell are ............. |
|
Answer» 7 N (He) `2p^(3)` OR [He]  N(Z=7) its has protons =7 ELECTRONS =7 |
|
| 10. |
The nucleus of tritium consists of |
|
Answer» 1 PROTON +1 neutron |
|
| 11. |
the nucleus of an element contain9protons itsvalencywould be |
|
Answer» `-1` |
|
| 12. |
The nucleus of an atom is located at x = y = z = 0. If the probability of finding an s orbital electron in a tiny volume around x=a, y=z=0 is 1. xx 10^(-5), choose the correct statements(s) regarding the probability of electron |
|
Answer» The probability of finding the electron in the same-sized VOLUME AROUND x = Z=0, y=a is the same i.e., `1 XX 10^(-5)` |
|
| 13. |
The nucleus of an atom contains |
|
Answer» ELECTRONS and protons |
|
| 14. |
The nucleus of an atom can be assumed to be spherical. The radius of the nucleus of mass number A is given by 1.25xx10^(-13) xx A^(1//3) cm Radius of atom is one Å. If the mass number is 64, Then the fraction of the atomic volume that is occupied by the nucleus is |
|
Answer» `1.0xx10^(-3)` `=1.25xx10^(-13) xx 64^(1//3) =5xx10^(-13)` cm Radius of ATOM =1Å=`10^(-8)` cm `"Volume of nucleus"/"Volume of atom"=((4//3)pi(5xx10^(-13))^3)/((4//3)pi(10^(-8))^3)` `=1.25xx10^(-13)` |
|
| 15. |
The nucles of an atom contains: |
|
Answer» ELECTRONS and PROTONS |
|
| 17. |
The nuclear radius is of the order of 10^(-13) cm while atomic radius is of the order 10^(-8) cm. Assuming the nucleus and the atom to be spherical, what fraction of the atomic volume occupied by the nucleus ? |
|
Answer» Solution :The volume of a sphere `= 4pi r^(3)//3` where r is the RADIUS of the sphere `:.` Volume of the NUCLEUS `= 4pi r^(3)//3 = 4pi (10^(-13))^(3)//3 cm^(3)` SIMILARLY, Volume of the atom `= 4pi r^(3)//3 = 4pi (10^(-8))^(3)//3 cm^(3)` `:.` Fraction of the volume of atom occupied by the nucleus `= (4pi (10^(-13))^(3)//3 cm^(3))/(4pi (10^(-8))^(3)//3) cm^(3) = 10^(-15)` |
|
| 18. |
The nuclei of tworadioactive isotopes of same substanceA^(236) and B^(234) are present in the ratio of 4:1 in an ore obtained form some other planet. Their half-lives are 30 and60 minutes respectibvely. Both isotopes are alpha emitters and acitity of isotopeA^(236) si 1 rutherford(10^(6) dps). Calcualte: (a) After how much time their activities will becomes identical? (b) The time required tobring the ratio of their atoms to 1:1 |
|
Answer» |
|
| 19. |
The normality of the mixture of HCl and H_(2)SO_(4) solution is N//5 . 0.287 g of AgCl is obtained when 20 mL of this solution is treated with excess of AgNO_(3). Calculate the percentage of both the acids in the mixture. |
|
Answer» |
|
| 20. |
The nuclear cross section is measured in barn'. It is equal to |
|
Answer» `10^(-20)m^(2)` |
|
| 21. |
The normality of aqueous solution of 1 M H_(3)PO_(4)is ........... |
|
Answer» 1.5 N `= 1 xx 3 = 3N` |
|
| 22. |
The normality of a 26 % "mass"//"volume" solution of ammonia (density 0.885 g//mL) is approximately : |
|
Answer» 1.5 |
|
| 23. |
The normality of 2.24 vol H_2O_2 is |
|
Answer» 1.786 |
|
| 24. |
The normality of 10 volume H_(2)O_(2) is : |
|
Answer» 0.176 |
|
| 25. |
The normality of 10 % ("mass"//"volume") acetic acid is : |
| Answer» Solution :N//A | |
| 26. |
The normality of 0.3 M phosphorous acid (H_(3) PO_(3)) is |
|
Answer» 0.1 |
|
| 27. |
The normality of 0.3 M phosphorous acid (H_(3)PO_(3)) is : |
|
Answer» 0.1 |
|
| 28. |
The normality of 0.3 H_3PO_3 is |
|
Answer» 0.1 n = 2 , `N = M xx n = 0.3 xx 2 = 0.6` |
|
| 30. |
The normal boiling point of the liquid is approximately |
|
Answer» 1.55 TIMES of its CRITICAL temperture |
|
| 31. |
The normal boiling point of a liquid 'A' is 350K. DeltaH_("vap") at normal boiling point is 35 kJ/mole. Pick out the correct statement(s). (Assume DeltaH_("vap") to be independent of pressure) |
|
Answer» `Delta S_("VAPORISATION") gt 100 J//K` mole at 350K and 0.5atm |
|
| 32. |
The no.of intermediate formed in the following conversion is |
Answer»  
|
|
| 33. |
The non-viable particulate is among the following is |
| Answer» Solution :Dust | |
| 34. |
The non metal which occur only in the oxidised form in nature is |
|
Answer» carbon |
|
| 35. |
The non planar molecule among the following is |
|
Answer» `B_2 H_6` |
|
| 36. |
The non metal which is generally occur in the oxidised form in nature is |
|
Answer» C |
|
| 38. |
The non-existence of PbI_(4) is due to |
|
Answer» HIGHLY oxidising NATURE of `Pb^(4+)` |
|
| 39. |
The non aromatic compound among the following is - |
|
Answer»
|
|
| 40. |
The non aromatic compound among the following is : |
|
Answer»
|
|
| 41. |
The non-aromatic compound among the following |
|
Answer»
|
|
| 42. |
The nodal plane in the pi-bond of ethene is located in |
|
Answer» the MOLECULAR plane |
|
| 43. |
The non-aromatic compound among the following is |
|
Answer»
|
|
| 44. |
The nodal plane in the -bond of ethene is located in |
|
Answer» The molecular PLANE |
|
| 45. |
The noble gases are stable due to their __________ outer shell electronic configuration . |
|
Answer» pantly FILLED |
|
| 46. |
The -NO_(2) group in nitrobenzene: (i) In resonance act as a electron donating and in inductive effect it act as an electron attracting. (ii) It acts as an electron donating in resonance as well as inductive effect. (iii) It acts as an electron attractive in resonance as well as inductive effect. (iv) It acts as an electron attracting in resonance and electron donating in inductive effect |
| Answer» SOLUTION :(i-F),(ii-F), (iii-T), (iv-F) | |
| 47. |
The no. of possible isomers of C_(4)H_(10)O is…. |
|
Answer» 7 (ii) `underset("ethoxy ethane")(CH_(3)-CH_(2)-O-CH_(2)-CH_(3))` (iii) `underset("methyl isopropyl ether")((CH_(3))_(2) -CH-O-CH_(3))` (IV) `underset("1-butanol")(CH_(3)-CH_(2)-CH_(2)-CH_(2)-OH)` (v) `underset("2-butanol")(CH_(3)-CH_(2)- underset(underset(OH)(|))(CH)-CH_(3))` (VI) `underset("2-methyl-propanol")(CH_(3)-underset(underset(CH_(3))(|))(CH)-CH_(2)-OH)` (VII) `underset("2-methyl 2-propaol")(CH_(3)-underset(underset(CH_(3))(|))overset(overset(CH_(3))(|))(C )-CH_(3))` |
|
| 48. |
The no of mono substituted derivatives of pyridine is ________ |
Answer» 
|
|
| 49. |
The no of moles of CO_2, formed during the oxidation of CH_2 = CH - CH = CH_2 with acidified KMnO_4 is |
|
Answer» |
|
| 50. |
The no. of isomers for the compound with molecular formula C_(2)BrClFl is….. |
|
Answer» 3 
|
|