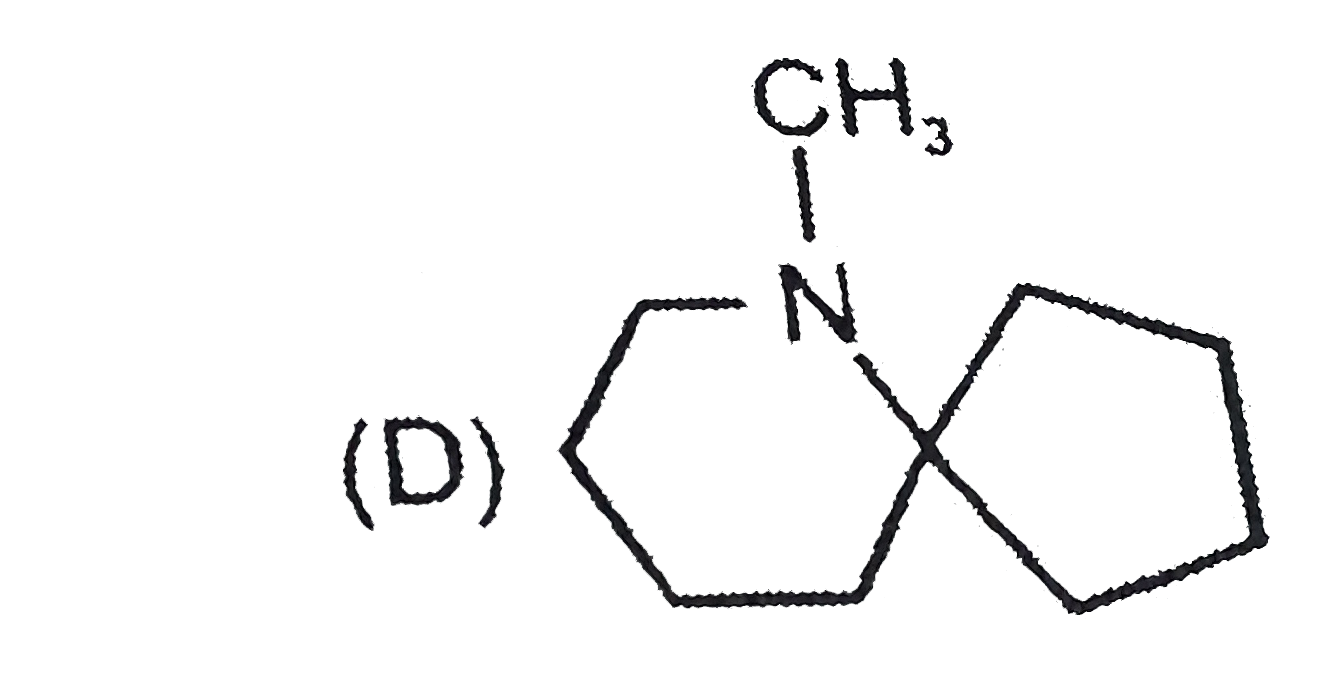Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The no.of hydroxyl ions produced by one molecule of Na_(2),CO_(3) , on hydrolysis is |
|
Answer» 4 |
|
| 2. |
The no. of electrons satisfying n+l=7 for Lu (atomic number:71) |
|
Answer» |
|
| 3. |
The no. of amides having formula C_4H_9NOare : |
|
Answer» 5 `C-C-overset(O)overset(||)C-NH-C""H-overset(O)overset(||)C-underset( C )underset(|)N-C-C` `C-overset(O)overset(||)C--NH-C-C""C-underset( C )underset(|)C-overset(O)overset(||)C-NH_(2)` `H-overset(O)overset(||)C-NH-C-C-C""H-overset(O)overset(||)C-NH-underset(C)underset(|)C-C` |
|
| 4. |
The no. of amides having formula C_(3)H_(7)NO each of which is capable of forming hydrogen bonding are : |
|
Answer» 1 
|
|
| 5. |
The number of lone pair of electrons on C-atom present in CO_(2) are ……………… |
|
Answer» `SeF_(4),XeO_(2)F_(2)` T-shaped, one lone pair on CENTRAL ATOM. |
|
| 6. |
C_(40) H_(56) is the empirical formula of |
|
Answer» 1 `H-overset(O)overset(||)C-NH-C-C""H-overset(O)overset(||)C-overset(CH_(3))overset(|)N-CH_(3)` (In this case hydrogen bond FORMATION not possible due to sterice FACTOR) |
|
| 7. |
The nitrogen oxide(s) that contains N-N bond(s) is (are) |
|
Answer» `N_(2)O` |
|
| 8. |
The nitrogen is converted into which form in Kjeldhl's method? |
|
Answer» `NH_(3)` |
|
| 9. |
The nitrogen containing compound produced in the reaction of HNO_(3) with P_(4)O_(10) |
|
Answer» can be prepared by reaction of `P_(4)andHNO_(3)` `N_(2)O_(5)` cannot be prepared by action of `HNO_(3)` on `P_(4)` but gives `H_(3)PO_(4)` `P_(4)+10HNO_(3)to4H_(3)PO_(4)+20NO_(2)+4H_(2)O` `N_(2)O_(5)` has 54 electrons `(2xx7+5xx8)` and hence is diamagnetic `N_(2)O_(5)` does not CONTAIN N-N bond. REFER to structure is Table 11.14, page 11//120. `N_(2)O_(5)` reacts with Na producing `NO_(2)` `Na+N_(2)O_(5)toNaNO_(3)+NO_(2)` (brown gas) Thus, option (b) and (d) are correct. |
|
| 10. |
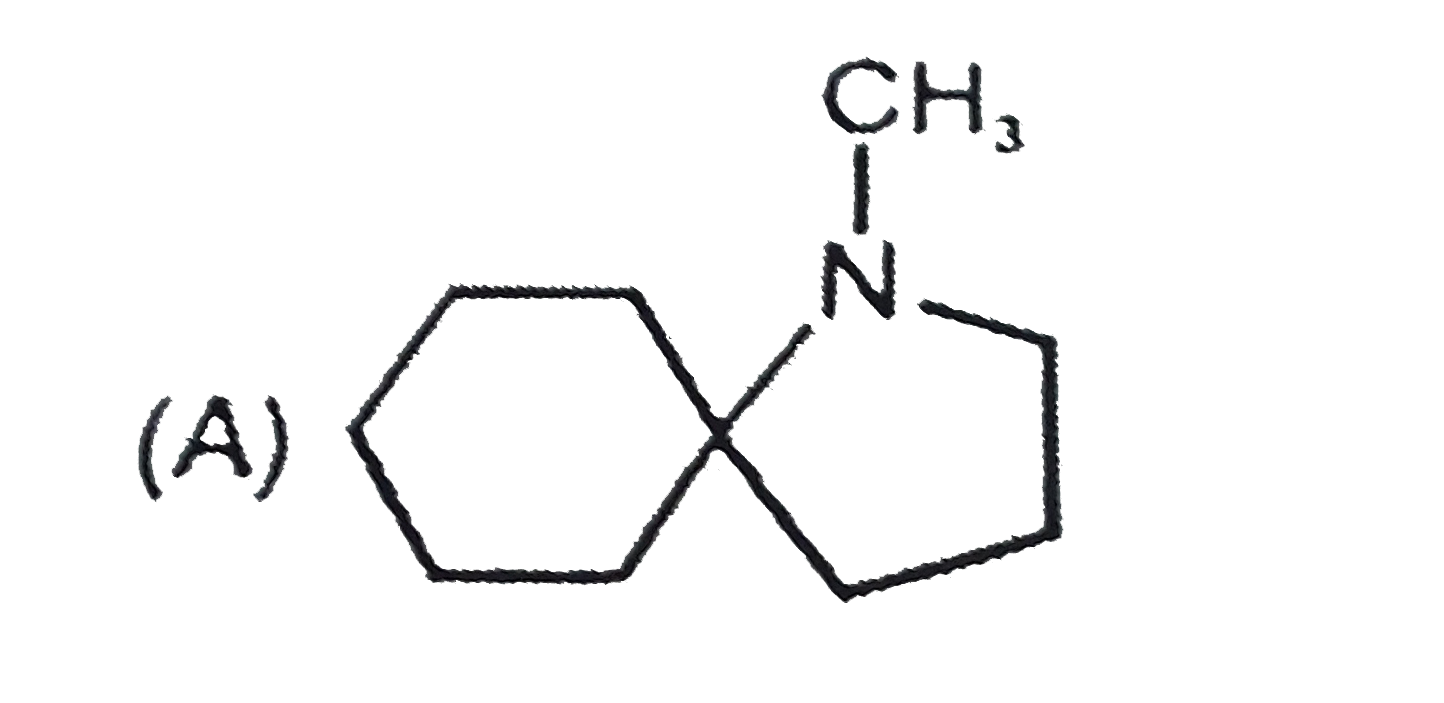
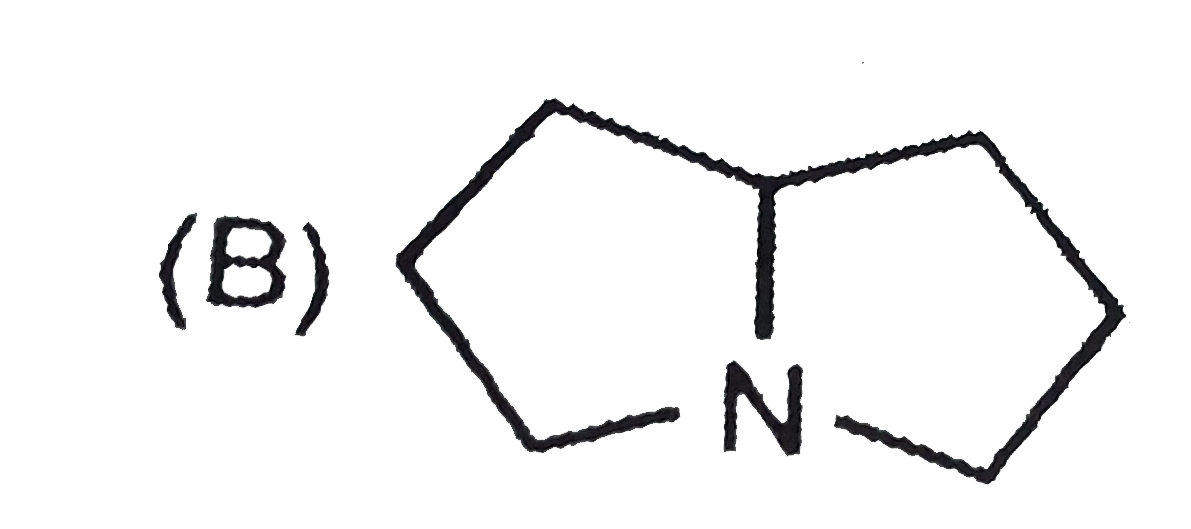
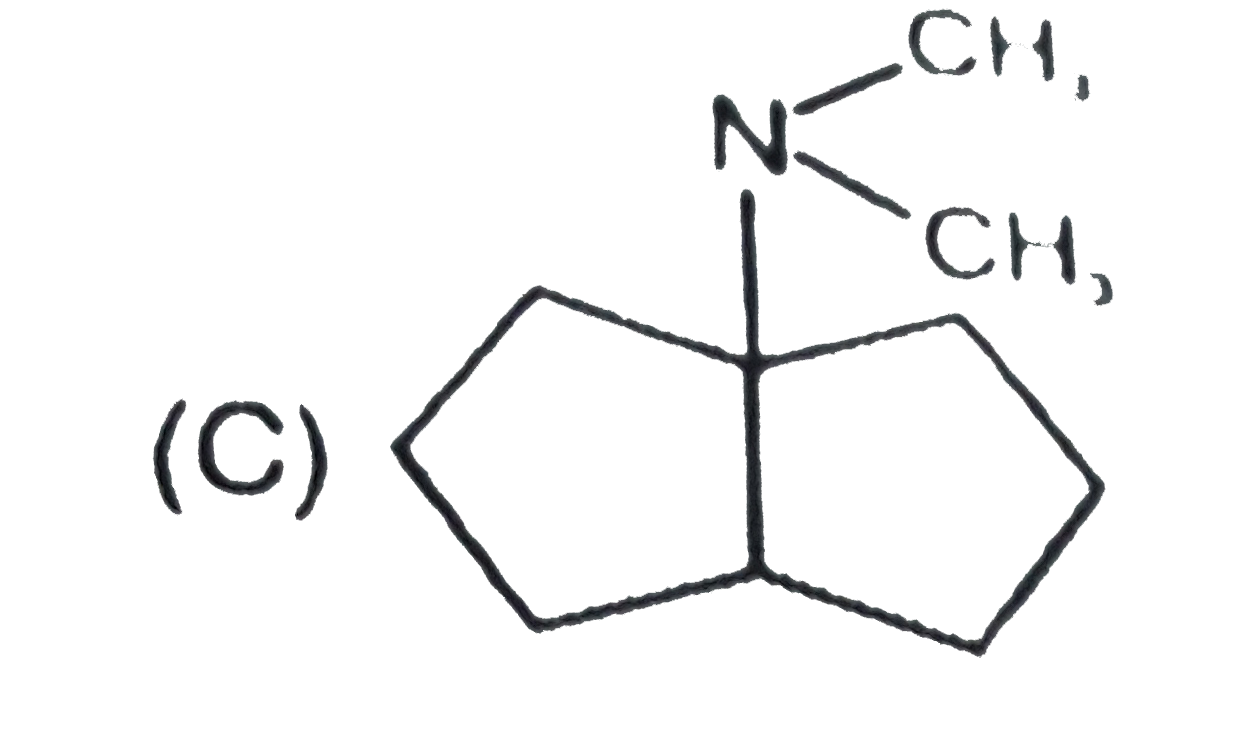
The nitrogen atom in the following cyclic compounds can be removed as trimethylamine by successive Hoffmann eliminations (involving exhaustive methylation followed by heating with AgOH). The amine which will require a greater number of Hoffmann eliminations is: |
|
Answer»
|
|
| 11. |
The nitro group is a very strong deactivating group Nitro benzene undergoes nitration at a rate only 10-4 times that of benzene. The nitro group is a meta-directing group. When nitrobenzene is nitratedwith nitration mixture 93% of meta substituted product is formed. Ring deactivating groups are : |
|
Answer» Ortho-PARA directing GENERALLY |
|
| 12. |
The nitro group is a very strong deactivating group Nitro benzene undergoes nitration at a rate only 10-4 times that of benzene. The nitro group is a meta-directing group. When nitrobenzene is nitratedwith nitration mixture 93% of meta substituted product is formed. When benzene is nitrated then Nitrobenzene is obtained and when nitro benzene is nitrated then the product which is 7% of the yield is_______ |
|
Answer»
|
|
| 13. |
The nitro group is a very strong deactivating group Nitro benzene undergoes nitration at a rate only 10-4 times that of benzene. The nitro group is a meta-directing group. When nitrobenzene is nitratedwith nitration mixture 93% of meta substituted product is formed. Ortho-para directing groups are |
|
Answer» <P>`-NO_2 , -CN` |
|
| 14. |
The nitro group in the benzene nucleus is ………. Directing. It ……………. The reactivity of the benzene ring. |
|
Answer» |
|
| 15. |
The nitration ofhigheralkanesinvolvessubstitutionas well as _______. |
|
Answer» C-H bondfissions |
|
| 16. |
We consider a thermodynamic system. If DeltaU represent the increase in the its energy and W the work done by the system, which of the following statements is true? |
|
Answer» <P> SOLUTION :`- W P DELTAV` |
|
| 17. |
The net enthalpy change of a reaction is the amount of energy required to break all the bonds in reactant molecules minus amount of energy required to form all the bonds in the product molecules. What will be the enthalpy change for the following reaction. H_(2(g)) + Br_(2(g)) to 2HBr_((g)). Given that Bond energy of H_(2), Br_(2) and HBr is 435 "kJ mol"^(-1), 192 "kJ mol"^(-1) and 368 "kJ mol"^(-1) respectively. |
|
Answer» Solution :bond energy of `H_(2) = 435 "kJ mol"^(-1)` bond energy of `Br_(2) = 192 "kJ mol"^(-1)` bond energy of HBr `= 368 "kJ mol"^(-1)` For the reaction `H_(2(g)) + Br_(2(g)) to 2HBr_((g))` `Delta_(r) H^( THETA ) = sum BE ("Reactants") - sumBE ("products")` `= BE (H_(2) ) + BE(Br_2) - 2BE (HBr)` `= 435 + 192 - (2XX 368) "kJ mol"^(-1)` `= -109 "kJ mol"^(-1)` |
|
| 18. |
The net enthalpy change of a reaction is the amount of energy required to break all the bonds in reactant molecules minus amount of energy required to form all the bonds in the product molecules. What will be the enthalpy change for the following reaction ? H_(2) (g) + Br_(2) (g) rarr 2HBr (g). Given that, bond energy of H_(2), Br_(2) and HBr is 435 kJ mol^(-1), 192 kJ mol^(-1) and 368 kJ mol^(-1) respectively |
|
Answer» Solution :Given that bond energy of `H_(2) = 435 kJ mol^(-1)` bond energy of `Br_(2) = 192 kJ mol^(-1)` bond energy of `HBr = 368 kJ mol^(-1)` For the REACTION `H_(2) (g) + Br_(2) (g) RARR 2HBr (g)` `Delta_(r) H^(Θ) = SigmaBE ("Reactants") - SigmaBE ("Products")` `= BE (H_(2)) + BE (Br_(2)) - 2BE (HBr)` `= 435 + 192 - (2 XX 368) kJ mol^(-1)` `= - 109 kJ mol^(-1)` |
|
| 19. |
The net enthalpy change of a reaction is the amount of energy required to break all the bonds an reactant molecules amount of energyrequiredto form all the bonds in the product molecules. What will be theenthalpy change for the following reaction : H_(2)(g) + Br_(2) rarr 2HBr(g) Given that bond energy of H_(2), Br_(2) and HBr is435 kJ mol^(-1) , 192 kJ mol^(-1) and 368 kJ mol^(-1) respectively. |
|
Answer» Solution :`H_(2)(G) +Br_(2)(g) rarr 2HBR(g)` `Delta_(r)H^(@) = SigmaBE` ( Reactants)`- Sigma `BE( Products)`= BE ( H_(2)) + BE ( Br_(2)) -2` BE ( HBr) `= 435 +192- ( 2 XX 368 ) KJ mol^(-1)` `=- 109 kJ mol^(-1)` |
|
| 20. |
The neopentyl halide in ethanol yields alkenes by E_(1) mechanism due to |
|
Answer» low CONCENTRATION of solvent |
|
| 21. |
The necessary condition for a molecule to exhibit optical isomerism is dissymmetry or chiral. Thus all organic compounds which contain one assymetric carbon atom are chiral and exist in two stereoisomers. Although the two forms have the same structure, they have different arrangements of groups about the assymmetric carbon. In fact, they represent assymetric molecules. They do not have a plane of symmetry. They are related to each other as an object to its miror image and are non superimposable. The two structures actually stand for dextro or (+) and leavo or (-) isomers. Since they are related each other as mirror images, they are commonly called enantiomers. Which of the following compounds is optically inactive |
|
Answer»

|
|
| 22. |
The Ne atom has 10 times the mass of H_2. Which of the following statements is true? I) At 25^@C the both have the same kinetic energy II) Ten moles of H_2 would have the same volume as 1 mole of Ne at same temperature and pressure III) One mole of Ne exerts the same pressure as one mole of H_2 at STP. IV) AH_2 molecule travels 10 times faster than Ne atom at same temperature. V) At STP, one litre of Ne has 10 times the density of 1 litre of H_2 |
|
Answer» II, IV, V II) `V prop n` at same P, T. III) at STP, both EXERT 1 atm. IV) `U prop 1/(sqrt(M))` V) `d prop M`. |
|
| 23. |
The raw material used in preparing permutit |
|
Answer» SODA ash `Al_(2)O_(3)` (Alumina) |
|
| 24. |
The nature of the aqueous solution after Kolbe's electrolysis is |
|
Answer» ACIDIC |
|
| 25. |
The nature of poisonous gas present in the exhausts of car is |
|
Answer» ACIDIC ,CO |
|
| 27. |
The natureof anoderaysdependsupon |
|
Answer» Natureof ELECTRONE |
|
| 29. |
The natural materials from which an element can be extracted economically are called |
|
Answer» ORES |
|
| 30. |
The names(s) of the following compound is : underset(CH(OH)COOH)underset(|)(CH(OH)COOH) |
|
Answer» tartaric ACID |
|
| 31. |
The names of compounds are given below. Which is/are the correct name(s) mentioned is correct against the structure given? |
|
Answer»
|
|
| 32. |
The name of CFCl_3 is ……………….. . |
| Answer» SOLUTION : Freon-11 | |
| 33. |
The name of CH_3CH(C_6H_5)CH_2undersetoverset(|)(OH)(C )H -CH_2CH_3 is |
|
Answer» 1-ethyl-3-phenyl-1-butenol |
|
| 34. |
The name of CH_(3)CH(C_(6)H_(5))CH_(2)underset("OH")underset("|")("C")-CH_(2)CH_(3) is |
|
Answer» 1-ethyl-3-phenyl-1-butenol |
|
| 35. |
The name of C_2H_4Cl_2 is …………. . |
| Answer» Solution :Freon -114 | |
| 36. |
The name of C_(2)F_(4)Cl_(2) is _____ |
|
Answer» Freon-112 |
|
| 37. |
The name of C_2F_4Cl_2 is …………………… . |
| Answer» Solution :Freon -114 | |
| 38. |
The name of C_(20)H_(42) and C_(30)H_(62) is…… and ……….respectively |
| Answer» SOLUTION :Icosen and Tricontene | |
| 39. |
The name glacial acetic acid is given to pure acetic acid : |
|
Answer» below 289-6 K it is white liquid |
|
| 40. |
The name 'Blue John' is given to which of the following compounds ? |
|
Answer» `CaH_(2)` |
|
| 41. |
The name chloromethyl acetylene implies |
|
Answer» `H_(3)C-C-=C-Cl` |
|
| 42. |
The name 'Blue John' is given to which of the following compounds? |
|
Answer» `CaH_2` |
|
| 43. |
The "N" which does not contribute to the basicity for the compound is: |
|
Answer» N 7 |
|
| 44. |
The N-O bond in NO_(2)^(-) is shorter than N-O bond in NO_(3)^(-). Account for this observation. |
Answer» Solution :`NO_(2)^(-)` is a resonance hybrid of two structures, I and II  while `NO_(3)^(-)` is a resonance hybrid of three structures, III, IV and V.  `:.` In `NO_(2)^(-)` BOND order of N-O bond is 3//2=1.5 since it has one duoble bond and one SINGLE bond, i.e., three bonds are shared between two O ATOMS. However, in `NO_(3)^(-)` bond order of N-O bond is 4//3=1.33 since it has one double bond and two single bonds, i.e., FOUR bonds are shared between three O atoms. Since higher the bond order shorter teh bond THEREFORE N-O bond is `NO_(2)^(-)` is shorter than N-O bond in `NO_(3)^(-)` |
|
| 45. |
The n-factor of FeS_2during its oxidation as Fes_(2) rarr Fe_(2)O_3+SO_2 |
|
Answer» 10 `S_2^(2-)rarr2SO_(2)+10C^(-)` TOTAL = 11 |
|
| 46. |
The (n+1) value for 4f sub shell is |
|
Answer» 4 |
|
| 47. |
The mutual sharing of one or more pair of electrons between the two combining atoms results in the formation of ……………………. |
| Answer» SOLUTION :COVALENT BOND | |
| 48. |
The mu of CH_(4) and "CCl"_(4) is zero. Than prediction of its shape and give reason of its right shape. |
| Answer» SOLUTION :Its SHAPE should be square planar or TETRAHEDRAL the bond angle is `109^(@)`28. so decide that the shape is tetrahedral not a square planar. | |
| 49. |
The movingbulletof gunhas 10gm massand10^(-5)muncertainty in position .Find uncertaintyin velocity |
| Answer» Solution :`5.27 XX 10^(28) MS^(-1)` | |