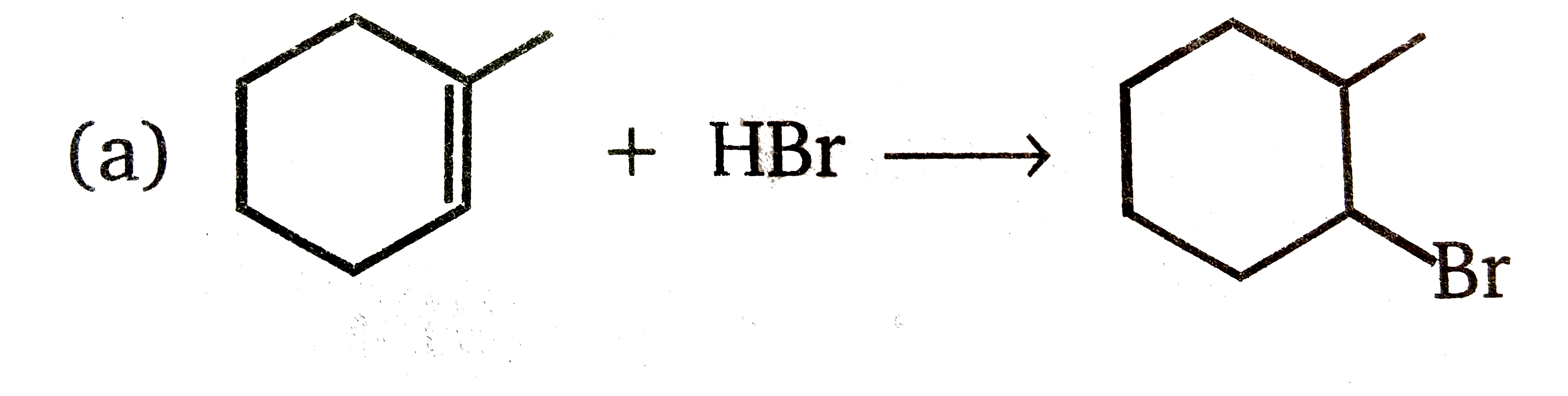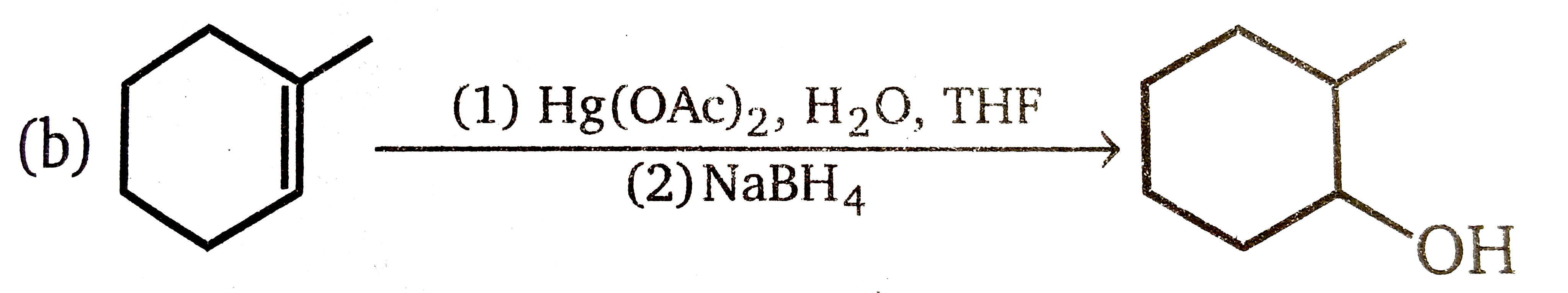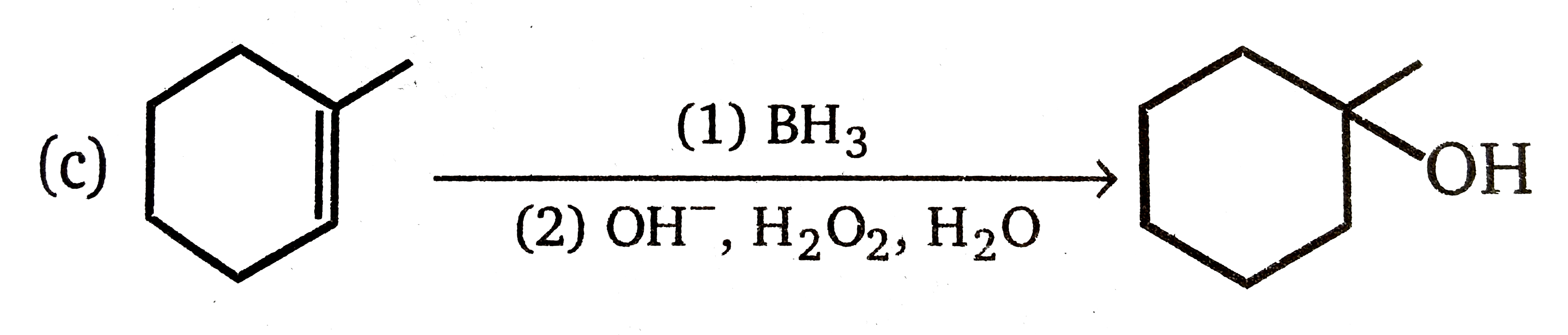Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What are biodegradable and non-biodegradable pollutants ? |
| Answer» Solution :Biodegradable pollutants are those which are decomposed by bacteria eg SEWAGE, cow-dung, etc. NON biodegradable pollutants are those which cannot be decomposed by bacteria, e g..MERCURY, ALUMINIUM, DDT, etc | |
| 2. |
Which of the following statements is not correct for a nucleophile? |
|
Answer» NUCLEOPHILES attack low electron DENSITY sites |
|
| 3. |
Which of the following 14 group elements have highest catenation property ? |
|
Answer» C |
|
| 5. |
What is theoretical plate? |
| Answer» Solution :Each SUCCESSIVE CONDENSATION and vaporization unit in the fractionating COLUMN is CALLED a theoretical plate. | |
| 6. |
When100mL of 1.0 M HCl was mixed with 100 mL of 1.0 M NaOH in an insulated beaker at constant pressure, a temerature increase of 0.7^(@)C was measured for the beaker and its contents (Expt. 1 ) . Because the enthalpy of neutralization of a strong acid with a strong base is a constant (-57.0 kJ "mol"^(-1)), the experiment could be used to measure the calorimeter constant. Ina second experiment (Expt. 2), 100 mL of 2.0 M acetic acid (K_(a)=2.0xx10^(-5)) was mixed with 100 mL of 1.0 M NaOH . (under identical conditions of Expt.1) where hte temperature rise of 5.6^(@)C was measured. (Consider heat capacity of all solutions as 4.2 Jg^(-1) K^(-1) and density of all solutions as 1.0 m mL^(-1)) The pH of the solution after Expt. 2 is |
|
Answer» 2.8 NAOH present in 100 mL of 1.0 M solution = 0.1 mole 0.1 mole of NaOH NEUTRALIZES 0.1 mole of NaOH to form 0.1 mole of `CH_(3)CO ON a` `:. ` In the final solution, `CH_(3)CO OH` = 0.1 mole, `CH_(3)CO ONa`= 0.1 mole, i.e., it is a buffer solution `pH = pK_(a) + log. (["Salt"])/(["Acid"])=-log(2XX10^(-5))+log.(0.1)/(0.1)` `=-log(2xx10^(-5))` `=5-0.301=4.699~= 4.7` |
|
| 7. |
By which process ,syngas is produced from coal ? |
| Answer» | |
| 8. |
Which molecule represents 4-ethyl-2-hexyne ? |
|
Answer» `(CH_3CH_2)_2 CHC -= "CCH" (CH_2CH_3)_2` |
|
| 9. |
When100mL of 1.0 M HCl was mixed with 100 mL of 1.0 M NaOH in an insulated beaker at constant pressure, a temerature increase of 0.7^(@)C was measured for the beaker and its contents (Expt. 1 ) . Because the enthalpy of neutralization of a strong acid with a strong base is a constant (-57.0 kJ "mol"^(-1)), the experiment could be used to measure the calorimeter constant. Ina second experiment (Expt. 2), 100 mL of 2.0 M acetic acid (K_(a)=2.0xx10^(-5)) was mixed with 100 mL of 1.0 M NaOH . (under identical conditions of Expt.1) where hte temperature rise of 5.6^(@)C was measured. (Consider heat capacity of all solutions as 4.2 Jg^(-1) K^(-1) and density of all solutions as 1.0 m mL^(-1)) Enthalpy of dissociation (in kJ "mol"^(-1)) of acetic acid obtained fromExpt. 2 is |
|
Answer» `1.0` `=100xx1` mmol = 0.1 mole Heat evolved = 0.1 mole `xx` 57.0 kJ `"mol"^(-1)` = 5.7 kJ = 5700 J Heat used to increase temperature of the solution (200 mL ) by `5.7^(@)` `=200 xx 4.2 xx 5.7` (SP. heat capacity of solution `= 4.2 JK^(-1)g^(-1)`) = 4788 J `:.` Heat used to increase the temperature of the calorimeter `=5700-4788 J = 912 J` ms `Delta t = 912` or ms `(5.7) = 912` or ms (calorimeter constant ) `= (912)/(5.7) = 160 J//^(@)C` In EXPT. 2, heat evolved by neutralization of `CH_(3)CO OH` with NaOH `{:(=,200xx4.2xx5.6,+,160xx5.6),(,("Absorbed by solution"),,("Absorbed by calorimeter")):}` `= 4704+896 J = 5600 J` `CH_(3)CO OH` present in 100 mL of 2.0 M solution `=100xx2 ` mmol = 0.2 mol NaOH present in 100 mL of 1.0 M NaOH `=100xx1` mmol = 0.1 mol `:. CH_(3)CO OH` neutralized by NaOH = 0.1 mol Thus, heat evolved by neutralisation of 0.1 mol of `CH_(3)CO OH = 5600 J` `:.` Heat used in the dissociation of 0.1 mol of `CH_(3)CO OH=5700-5600J = 100 J ` `:.` Enthalpy of dissociation of acetic acid per mole = 1000 J = 1 kJ |
|
| 10. |
Volume strength of 10%(w//v)H_(2)O_(2) is approximately |
|
Answer» 100 |
|
| 11. |
The total number of valency electrons in PO_(4)^(3-) ion is |
|
Answer» `32` |
|
| 12. |
Which of the following statements is not correct regarding enantiomers? |
|
Answer» They have identical melting and boiling points |
|
| 13. |
When MnO_4– reacts with H_2O_2 in basic medium then following species are involved, except |
|
Answer» `MnO_(2)` |
|
| 14. |
Water of crystallisation in compound P: |
|
Answer» 4 moles 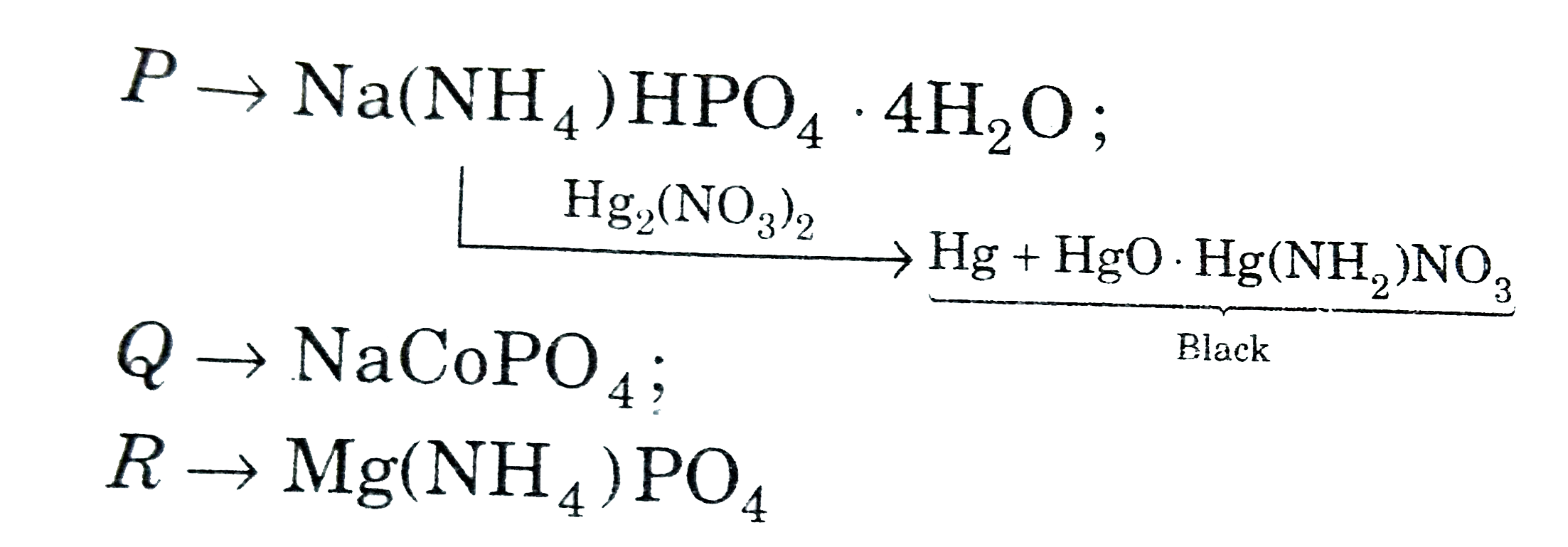 `Q RARR NaCoPO_(4)`, `R rarr Mg(NH_(4))PO_(4)` |
|
| 15. |
The value of universal gas constant depends upon ................ |
|
Answer» TEMPERATURE of the gas |
|
| 16. |
Which of the following is correct statement? |
|
Answer» <P>The members of secondperiod have lower ELECTRON affinity than the next member in their RESPECTIVE GROUPS of p-block |
|
| 17. |
Which of the following indicates the mathematical expression for Dalion's law of partial pressures a) Partical pressure = ("The component gas volume ")/("Total pressure") xx "Total volume " b) Partial pressure = mole fraction of component gas xx Total pressure c) Partial pressure = Partial volume xx Total pressure d) Partial pressure = No. of moles of component gas xx Total pressure |
|
Answer» a,d |
|
| 18. |
Which of the following is independent on slight variation of temperature? |
|
Answer» Molarity |
|
| 19. |
The volume of 0.1 M AgNO_(3) should be added to 10.0 ml of 0.09 M K_(2)CrO_(4) to precipitate all the chromate as Ag_(2)CrO_(4) is |
|
Answer» Solution :eq. `AgNO_(3)` = eq. `K_(2)CrO_(4)` `0.1xx1xxV_(mL)=10xx0.09xx2impliesV_(mL)=18mL` |
|
| 20. |
When three moles of ozone completely reacts with SO_(2), the number of moles of oxygen formed is |
|
Answer» 1 |
|
| 21. |
Which of the following is true |
|
Answer» It occurs during warm WEATHER |
|
| 22. |
Which of the following reactions generates the major product? Ignore stereoisomerism |
|
Answer»
|
|
| 23. |
What is permanent hardness of water? How it will be removed? |
|
Answer» Solution :Permanent hardness of water is due to the presence of soluble salts of magnesium and calcium in the form of chlorides and SULPHATES in water. It can be removed by adding washing soda which REACTS with these metal chlorides and sulphates in hard water to form insoluble carbonates. `MCI_(2(aq))+NaCO_(3(aq))toMCO_(3(s))+2NaCI_((aq))` `MSO_(4(aq))+ Na_(2)CO_(3(aq))toMCO_(3(s))+Na_(2)SO_(4(s)0` Where M = CA or Mg. |
|
| 24. |
What is the mass of glucose (C_(6) H_(12) O _(6)) in it one litre solution which is isotonic with 6 gL ^(-1) of urea (NH_(2) CONH_(2)) ? |
|
Answer» Solution :Osmotic PRESSURE of urea solution `(pi_(1)) = CRT` `= (W_(2))/(M _(2) V) RT = (6)/( 60 XX 1) xx RT` Osmotic pressure of glucose solution `(pi _(2)) = (W_(2))/(180 xx 1) xx RT` For isotonic solution, `pi _(1) = pi_(2)` `(6)/(60) RT= (W_(2))/(180) RT implies W_(2) = (6)/(60) xx 180` `W_(2) = 18g` |
|
| 25. |
What is the aim of the study of chemical thermodynamics? |
|
Answer» SOLUTION :The MAIN aim of the study of chemical thermodynamics is to learn . (i)Transformation of ENERGY from one FORM into another form. (ii) Utilization of various forms of energies. (iii) Change in the properties of system produced by chemical or PHYSICAL effects. |
|
| 26. |
What is allotropy ? Mention about the allotropic forms of Pb, Ge , Si. |
|
Answer» Solution :Allotropy : The existence of the same element in two or more FORMS in the same physical state but having different PROPERTIES is called allotropy. The different forms are called allotropes. PB: `to` No allotrope exists. Ge : `to` Two ( Both are crystalline forms ) Si : `to` Two ( Both are crystalline forms ) |
|
| 27. |
What is form when heavy water reacts with CaC_2 ? |
|
Answer» `CaD_2` |
|
| 28. |
Which of the following is an example of an organic reaction? |
|
Answer» RUSTING of iron |
|
| 29. |
Which is incorrectly matched? |
|
Answer» `{:("Disease","CAUSE"),((1)"Fluorosis ",Fluorides in WATER"):}` |
|
| 30. |
Which is useful is explaining certain properties such as acidity of phenol? |
|
Answer» Inductive |
|
| 31. |
When ortho dibromobenzene is subjected to mononitration X number of product are formed and when meta dibromobenzene is subjected to mononitration, Y number of products are formed. Report answer as XY. |
|
Answer» |
|
| 32. |
Which one is not tetrahedral ? |
|
Answer» `BF_(4)^(-)` `CO_(3)^(2-)` is `AB_(3)` While `BF_(4)^(-) , NH_(4)^(+) and SO_(4)^(2-)` all three are `AB_(4)` type TETRAHEDRAL and POSSESS `sp^(3)` hybridization .  
|
|
| 33. |
Which of the following is not possible ? |
|
Answer» `dsp^(3)` |
|
| 34. |
Which of the following is not a spontaneous process? |
|
Answer» All WATER fall runs downhill |
|
| 36. |
Write the IUPAC names of the products obtained by the ozonolysis of the following compouds : 3,4 Dimethyl hept-3-ene |
Answer» SOLUTION :
|
|
| 37. |
What is the main difference between wave emitted by a bulb or heater and that associated with a particle ? |
| Answer» Solution :The wave emitted by bulb/heater is an electromagnetic wave (ASSOCIATED with ELECTRIC and MAGNETIC FIELD) while that of a particle is only MATTER wave | |
| 38. |
Which out of sodium or potassium has higher melting point ? |
| Answer» Solution :On going from Na to K , the size of the atom INCREASES and hence the melting BONDING weakens . In other WORDS , SODIUM has higher melting POINT than potassium because of stronger metallic bonding . | |
| 39. |
Which salt is water soluble |
|
Answer» CALCIUM STEARATE |
|
| 40. |
Write IUPAC names of the products obtained by the ozonolysis of the following compounds : (i)Pent-2-ene , (ii)3,4-Dimethylhept-3-ene , (iii)2-Ethylbut-1-ene , (iv)1-Phenylbut-1-ene |
|
Answer» Solution :(i)`underset"Pent-2-ene"(overset5CH_3-overset4CH_2-overset3CH=overset2CH-overset1CH_3) underset((II)Zn//H_2O)overset((i)O_3//CH_2Cl_2, 196 K)to underset"PROPANAL"(CH_3-CH_2-CH)=O+O=underset"ETHANAL"(CH-CH_3)` (ii)`underset"3,4-Dimethylhept-3-ene"(overset7CH_3overset6CH_2overset5CH_2-undersetunderset(H_3C)(" "|)overset4C=undersetunderset(CH_3)|overset3C=overset2CH-overset1CH_3underset((ii)Zn//H_2O)overset((i)O_3//CH_2Cl_2, 195 K)to underset"Pentan-2-one"(CH_3CH_2CH_2-oversetoverset(CH_3)|C=O)+underset"Butan-2-one"(O=undersetunderset(CH_3)|C-CH_2H_3)` 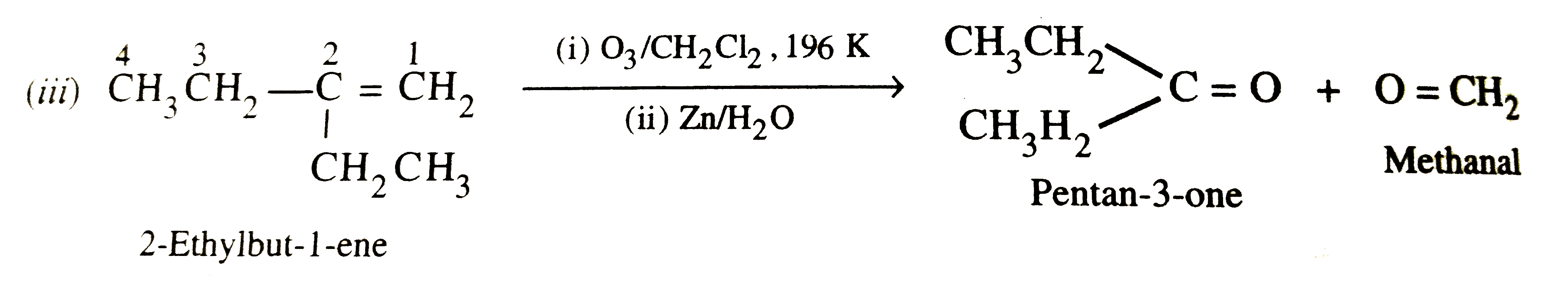 (IV)`underset"1-Phenylbut-1-ene"(overset4CH_3overset3CH_2-overset2CH=overset1CH-CH_6H_5) underset((ii)Zn//H_2O)overset((i)O_3, CH_2Cl_2 , 196 K) to underset"Propanal"(CH_3CH_2CH=O)+underset"Benzaldehyde"(O=CH-C_6H_5)` |
|
| 41. |
What is difference between real gas and ideal gas is graph of pV to p at constant temperature. |
|
Answer» SOLUTION :At high PRESSURE, volume of real GAS is higher than volume if IDEAL gas. When pressure decreases volume of real gas decreases then ideal gas. Real gas do not follow rules of ideal gas. |
|
| 42. |
Which element is most positive ? |
|
Answer» `[He]2s^(1)` |
|
| 43. |
What are ogranometallic compounds ? Give one example. Explain the nature of carbon-metal bond. |
|
Answer» Solution :(i) Organometallic compounds are organic compounds in which there is a direct CARBON-metal bond. (ii) EXAMPLE : `CH_3MgI`, METHYL magnesium iodide. (iii) The carbon - magnesium bond in Grignard reagent is covalent but highly polar. The carbon atom is more electronegative than magnesium. HENCE, the carbon atom has partial negative charge and magnesium atom has a partial positive charge. `overset(delta-)R..................overset(delta+)(MgX)` |
|
| 44. |
What is siltation ? |
| Answer» Solution :MIXING of soil or ROCK PARTICLES into WATER is called siltation | |
| 45. |
The vacant space in bcc lattice unit cell is |
|
Answer» 0.23 VACANT space in bcc lattice = 100 - 68 = 32% |
|
| 46. |
There are certain substances which behave as normal, strong electrolytes at low concentration but at higher concentration they behave as colloidal solutions due to the formation of aggregated particles. Such colloids are called associated colloids and the aggregated particles are called micelles. Soaps and detergents are the example of associated colloids. the formation of micelles takes place above certain concentration called critical micellization concentration (CMC) and a characteristic temperature. Above CMC, the surfactant molecules undergo : |
|
Answer» DISSOCIATION |
|
| 47. |
There are certain substances which behave as normal, strong electrolytes at low concentration but at higher concentration they behave as colloidal solutions due to the formation of aggregated particles. Such colloids are called associated colloids and the aggregated particles are called micelles. Soaps and detergents are the example of associated colloids. the formation of micelles takes place above certain concentration called critical micellization concentration (CMC) and a characteristic temperature. Micelles are formed only: |
|
Answer» below the CMC and the KRAFT TEMPERATURE |
|
| 48. |
What is the difference between terms: "hydrolysis" and "hydration"? |
|
Answer» Solution :Hydrolysis is a chemical reaction of a salt with WATER. `PCl_(3)+3H_(2)O to H_(3)PO_(3)+3HCL` Addition of `H_(2)O` through a physical process is HYDRATION. `CuSO_(4)+5H_(2)O to CuSO_(4).5H_(2)O` |
|
| 49. |
There are certain substances which behave as normal, strong electrolytes at low concentration but at higher concentration they behave as colloidal solutions due to the formation of aggregated particles. Such colloids are called associated colloids and the aggregated particles are called micelles. Soaps and detergents are the example of associated colloids. the formation of micelles takes place above certain concentration called critical micellization concentration (CMC) and a characteristic temperature. What type of molecules form micelles? |
|
Answer» Non-polar molecules |
|