Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following chemical reaction will not feasible? |
|
Answer» `PbO_(2)+dil.H_(2)SO_(4)rarrPbSO_(4)+H_(2)O+O_(2)` |
|
| 2. |
What are different isotopes of hydrogen? |
| Answer» | |
| 3. |
When 3-chlorocyclopropene (A) is treated with SbCl_5, it gives a stable salt (B), C_3H_3SbCl_6, which is highly polar. Further, when (A) is treated with AgBF_4, it gives a white precipitate of AgCl and a crystalline salt (C). Explain. |
Answer» SOLUTION :In both cases, salts of SYMMETRICAL cyclopropenyl CATION are formed, which are aromatic in character and hence STABLE. 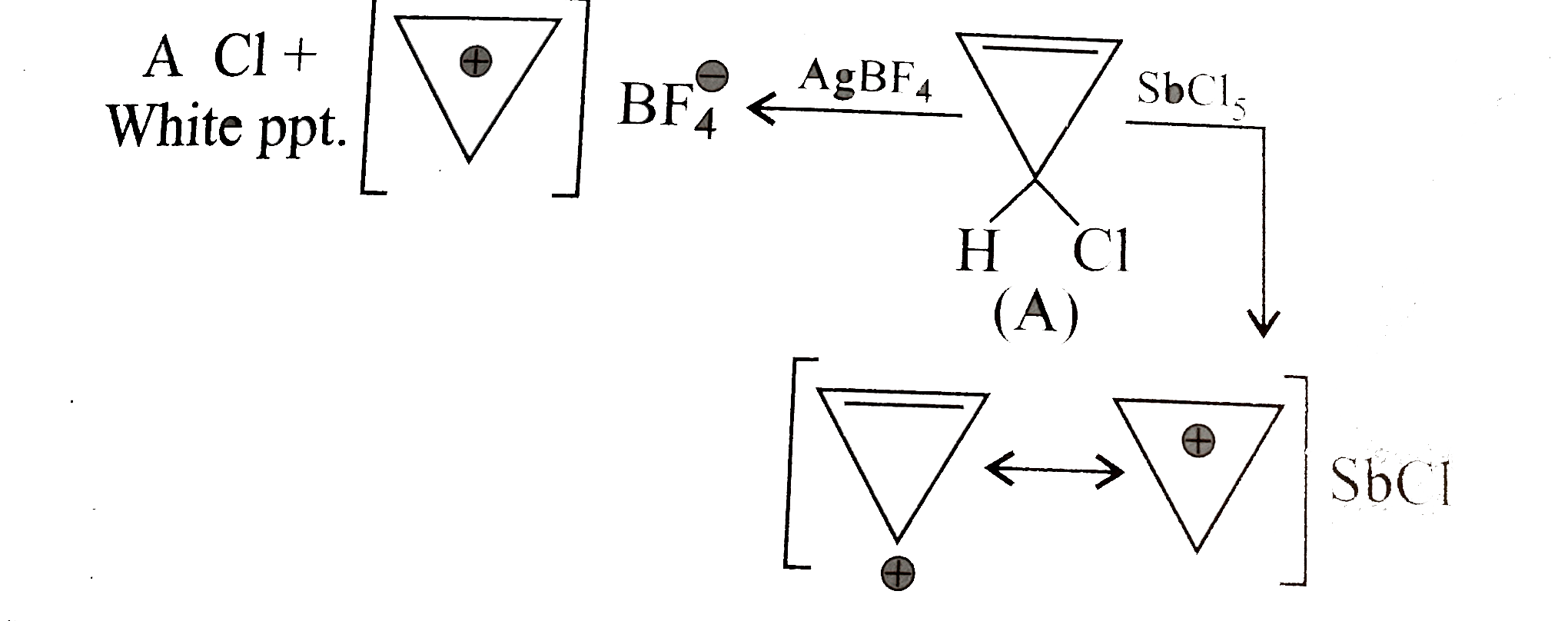 . .
|
|
| 4. |
Which of the following will react with 1-butyne ? 1) AgNO_3 + NH_4OH 2) Cu_2Cl_2 + NH_4OH 3) Na4) KMnO_4 // OH |
|
Answer» 1&4 |
|
| 5. |
Complete the chemical reaction :- CO + H_2 --> |
| Answer» | |
| 6. |
Which catalyst is used for preparation water-gas from methane ? |
|
Answer» Co |
|
| 7. |
When CO_(2) under high pressure is released from a fire extinguisher. Particles of soloid CO_(2) are formed, despite the low sublimation temperature (-77^(@)C) of CO_(2) at 1.0atm it is: |
|
Answer» the gas does WORK pushingback the atmosphere using KE of molecuels and THUS lowering the TEMPERATURE |
|
| 8. |
What is bond enthalpy? Explain bond enthalpy in polyatomic molecule like H_(2)O . |
|
Answer» Solution :It is defined as the amount of energy required to break one mole of bonds of a particular type between two atoms in gaseous state. The UNIT of bond enthalpy is kJ`mol^(-1)` . e.g.-1 : (i) The H - H bond enthalpy in hydrogen molecule is 435.8 kJ` mol^(-1)` `H_(2(g)) rarr H_((g)) + H_((g)) , Delta_(a)H^(O) = 435.8" kJ " mol^(-1)` e.g.-2 : (ii) The bond enthalpy for molecules containing multiple bonds, for example `O_(2) and N_(2)` will be as under : `O_(2) (O = O)_((g)) rarr O_((g)) + O_((g)) , Delta_(a) ` H = 408 kJ `mol^(-1)` `N_(2)(N equiv N)_((g))rarr N_((g)) , Delta_(a) H = ` 946.0 KK `mol^(-1)` Characteristic of bond enthalpy : Here bondenthalpy is energy of BROKEN the bond (dissociation) so, it endothermic, but making enthalpy is exothermic. It is important that larger the bond dissociation enthalpy, stronger will be the bond in the molecule. So enthalpy is increase in this order : single bond `rarr` double bond `rarr`Triple bond. HCl is heteronuclear diatomic molecules and its bond dissociation enthalpy is as under `HCl_((g)) rarr H_((g)) + Cl_((g)) : Delta_(a) H = 431.0 " kJmol"^(-1)` Bond dissociation enthalpy for polyatomicmolecule `H_(2)O` : In case of polyatomic molecule, the measurement ofbond strength is more complicated. Example: In case of `H_(2)`O molecule, the enthalpy needed to break the two O - H bond is not the same but different. `H_(2)O_((g)) rarr H_((g)) + OH_((g)) , Delta_(a) H_(1)^(Ө) = 502 " kJ mol"^(-1)` ` OH_((g)) rarr H_((g))+ O_((g)): Delta_(a)H_(2)^(Ө) = ` 427 kJ `mol^(-1)` The difference in the `Delta H^(Ө)` value shows that the SECOND O - H bond undergoes some change because of changed chemical environoment . This is the reason for some different in energy of the same O - H bond. In different molecules like `C_(2)H_(5)` OH (ethanol ) and water. Average Bond enthalpy: The mean or average bond enthalpy is used in more than one same bond containing polyatomic molecules. It is obtained by dividing total bond dissociation enthalpy by the NUMBER of bonds broken. Average bond enthalpy = `("Total enthalpy of broken bond")/("Number of all broken bond") ` `({:("Average enghalpy of"),("O - H bond in"H_(2)O):}) = (Delta_(a) H_(1)^(Ө) + Delta_(a) H_(2)^(Ө))/(2)` ` = (502 + 427)/(2)` = `464.5 kJ mol^(-1)` |
|
| 9. |
The set representing the correct order of first ionization potential is..... |
|
Answer» `K gt Na gt Li` |
|
| 10. |
Which real product is obtaine on hydrolysis of alkyne ? Why ? |
| Answer» Solution :HYDROLYSIS product of alkyne is ALKANOL and on isomerism it gives ALDEHYDE or ketone as FINAL product. (O the BASIS of nature of alkyne). | |
| 11. |
When oxalic acid (H_(2)C_(2)O_(4)) is used as reducing agent, the acid is oxidised to carbondioxide. Calculate the equivalent weight of the acid. |
|
Answer» |
|
| 12. |
The volume strength of 1.5 N H_2O_2 is |
|
Answer» 4.8V |
|
| 13. |
Which of the following is not chemically CaCO_3 ? |
|
Answer» Precipitated chalk |
|
| 14. |
Which of the following is a favourable factor for cation formation? |
|
Answer» HIGH electronegativity |
|
| 15. |
When copper pyrites is roasted in excess of air, amixture ofCuO + FeO is formed . FeO is present as impurities . This can be removed as slag during reduction of CuO . The flux added to form slag is |
|
Answer» `SiO_2` which is an acid FLUX |
|
| 16. |
Thesize ofisoelectronicspecies-F^(-) ,Neand Na^(+)isaffected by ( a)nuclear charge ( Z)( b)valenceprincipalquantumnumber (n) (c ) electron- electron interactionin theoutherorbital ( d) noneof thefactorsbacausetheir size is thesame . |
| Answer» Solution :The SIZEOF the isoelectronic ionsdependsupon thenuclear CHARGE (Z) . As thenuclearchargeincreases the sizedecreases . Forexample `F^(-)(+9) gt Ne + (+10) gt Na^(+) (+11)`. Thereforestatement (a) iscorrect while all otherstatementare wrong . | |
| 17. |
Which of the following pair of molecules has not same IUPAC name? |
|
Answer»
|
|
| 18. |
Which of the following compounds may give blood red colouration while performing Lassaigne.s lest for nitrogen |
|
Answer» <P>`(NH_2)_2C = S` |
|
| 19. |
Describe structure of BuckminsterFullerence ? |
| Answer» | |
| 20. |
ThethresholdfrequencyV_(0)for ametalis7.0 xx 10^(14) s^(-1)Calculationthe kineticenergyofandelectron emittedwhenradiationoffrequencyv=1.0 xx 10^(15) s^(-1)hitsthe metal . |
|
Answer» Solution :Accordingto Einsteinequation Kinetic energy`=(1)/(2) m_(e ) v^(2)` where`v_(0)= 7.0xx10^(14)s^(-1)` `=(6.626 xx 10^(34) j s ) ( 1.0 xx 10^(15) -7.0xx 10^(14)) s^(-1)` `=6.626 xx 10^(34)(10 xx 10^(14)-7.0xx 10^(14))` `19.878xx 10^(-20)` `=1.9879 xx 10^(19)J` |
|
| 21. |
Vinyl acetelyne overset(x)to chloroprene: x is |
|
Answer» `Cl_(2)//C Cl_(4)` |
|
| 22. |
The type of hybridisation of carbon atoms numbered 2 and 3 in CH_(2) = CH-CH= CH_(2) |
|
Answer» `SP^(2), sp^(3)` |
|
| 23. |
Zeolite used to soften hardness of water is, hydrated |
|
Answer» Sodium aluminium silicate `( NAISi_(2)O_(6).H_(2)O)` |
|
| 24. |
The uncertainity in the position of an electron (mass 9.1 xx 10^(28)g) moving with a velocity of 3.0 xx 10^(4) cm^(-1) accurate up to 0.011%, will be |
|
Answer» 1.92 cm |
|
| 25. |
What is the molecular formula of gypsum? |
|
Answer» `CaSO_(4)..H_(2)O` |
|
| 26. |
What is the difference between vapour and gas ? |
| Answer» Solution :A gas can be liquefied below the CRITICAL temperature by APPLYING PRESSURE. Hence, above the critical temperature, it is a gas but below the vritical temperature, it is VAPOUR. | |
| 27. |
What is the value of: (27RbT_( c))/a? |
|
Answer» |
|
| 28. |
Total number of fundamental particles present in N^(3-) ion (the nuclide of N is ""_(7)^(14)N |
|
Answer» |
|
| 29. |
The some amount of CH_3COONa_((s)) is added in CH_2COOH solution then.... |
| Answer» Answer :A | |
| 30. |
Which one of the following acids is not present in Acid rain? |
|
Answer» |
|
| 31. |
Whichelementsare determineby spectroscopy |
| Answer» SOLUTION :Rubidium (Rb)CAESIUM(cs) . Thallium (Ti)Indium (in)GALLIUM (Ga) and SCANDIUM (SC ) | |
| 32. |
Which of the following selected chain is correc to tname the given compound according to IUPAC system? |
Answer» Solution :The solected CARBON chain CONTAINING four carbon atom is correct since this is the longest posibile carbon chain containing both the functional group i.e. carboxyl `(-COOH)` and HYDROXYL `(-OH)`  The remaining three carbon chains containing seven, FIVE and six carbon atoms respectively are incorrect since NONE of them contains both the functional groups |
|
| 33. |
Which of the following is not a property of liquid state ? |
|
Answer» Intermolecular force of attraction in a liquid is quite large |
|
| 34. |
What is water-gas shift reaction? |
|
Answer» Solution :The CARBON monoxide of the water gas can be converted carbon dioxide be mixing the gas mixture with more steam at `400^(@)C` and passed over a shift converter containing iron/copper catalyst. This reaction is CALLED as water -gas shift reaction . `CO+H_(2)OrarrCO_(2)+H_(2)` The `CO_(2)` formed in the above process is absorbed in a solution of potassium CARBONATE. `CO_(2)+K_(2)CO_(3)+H_(2)Orarr2KHCO_(3)` |
|
| 35. |
When CI_(2) gas reacts wioth hot and concentrated solution of sodium hydroxide the oxidation number of chloirine changes from: |
|
Answer» `ZERO to +1 and zsero to -5` |
|
| 36. |
Which of the followings are Lewis acids ? H_2O, BF_3 , H^(+) , NH_4^(+) |
| Answer» SOLUTION :`BF_3, H^(+) , NH_4^+` | |
| 37. |
What do you understand by the terms : (i) Hydrogen economy (ii) hydrogenation(iii) syngas (iv) water-gas shift reaction and (v) fuel cell ? |
|
Answer» Solution :(i) Hydrogen economy . The proposal to use hydrogen as a fuel in industry , power plants and possibly also in homes and motor vehicles is calledhydrogen economy. The basic principle of hydrogen economy is production , storage asnd trasnportation of energy is the form of liquid or gaseous dihydrogen . (ii) HYDROGENATION means addition of hydrogen across double and triple bonds to form saturated compounds. The vegetable oils such soyabean oil, cotton SEED oil, groundnut oil, etc. are called POLYUNSATURATED oils since they contain many C=C bonds. When these oils are exposed to air for prolonged periods, the double bonds present in them undergo oxidation, i.e., develop unpleasant taste. To avoid this, double bonds are hydrogenated . For this purpose, dihydrogen is bubbled through edible oils in presence of finely divided nickel at 473 K when the oils are converted into solid fats. Vegatable oil `+ H_(2) overset(Ni, 473 K) to ` Vegetable Ghee THis process is called hdyrogenation or hardening of oils and is used in the manufacture of vegetable ghee like Dalda, Gagan, Rath etc. From vegetables oils. It MAY, however, be noted that hydrogenation reduces the number of double bonds but does not completely eliminate them. (iii) Syngas. The mixtures of CO and `H_(2)` are called synthesis gas or syngas. It can be produced by thereaction of steam on hydrocarbon or coke at high temperature in the presence of nickel as catalyst. `C_(n)H_(2n+2)+ nH_(2)O(g) underset(Ni) overset(1270 K)to nCO+(2n+1)H_(2)` e.g. `CH_(4)(g) + H_(2)O(g) underset(Ni)overset(1270K)to CO(g) + 3H_(2)(g)` These days, 'syngas' is produced from sewage, saw dust, scrap, wood, newspapers, etc. The process of producing syngas form COAL is called 'coal gasification' `C(s) + H_(2)O(g) underset(Ni)overset(1270K)to CO(g) + H_(2)(g)` (iv) Water -gas shift reaction. The amount of hydrogen in water gas can be further increased by oxidising CO to `CO_(2)` by mixing it with steam and passing the mixture over `FeCrO_(4)` as catalyst at 673 K. `underset("Syngas")ubrace(CO(g) + H_(2)O(g))+ underset("Steam")(H_(2)O(g)) underset(FeCrO_(4))overset(673K)to CO_(2)(g)+ 2H_(2)(g)` This chemical reaction is which CO of the which CO of the water gas reacts with steam to form `CO_(2)` and more `H_(2)` is called water-gas shift reaction. (v) Fuel Cell. Fuel cell is a device which converts the energy produced during the combustion of a fuel directly into electrical energy. Dihydrogen is used in hydrogen -oxygen fuel cells for generating electrical energy. It has many advantages over the conventional fossil fuels. It is eco-friendly, i.e., it does not cause and pollution and (ii) Its octane number is about three times than that of gasoline , i.e., it releases three times more energy per unit mass of fuel as compared to gasoline. |
|
| 38. |
Total number of stereoisomer in given compound are : CH_(3)-overset(OH)overset(|)CH-CH=CH-C=overset(Me)overset(|)C-CH_(3) |
Answer»  So STEREOISOMER `=2^(N)=2^(2)=4` |
|
| 39. |
Which one of the ions in the belowwould have thelargest value of enthalpy of hydration ? |
|
Answer» `{:("IONIC radius in nm " ,"Charge of ion"),(0.0065,+2):}` |
|
| 40. |
Among underset(* *)overset(* *)O=C=underset(* *)overset(* *)O and :O-=C-underset(* *)overset(* *)O:, which is a preferable structure for CO_(2) molecule. Why? |
|
Answer» `UNDERSET(* *)OVERSET(* *)O=C=underset(* *)overset(* *)O` |
|
| 42. |
Which of the following is a measure of bacteria present in water |
|
Answer» D.O |
|
| 43. |
The temperature at which Helium atoms have the same R.M.S. velocity as that of Methane at 927^@C is |
| Answer» ANSWER :C | |
| 44. |
trans-2-butene underset("addition")overset(Br_2)to Major product |
|
Answer» d-2,3-dibromobutane |
|
| 45. |
What is the oxidation state of Ni in Ni (CO)_(4) ? |
| Answer» Solution :ZERO OXIDATION STATE of METAL in metal carbonyls is zero | |
| 46. |
What is the mass of precipitate formed when 50 ml of 8.5% solution of AgNO_3 is mixed with 100 ml of 1.865% potassium chloride solution? |
|
Answer» 3.59 G 50 mL of 8.5% solution contains 4.25 g of `AgNO_3` Number of moles of `AgNO_3` PRESENT in 50mL of 8.5% of `AgNO_3` Number of moles of `AgNO_3` present in 50 mL of 8.5% `AgNO_3` solution. =Mass/Molar mass = 4.25/170 = 0.05 moles Similary, number of moles of KCl present in 100 mL of 1.865% KCl solution. `=1.865 //74.5 =0.025` moles So total amount of AgCl formed is 0.025 moles (based on the stoichiometry ) Amount of AgCl present in 0.025 moles of AgCl =No. of moles `xx` molar mass `=0.025 xx 143.5 = 3.59 g` |
|
| 47. |
Which of the following statement are true ? |
|
Answer» Bridgehead halide are inert for both `S_N1` and `S_N2` reaction. |
|
| 48. |
Which of the following compounds will be least susceptible to elimination of HBr? |
|
Answer»
|
|
| 49. |
Which one of the following is slowly degradable pollutants ? |
|
Answer» AGRICULTURE WASTE |
|






