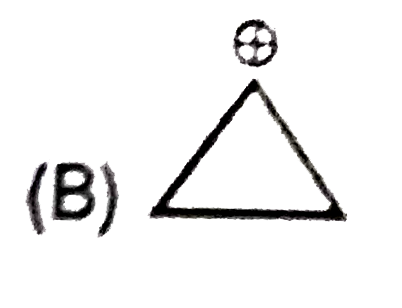Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Writes short notes on Hyperconjucation |
|
Answer» Solution :Hyper conjucation : This delocalisation of electrons of ` sigma ` bond is called as hyper conjugation . It is a SPECIAL stabilising effect that results due to the interaction of electrons of a ` sigma ` - bond (USUALLY -H or C-C) with the adjacent, empty non-bonding p-orbital or an anti-bonding ` sigma^(**) " or" pi^(**)` - orbitals resulting in an extended MOLECULAR orbital . Unlike electromeric effect, hyper conjugation is permanent effect . In PROPENE , the` sigma `-electrons of C-H bond of methly group can be delocalised into the `pi-` orbitals of doubly bonded carbon as represeted below, ` (##SUR_CHE_XI_V02_C12_E01_017_S01.png" width="80%"> |
|
| 2. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 3. |
Which of the following species are Isoelectronic with Ne? (Atomic no of Ne=10). N^(3-),Na^(+),Al^(3+),Ar,Rb^(+) & F^(-) |
|
Answer» |
|
| 4. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 5. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 6. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 7. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 8. |
Which of the following mainlycauses water pollution? |
|
Answer» FLY ash |
|
| 9. |
When compound A (orange red) is heated then green coloured oxide of (B) is formed and inert gas. (C) is formed then select the correct statements: |
|
Answer» Compound (A) is `(NH_(4))_(2)Cr_(2)O_(7)` |
|
| 10. |
Which of the following converts CH_(3)CONH_(2) to CH_(3)NH_(2) ? |
|
Answer» NaBr<BR>NaOBr |
|
| 11. |
What is the total number of orbitals associated with the principal quantum number n=3 ? |
|
Answer» <P> Solution :For N = 3, the possible values of l are 0,1 and 2. Thus, there is one 3s orbital (n = 3, l = 0 and `m_l` = 0), there are THREE p ORBITALS (n = 3, l = 1 and ml = -1, 0, 1) there are five 3d orbitals (n = 3, l = 2, ml = -2, -1, 0, 1, 2).THEREFORE, the total number of orbitals is 1+3+5 = 9. |
|
| 12. |
What is the product of the reaction C_(6)H_(5)COOCH_(3) overset(LiAlH_(4))rarr...... + ........ |
|
Answer» `C_(6)H_(5)COOH + CH_(3)OH` |
|
| 13. |
Under similar conditions of temperature and pressure gas having highest rate of diffusion is |
|
Answer» oxygen |
|
| 14. |
When 11.2 g of KOH is added to one litre of 'X' M H_(2) SO_(4) solution , the pOH of the solution becomes 13.301 . Then the value of 'X' is (assume no changein the volume of solution by the addition of KOH) |
|
Answer» `0.4` |
|
| 15. |
Write the electronic configuration of Lithium molecule. |
| Answer» SOLUTION :`SIGMA1S^(2),sigma1s^(2),SIGMA2S^(2)` | |
| 16. |
What is known as asymmetrical alkene ? Give examples. |
|
Answer» SOLUTION :When different alkyl groups attached to the two carbon having double BOND then such TYPE of ALKENE is known as asymmetrical alkene. e.g., `CH_(3)CH=CHC_(2)H_(5)` |
|
| 17. |
What is in reactivity in same period in left to right? |
| Answer» SOLUTION :Reactivity of ELEMENTS of the end of the PERIODIC table is more, but in the middle reactivity of elements DECREASES. | |
| 19. |
Which sulphides are soluble only in aqura regina ? |
|
Answer» NiS |
|
| 20. |
When a rod of copper is immersed in a solution of ZnSO_(4) zinc gets precipitated. |
|
Answer» |
|
| 21. |
What do you expect the nature of hydrides is, if formed by elements of atomic numbers 15,19,23,44 with dry dihydrogen ? Compare their behavoiur towards water. |
|
Answer» Solution :(i) ELEMENT with Z =15 is a non-metal (i.e. P)and hence forms covalent hydride (i.e. `PH_(3)`) (ii) Element with Z=19 is an alkali metal (i.e. K) and hence forms saline or ionic hydride (i.e. `K^(+)H^(-)`) (III) Element with atomic number Z=23 is a transition metal (i.e. V) of group 3 and hence forms a metallic or insterstitial hydride (i.e. `VH_(0.56)`) (iv) Element with Z=44 is a transition metal (i.e. Ru) of group 8 and hence does not FORM any hydride (hydride GAP). Behaviour towards water. Only ionic hydrides react with water EVOLVING `H_(2)` gas. Thus, `2KH(s) + 2H_(2)O(s) to 2KOH (aq) + 2H_(2)(q)` |
|
| 22. |
Which of the following compounds have same empirical formula ? a) formaldehyde "" b) Glucose c) Sucrose "" d) Acetic acid |
|
Answer» only a , B |
|
| 23. |
Which of the following statement/s is/are not correct |
|
Answer» Physical adsorption is directly related to the critical temperature of the gas (adsorbate) (c) is wrong because at about `0^(@)C`, adsorption remains constant after a particular pressure. (d) Is wrong because there can be negative adsorption also. |
|
| 24. |
What is ring - chain tautomerism ? |
Answer» Solution :Ring - Chain tautomerism occurs in sugars. (aldehyde vs the pyranose of FURANOSE STRUCTURE) - Oxocarboxylic ACIDS etc. 
|
|
| 25. |
Which of the following produces four monochloro derivatives on free radical chlorination ? |
|
Answer» 2,2-dimethylbutane |
|
| 26. |
Which order is true with reference to size of species? |
|
Answer» `M^(4) lt M^(2+) lt M ` |
|
| 28. |
What type of semiconductro is obtained when siliicon is doped with arsenic ? |
| Answer» SOLUTION :N -TYPE SEMICONDUCTOR. | |
| 29. |
Write the functional grop for (i) Aldehyde (ii) Ketone. |
|
Answer» |
|
| 30. |
The solubility of metal halides depends on their nature, lattice enthalpy and hydration enthalpy of the individual ions. Amongst fluorides of alkali metals, the lowest solubility of LiF in water is due to |
|
Answer» ionic nature of lithium fluoride |
|
| 31. |
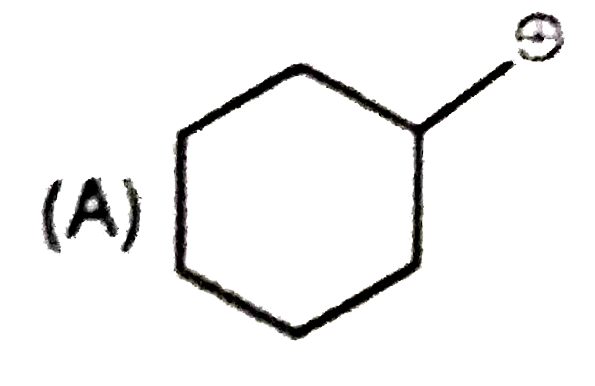
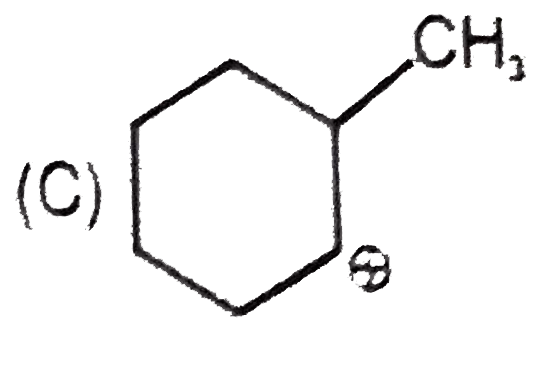
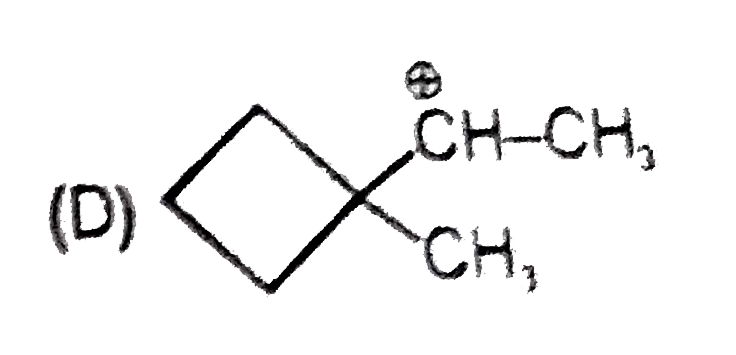
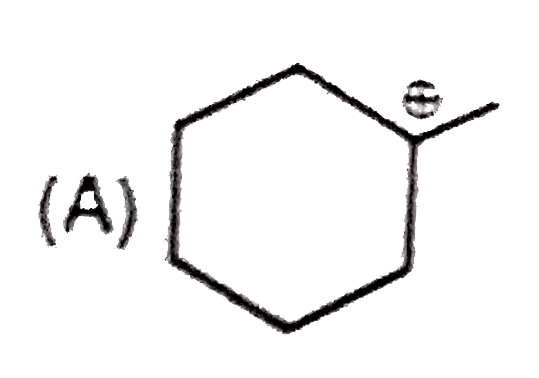
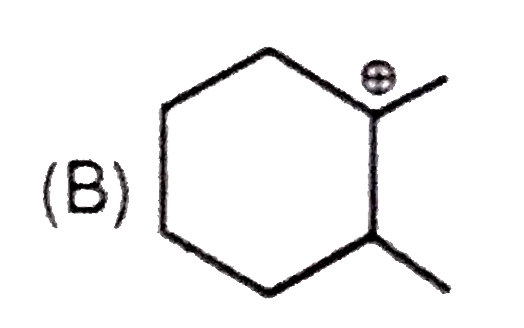
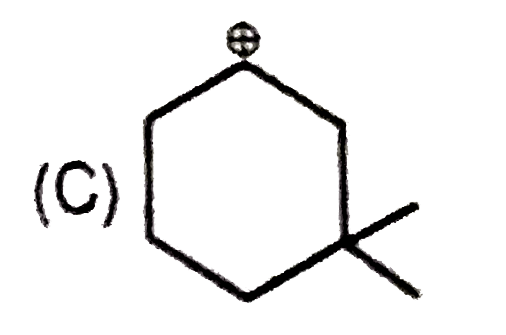
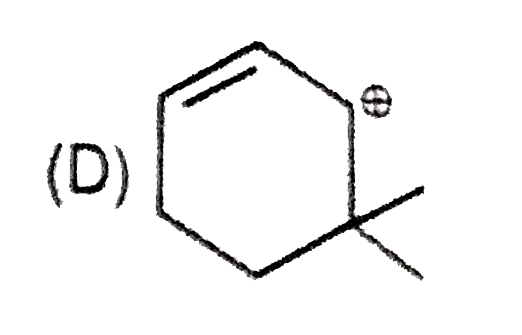
Whenever an intermediate carbocation is formed in reaction it may rearranges. Only those carbocation will rearrange which can produce more stable species . It can be done either by : (i) Shifting of H, alkyl , aryl, bond (1,2-shifting ) (ii) Ring expansion (iii) Ring contraction In which of the following carbocation rearrangement will not take place ? |
|
Answer»
|
|
| 32. |
Write formulas for the following compounds : (a) Mercury (II) chloride (b) Nickel (II) sulphate ( c) Tin (IV) oxide (d) Thallium (I) sulphate (e) Iron (III) sulphate (f) Chromium (III) oxide |
|
Answer» Solution :(a) Mercury (II) chloride - `Hg(II)Cl_(2)` (B) NICKEL (II) sulphate - `NI(II)SO_(4)` ( c) TIN (IV) oxide - `Sn(IV)O_(2)` (d) Thallium (I) sulphate - `Tl_(2)(I)SO_(4)` (e) Iron (III) sulphate - `Fe_(2)(III)(SO_(4))_(3)` (f) Chromium (III) oxide - `Cr_(2)(III)O_(3)` |
|
| 34. |
Whenever an intermediate carbocation is formed in reaction it may rearranges. Only those carbocation will rearrange which can produce more stable species . It can be done either by : (i) Shifting of H, alkyl , aryl, bond (1,2-shifting ) (ii) Ring expansion (iii) Ring contraction Most stable rearranged carbocation is is |
|
Answer»

|
|
| 35. |
Which of the following compound is given red colour in Lassaigne's test? |
|
Answer» NACNS |
|
| 36. |
Which of the following electronic configuration is n o t possible ? |
|
Answer» `3d^(2)` |
|
| 37. |
Which metal ion is present in chlorophyl the green pigment of plants |
|
Answer» `Ca^(2+)` |
|
| 38. |
When the pressure on a gas is decreased to 1/4 and the absolute temperature is increased four-fold the volume of the gas |
|
Answer» INCREASES by 16 TIMES |
|
| 39. |
Which one of the following reacts with CH_3Mglfollowed by acid hydrolysis to yield acetic acid? |
|
Answer» `CNCL` |
|
| 40. |
Which of the following compounds do not contain pi-bond? |
|
Answer» `CH_(3)CHO` 
|
|
| 41. |
Which of the followingg compounds is not correctly matched wiith its IUPAC name? |
|
Answer» `CH_(3)CH_(2)CH_(2)COOCH_(2)CH_(3)`-Ethyl butanoate The correct name is 3-methylbutan-2-ol since functional group should GET lowest NUMBER. |
|
| 42. |
Which of the following is metal displacement reaction |
|
Answer» `Zn + CuSO_(4)rarrZnSO_(4) + Cu` |
|
| 43. |
Which is formed when SiCl_(4) vapours are passed over hot Mg |
|
Answer» `SiCl_(2) + MgCl_(2)` |
|
| 44. |
Which of the following sets will have highest hydration enthalpy and highest ionic radii ? |
|
Answer» Na and Li |
|
| 45. |
The solubility of alkali metal hydroxides follows the order |
|
Answer» `LiOH LT NaOH lt KOH lt RbOH lt CSOH` |
|
| 46. |
Threshold frequency, v_(0) is the minimum frequency which a photon must posses to eject an electron from a metal. It is different for different metals. When a photon of frequency 1.0 xx 10^(15) s^(-1) was allowed to hit a metal surface, an electron having 1.988 xx 10^(-19) J of kinetic energy was emitted. Calculate the threshold frequency of this metal. Show that an electron will not be emitted if a photon with a wavelength equal to 600nm hits the metal surface. |
|
Answer» Solution :`hv = hv_(0) + K.E. or hv_(0) = hv - K.E.` or `v_(0) = v - (K.E.)/(h) = 1.0 xx 10^(15) s^(-1) - (1.988 xx 10^(-19) J)/(6.626 xx 10^(-34) Js)` `= (1.0 xx 10^(15) - 0.30 xx 10^(15)) s^(-1) = 0.97 xx 10^(15) s^(-1)` `9.7 xx 10^(14) s^(-1)` When `LAMDA = 600nm, " i.e., " 600 xx 10^(-9) m or 6.0 xx 10^(-7) m` `v = (c)/(lamda) = (3.0 xx 10^(8) ms^(-1))/(6.0 xx 10^(-7) m) = 0.5 xx 10^(15) s^(-1) = 5 xx 10^(14) s^(-1)` THUS, `v lt v_(0)` Hence, no electron will be emitted |
|
| 47. |
Which of the folowings statements are not true ? |
|
Answer» Vacancy defect results in a decrease in the DENSITY of the substance. (d) is not true because Frenkel defect neither changes mass nor volume. |
|
| 48. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 49. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|
| 50. |
Write the possible structures for the following molecules .Show the direction of the movement of electrons with the help of arrows. |
Answer» SOLUTION :
|
|