Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following behaves as Lewis base as well as Bronsted-Lowry base ? |
|
Answer» Carbonium ION |
|
| 2. |
What is meant by localized orbitals ? |
| Answer» SOLUTION :LOCALIZED molecular orbitals are molecular orbitals which are concentrated in a LIMITED spatial region of a MOLECULE | |
| 3. |
What is the difference in the angular momentum of an electron present in 3p and that present in 4p orbital ? |
| Answer» Solution :No difference because ANGULAR momentum `= (h)/(2pi) SQRT(l (l +1))` and for 3P or 4p orbital, `l = 1` | |
| 4. |
Write the anomalous behaviours of boron. |
|
Answer» SOLUTION :The ANOMALOUS behaviours of boron are as follows : (a) Boron is a harder element than the other elements of its GROUP . (b) It has high MELTING and boiling points compared to other members of the group . (c ) It forms only covalent and ionic compounds . (d ) Boron oxide is a weak acid while ALUMINIUM and gallium oxides are amphoteric. Indium and thalium oxides are acidic in nature. |
|
| 5. |
Two elements A and B have the following electronic configurations. The formula of the compound formed between them can be A=1s^2 2s^2 2p^6 3s^2 3p^1 , B=1s^2 2s^2 2p^4 |
|
Answer» AB |
|
| 6. |
Which of the following relate to wave nature of light or particle nature or both ? (a) Interference (b) Diffraction (c) Photoelectric effect (d) Black body radiation (e) Planck's equation, E = hv (f) Einstein equation, E = mc^(2) |
| Answer» SOLUTION :(a) WAVE nature (b) Wave nature (C) Particle nature (d) Particle nature (e) Both particle and wave nature (F) Particle nature. | |
| 8. |
Which of the anions is not hydrolyzed in aqueous solution ? CI^(-),(ii) NO_(3)^(-) (iii) Br^(-),(iv) CIO_(4)^(-) |
|
Answer» (i), (ii), (iii), (iv) |
|
| 9. |
"The total mass of reactants is always equal to the total mass of products in a chemical reaction." This statement is known as |
|
Answer» LAW of CONSERVATION of mass |
|
| 10. |
Which of the following are less acidic than phenol ? |
|
Answer» `H_(2)O` |
|
| 11. |
What is the state of hybridisation of carbon atoms in benzene? |
| Answer» SOLUTION :`sp^2` | |
| 12. |
The standard electrode potential corresponding to the reaction : Au^(3+)(aq) +3e^(-) rarrAu(s) is 1.42 V . Predict (i) if gold can be can be dissolved in 1 M HCl solution and (ii) on passing hydrogen gas through gold solt solution, metallic gold will be precipitated or not . |
| Answer» Solution :The standard reduction potential is greater than zero , therefore , reduced form of gold is more STABLE than hydrogen . In other words , gold does not dissolve in 1 M HCl solution . On the other HAND , if hydrogen gas is passes through the saltsolution of the gold , it will reduce `Au^(3+)` ion and METALLIC gold will be PRECIPITATED . | |
| 13. |
What is relation between compressibility and temperature an liquifaction of gas ? |
| Answer» SOLUTION :GASES do not liquify on COMPRESSION only, although molecules COME very close to each other and intermolecular forces operate to the MAXIMUM. | |
| 14. |
underset(("Orange solid"))((A))overset(Delta)rarrunderset(("Yellow"))((B))+underset(("Green"))((C)) +underset(("Paramagnetic"))((D)) Compound (A) is: |
|
Answer» `K_(2)Cr_(2)O_(7)` |
|
| 15. |
Which one of the following represents the composition of carnallite mineral ? |
|
Answer» `K_(2)O.Al_(2)O_(3). 6 SiO_(2)` |
|
| 16. |
What is the nuclear reaction that take place in the sun and other stars? |
| Answer» Solution :The sun and other stars are composed MAINLY of 85-95% hydrogen which GENERATES their ENERGY by nuclear FUSION of hydrogen nuclei into helium. | |
| 17. |
What is the type of hybridisation of each carbon in the following compounds? (CH_(3))_(2)CO |
| Answer» Solution :`CH_(3)-OVERSET(O)overset(||)(C)-CH_(3)-SP^(3)-sp^(2)-sp^(3)` | |
| 18. |
When the oxidation number of an element is maximum , it can act only as ........ |
| Answer» SOLUTION :OXIDISING AGENT | |
| 19. |
What statement is correct for Keto-enol tautomerism ? |
|
Answer» Tautomersim is catalysed by acid and base. |
|
| 20. |
The value of n_(1) in Balmer series is |
|
Answer» 2 |
|
| 21. |
Which of the following biological activity is not related with calcium ? |
|
Answer» Interneuronal transmission |
|
| 22. |
Thespecificheatof wateris |
|
Answer» Solution :(i) Even a small rise in temperature in the surroundings will raise the temperature of the body because the SPECIFIC heat of ALCOHOL is much less than the specific heat of water. So, in order to cool the body, more sweating will take place. (ii) As there is less H bonding in alcohol, it will get evaporated faster. The alcohol will be evaporated at such a faster rate that the liquid has to be ingeasted all the time. (iii) Ice which floats on water helps aquatic life to EXIST even in winter as water insulates the heat from liquid below it to go back to the surroundings. Solid alcohol does not have such special properties. (b) Praise is to the ALMIGHTY that has so thoughtfully given such special properties to water and made it a liquid that could sustain life on earth. |
|
| 23. |
Write the correct resonance structure of the given compound. . |
|
Answer» Solution :There are four POSSIBILITIES, either `H^(Ө)` shifts from `C-2` or `C-4` or `Me` shifts from `C-2` or `C-4`. But `H^(Ө)` shift produces more stable resonance structure. MOREOVER, `H^(Ө)` shift from `C-2` would give more stable structure DUE ti conjugation of positive charge with the `L P overline E^, s` on `O` atom of `OH` group.  , ,  . . 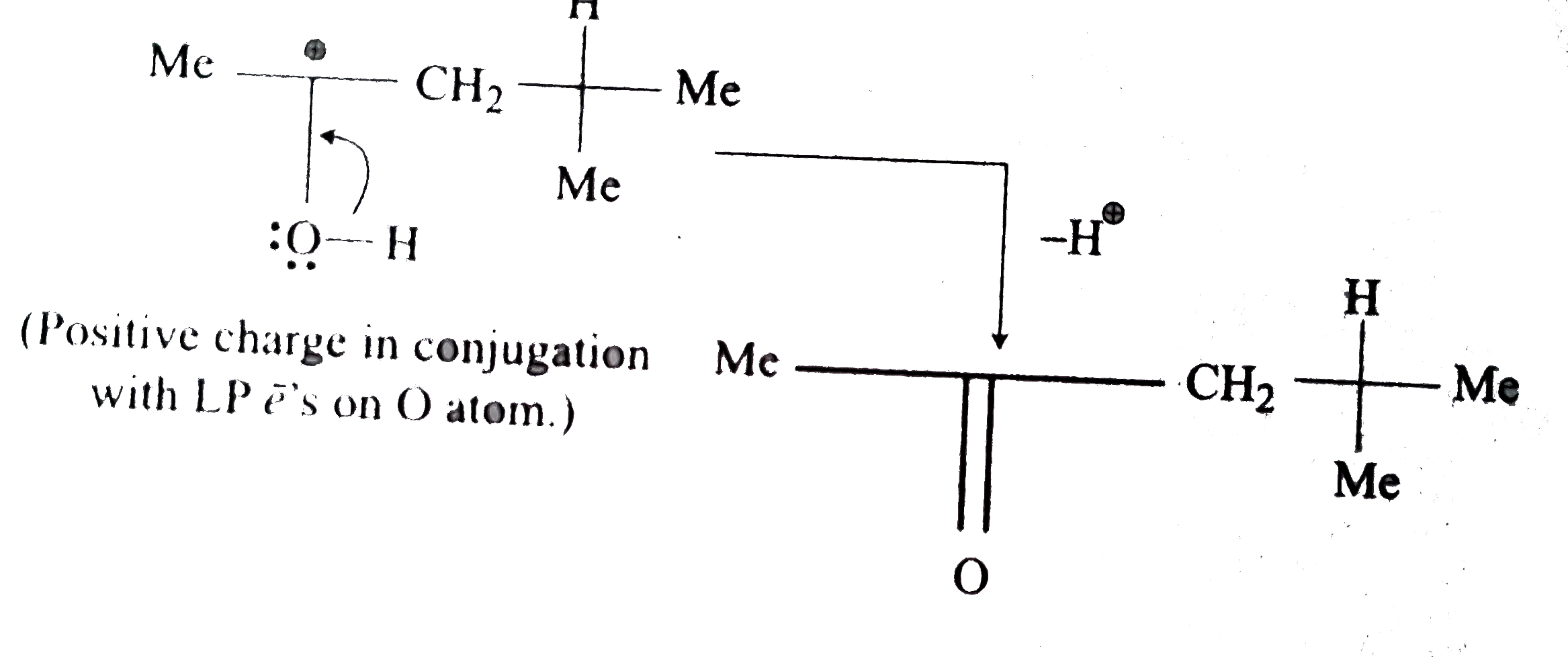
|
|
| 24. |
Which of the following compound is having the highest P^(ka) value |
|
Answer» PHENOL |
|
| 25. |
Which of the following statements is correct with respect to the property of elements wiith increase in atomic number of ini the carbon family (group 14)? |
|
Answer» Their METALLIC character decreases. |
|
| 26. |
Two buffers (X) and (Y) of pH 4.0 and 6.0 respectively are prepared from acid HA and the salt NaA. Both the buffers are 0.50 M in HA. What would be the pH of the solution obtained by mixing equal volumes of the two buffers ? (K_(HA)=1.0xx10^(-5)) |
|
Answer» Solution :For BUFFER X, `PH=pK_(a) + log. (["Salt"])/(["Acid"]), i.e., 4 = 5 + log. (["Salt"])/(0.5) or log .(["Salt"])/(0.5) = - 1 or (["Salt"])/(0.5) = 10^(-1) or ["salt"]=0.05 M` For buffer Y, `pH = pK_(a) + log. (["Salt"])/(["Acid"]), i.e., 6 = 5 + log .(["Salt"])/(0.5) orlog. (["Salt"])/(0.5) or (["Salt"])/(0.5) = 10 or ["Salt"]=5 M` When EQUAL VOLUMES of both the buffers are mixed, [Acid] = 0.5, [Salt] `=(5+0.05)/(2) = 2.5025` `pH = pK_(a) + log. (["Salt"])/(["Acid"])=5 + log. (2.5025)/(0.5) = 5 + log 5.05 = 5.7 ` |
|
| 27. |
Thermite is a mixture of iron oxide and |
|
Answer» ZINC powder |
|
| 28. |
When a small dry piece of sodium metal is throw in water, it immediately fire. What actually happens |
|
Answer» SOLUTION :The hydrogen envolved in the reaction catches fire and HOT the sodium metal because it is a highly combustilble GAS. `2Na(s)+H_(2)O(l)to 2NaOH(aq)+H_(2)(g)` |
|
| 29. |
Value of gas constant (R) is |
|
Answer» 0.082 lit ATM |
|
| 30. |
Which of the following groups of elements have properties that are most similar ? |
|
Answer» Na, K , Cs |
|
| 31. |
What is the shape of the curve obtained on plotting P against PV at constant temperature for an ideal gas ? |
| Answer» Solution :Straight LINE PARALLEL to PRESSURE AXIS | |
| 32. |
Three moles of an ideal gas at 27^(@)C are compressed reversibly and isothermally from a volume of 10 dm^(3) to 5 dm^(3). Calculate the work done on the gas. |
|
Answer» Solution :`T = 27^(@)C = 27 + 273 = 300 K, V_(1) = 10 dm^(3), V_(2) = 5 dm^(3)` `R = 8.314 J//K//Mol, n = 3 "MOLE"` `R = 8.314 w = -2.303 nRT log ((V_(2))/(V_(1))) = -2.303 xx 3 xx 8.314 xx 300 xx log[(5)/(1)]` `= -2.303 xx 3 xx 8.314 xx 300 xx bar(1).6990` `= -2.303 xx 3 xx 8.314 xx 300 xx (-0.3010) "" because -1 + 0.6990 = -3010`. `= +5,186.96 "joule" = 5.187 kJ` |
|
| 33. |
Uncertainty principle gave the concept of |
|
Answer» probability |
|
| 34. |
Water cannot be used to extinguish petrol fires. Why ? |
| Answer» Solution :WATER is used to extinguish most fires because it LOWERS the temperature of BURINING material. However, in case of petrol fires, petrol being LIGHTER than water, floats over water and hence fire spreads INSTEAD of being extinguished. | |
| 35. |
Write the oxidation state of the constituents in chromium hydride. Mention reasons. |
| Answer» Solution :Oxidation state of CHROMIUM = 0. Oxidation state of HYDROGEN = 0 Chromium hydride is a metallic hydride. It is a non - stoichimetric compound and involves no CHEMICAL change. The oxidation state of CONSTITUENTS is zero. | |
| 36. |
Write the valence shell electronic configuration of P-block elements. |
|
Answer» |
|
| 37. |
Under what condition, the heat evolved or absorbed in a reaction is equal to its free change ? |
| Answer» Solution :As `DeltaG = Delta H-T DELTAS`. Thus, `DeltaG = DeltaH ` only when EITHER the REACTIONIS carried out at `0^(@)K` or the reaction is not ACCOMPANIED by any entropy change ,i.e., `Delta S = 0`. | |
| 38. |
Which of the following compounds with molecular formula C_(5)H_(10) yields acetone on ozonolysis? |
|
Answer» `2`-Methylbut-`1`-ene  `CH_(3)-underset(3"-Methylbut-1-ene")(underset(CH_(3))underset(|)(CH_(2)))-CH=CH_(2)underset((2)Zn//H_(2)O)OVERSET((1)O_(3)//CHCl_(3))rarrCH_(3)underset(CH_(3))underset(|)(CH)CH=O+O=CH_(2)` Cyclopentane does not UNDERGO ozonolysis. |
|
| 39. |
Which of the following element is extracted commercially by the electrolysis of an aqueous solution of its compound ? |
|
Answer» CHLORINE |
|
| 41. |
Which statement (s) is/are correct ? |
|
Answer» Ratio of De-broglie wave lentghs of ELECTRONS and proton moving with same speed is 1:1836 |
|
| 43. |
Which one having less radius between Al and Al^(3+)? Why? |
|
Answer» Solution :`Al^(3+)`having LESS radius because Al is parent action when 3 ELECTRONS REMOVED than `Al^(3+)`is obtain. So ATTRACTION becomes more in `Al^(3+)` than Al. |
|
| 44. |
Which type of hybridisation is found in atoms of acetylene (ethyne)? |
| Answer» ANSWER :A | |
| 45. |
Under what conditions can the process of team distillation is used? |
| Answer» Solution :Steam DISTILLATION is used to PURIFY the liquids which are steam VOLATILE and not MISCIBLE with water. | |
| 46. |
When ammonical solution of common salt is saturated with carbon dioxide, we obtain |
|
Answer» `(NH_(4))_(2)CO_(3)` |
|
| 47. |
Which of the following molecules is expected to rotate the plane of polarized light? |
|
Answer»
 Due to the PRESENCE of CHIRAL carbon atom it is OPTICALLY ACTIVE , hence it is expected to rotate plane of polarized LIGHT. |
|
| 48. |
Write the structural and IUPAC names of different structural isomers of alkenes corresponding to C_5H_10. |
|
Answer» Solution :The VARIOUS structural isomers and their IUPAC names are GIVEN below : (i)`underset"Pent-1-ene"(overset1CH_2=overset2CH-overset3CH_2-overset4CH_2-overset5CH_3)` (ii)`underset"Pent-2-ene"(overset1CH_3-overset2CH=overset3CH-overset4CH_2-overset5CH_3)` (iii)`underset"2-Methylbut-1-ene"(overset1CH_2=.^2oversetoverset(CH_3)|C-overset3CH_2-overset4CH_3)` (IV)`underset"3-Methylbut-1-ene"(overset1CH_2=overset2CH-.^3oversetoverset(CH_3)|CH-overset4(CH_3)` (v)`underset"2-Methylbut-2-ene"(overset1CH_3-.^2oversetoverset(CH_3)|C=overset3CH-overset4CH_3)` |
|
| 49. |
What will be the correct order of vapour pressure of water, acetone and ether at 30^@C. Given that among these compounds, water has maximum boiling point and ether has minimum boiling point ? |
|
Answer» WATER `lt` Ether `lt` Acetone `{:("Water,","Acetone,","Ether"),("(Maximum b.p.)",,"(Minimum b.p.)"):}` More the boiling point, LOWER is the vapour PRESSURE of the solvent. So, the correct order of vapour pressure will be Water `lt` Acetone `lt` Ether |
|