Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which elements have the electronic configuration ns^(2)np^(4)in their outer most orbit? |
|
Answer» Inert gases |
|
| 2. |
Which of the following hydrides has the largest bond angle ? |
| Answer» Solution :`H_(2) O` | |
| 3. |
Which free radical is the most stable ? |
|
Answer» `OVERSET(*)(C)H_(3)` |
|
| 4. |
Which of the following represents the best resonance form for H_2C = CHF ? |
|
Answer»
|
|
| 5. |
Whichof the following is truefor number ofelectronsorbitalsand type , orbitalspresentin N-orbitrespectively? |
|
Answer» `4,4 ` and 8 |
|
| 6. |
What is Birch reduction ? What is it used for ? |
Answer» SOLUTION :Reduction of alkynes with sodium in liquid ammonia is CALLED BIRCH reduction. It is used for PARTIAL reduction of alkynes to trans-alkenes. For example, 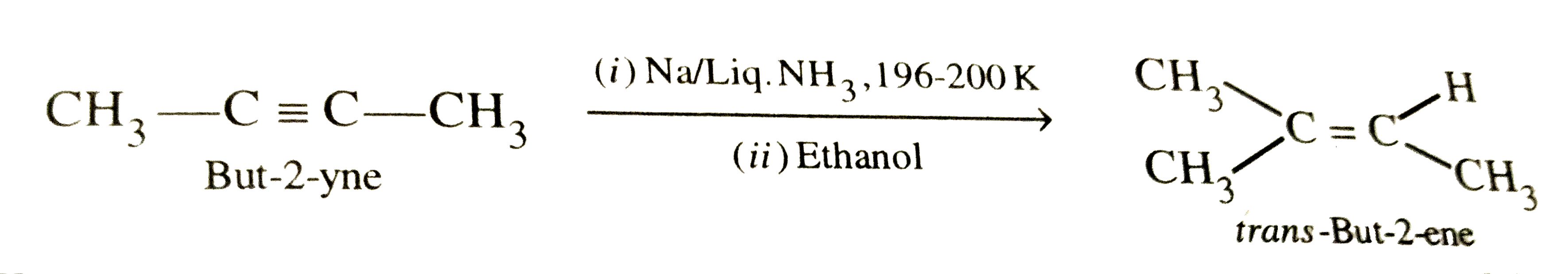
|
|
| 7. |
Which of the following gives red colour in Victor Meyer's test? |
|
Answer» n-Propyl alcohol `underset((3^(@)-"alcohol"))underset("tert-Butyl alcohol")(CH_(3)-underset(OH)underset(|)overset(OH)overset(|)(C)-CH_(3)) "" underset((2^(@)-"alcohol"))underset("sec-Butyl alcohol")(CH_(3)-overset(OH)overset(|)(CH)-CH_(2)-CH_(3))` A `1^(@)` alcohol GIVES RED colour in Victor Meyer's TEST. Therefore, n-propyl alcohol (i.e., only `1^(@)` alcohol) will give a red colour in the test. |
|
| 8. |
Which of the followingsetscontainonly isoelectronicions ? |
|
Answer» `Zn^(2+) , CA^(2+) , Ga^(3+) ,A1^(3+)` havedifferentno of electron and henceare notisoelectronicions. Species ofset ( b) i.e.,`K^(+)(19-1=18) ,Ca^(2+)(20-2 =18) , Sc^(3+)(21-3=18) ,C1^(-)(17+1=18) `are allisoelectron ionssince each one of themhas 18electrons. Speciesof set (c )i.e., `P^(3-)(15+3 =18),S^(2-)(16+2 =18),C1^(-)(17+ 1=18) , K^(+)(19-1=18) ` are allisoelectron ionssince EACHONE of themhas 18 electrons . Species ofset (d) i.e., `Ti^(4+) (22 -4 =18),Ar (18), Cr^(3+)(24-3=21) ,V^(5+)(23 -5= 18)` have DIFFERENT numberof electron and henceare notisoelectronicions. |
|
| 10. |
Two compounds with molecular formula C_(4)H_(6) is available , if one of the compound can react with soda amide while second can't react with ti, then find out that organic compound ? |
|
Answer» Solution :These compounds MAY be alkyne in nature. `underset("But-2-yne")(CH_(3)C-=C CH_(3)) overset(NaNH_(2))rarr "Not a reaction"`. `underset("But-1-yne")(CH_(3)CH_(2)C-=CH) overset(NaNH_(2))rarr underset("Sodium butinide")(CH_(3)CH_(2)C-=CNa)` So, but-2-yne and But-1-yne are the compounds with MOLECULAR formula `C_(4)H_(6)` |
|
| 11. |
Which is not correctly matched? I) Basic strength of oxides Cs_(2)OltRb_(2)OltK_(2)OltNa_(2)OltLi_(2)O II) Stability of peroxides Na_(2)O_(2)ltK_(2)O_(2)ltRb_(2)O_(2)ltCs_(2)O_(2) III) Stability of biocarbonate LiHCO_(3)ltNaHCO_(3)ltKHCO_(3)ltRbHCO_(3) IV) Melting point NaFltNaClltNaBrltNaI |
|
Answer» 1 and 4 |
|
| 12. |
The total number of lattice arrangements in different crystal systems is |
|
Answer» `7.0` |
|
| 13. |
Which of the following is Bragg's equation ? |
|
Answer» `nlambda=2thetasintheta` |
|
| 14. |
What angles are associated with the following orbitals? sp,sp^(2)andsp^(3) |
|
Answer» Solution :`SP=180^(@)` `sp^(2)=120^(@)` `sp^(3)=109^(@)28` |
|
| 15. |
What is mean by meta directing group ? Give examples of it and explain meta directing effect of -NO_(2). |
|
Answer» Solution :(a) Definition : Due to presence of certain functional group on benzene ring if on second substitution of any functional group acquired meta POSITION in benzene then FIRST substituted benzene is known as meta directing group. (b) EXAMPLES : `-NO_(2), -COOH, -SO_(3)H, -CHO, -COOH_(3), -COCl, -COR, -COOR` etc are meta directing groups.  `-NO_(2)` group is present in nitrobenzene. This `-NO_(2)` group is meta directing. Following given structure (I) to (V) are resonating structures and (VI) is hybride structure.  In above given structure of nitrobenzene has positive charge of ortho and PARA position but meta poisiton is neutral. So more electro negative density is present on meta position. Therefore, postive charge electrophilic `-NO_(2)` group is attaches to meta position. So `-NO_(2)` is meta directing in nature. |
|
| 16. |
Wrtie the reasonance structure forSO_(3) ,NO_(2) and NO_(3)^(-) . |
Answer» SOLUTION :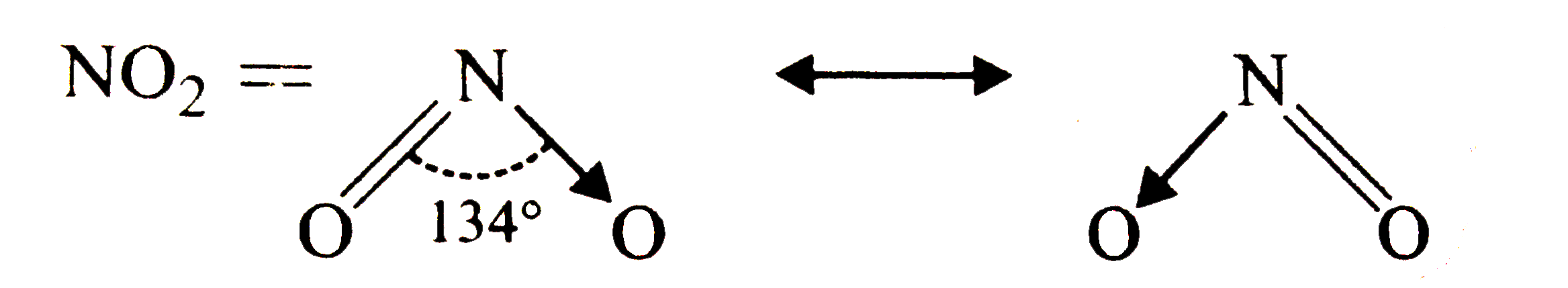 `SO_(3) and NO_(3)^(-)` = |
|
| 17. |
What is displacement reactions ? Give its types. Explain with example. |
|
Answer» Solution :The reactions in which one ion or ATOM in a compound is REPLACED (or substituted) by an ion or atom of the other element are called DISPLACEMENT reactions. AB + C `gt` AC + B Example: Metal displacement `CuSO_(4) + Zn rarr ZnSO_(4)` + Cu Example: Non-metal displacement 2KBr + `Cl_(2) rarr 2KCL + Br_(2)` |
|
| 18. |
Whichof the followingsequencescontainatomicnumbersof onlyrepresentativeelements |
|
Answer» `3, 33, 53, 87 ` |
|
| 19. |
Which group is known as p-block elements ? |
| Answer» SOLUTION :CONSEQUENTLY there are SIX groups of p-block elements in the PERIODIC table NUMBERING from 13 to 18. | |
| 20. |
What is Lindlar's catalyst ? What is it used for ? |
Answer» Solution :Pd deposited over `CaCO_3` or `BaSO_4` and partially poisoned by addition of LEAD acetate or SULPHUR or QUINOLINE is called Lindlar's CATALYST. It is used for partial reduction of alkynes to cis-alkenes . For EXAMPLE, 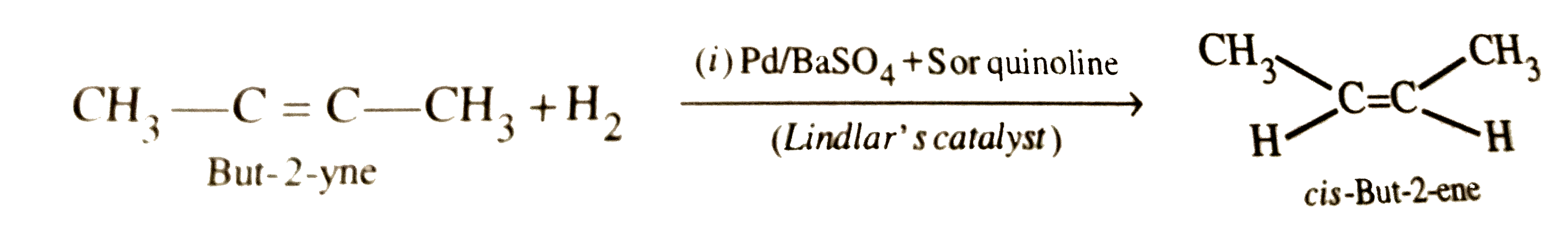
|
|
| 21. |
What are the products in the following sequence of reaction Compound (D) is |
|
Answer» `H_(2)N(CH_(2))_(4)NH_(2)` |
|
| 22. |
Which of the following pairs will have the same number of atoms ? [ atme wt. H=1, O=16, C=12, Cl=35.5 g "mol"^(-1)] |
|
Answer» 28 g CO and 36.5 g HCl `= 28 g "MOL"^(-1)` `:.28 g CO = 1 ` moleCO (ii) Molecular mass of HCL `=(1+35.5)` `= 36.5 f "mol"^(-1)` `:.36.5 g HCl = 1 ` mole HCl As CO and HCl both are 1 mole and they have same 2 atoms, hence will have same number of atoms (B) `44 g CO_(2) =1 "mole" CO_(2)` but `44g CO != 1 ` mole CO `:.` They do not have equal number of molecules (C) `14 g N_(2)` `= (14)/(28) "mole" N_(2)` `= 1/2 "mole" N_(2)` `28 g CO_(2)` `= (28)/(44) ` mole `CO_(2)` `= 0.63.63` mole `CO_(2)` `:. 14 g N_(2)` and `28 g CO_(2)` have have different moles and as a result different no. of molecules (D) `28 g N_(2)` `= (28)/(28) ` mole `N_(2)` ` = 1` mole `N_(2)` `=2` mole N atoms `18 g H_(2)O` `= (18)/(18) ` mole `H_(2)O` `= 1` mole `H_(2)O` ` = 3` mole atoms `:.N_(2)` and `H_(2)O` have different no. of atoms |
|
| 23. |
Which of the following will have the most negative electron gain enthalpy and which has the least negative? P, S, Cl, F. Explain your answer. |
| Answer» Solution :Electron gain enthalpy generally becomes more negative across a period as we move from left to RIGHT. Within a group, electron gain enthalpy becomes less negative down a group. However, adding an electron to the 2p orbital LEADS to greater repulsion than adding an electron to the LARGER 3p orbital. Hence the element with most negative electron gain enthalpy is chlorine, the ONE with the least negative electron gain enthalpy is phosphorus. | |
| 24. |
Two different acid HA and HX have same pH than their concentration is same ? Why ? |
| Answer» SOLUTION :YES, their concentration is same as their PH is same `[H^+]` is same in both and `[H^+]` = [HA]=[HX] | |
| 25. |
Which of the following statemernts pegarding projection shown below is true? |
|
Answer» Both (I) and (II) are OPTICALLY ACTIVE 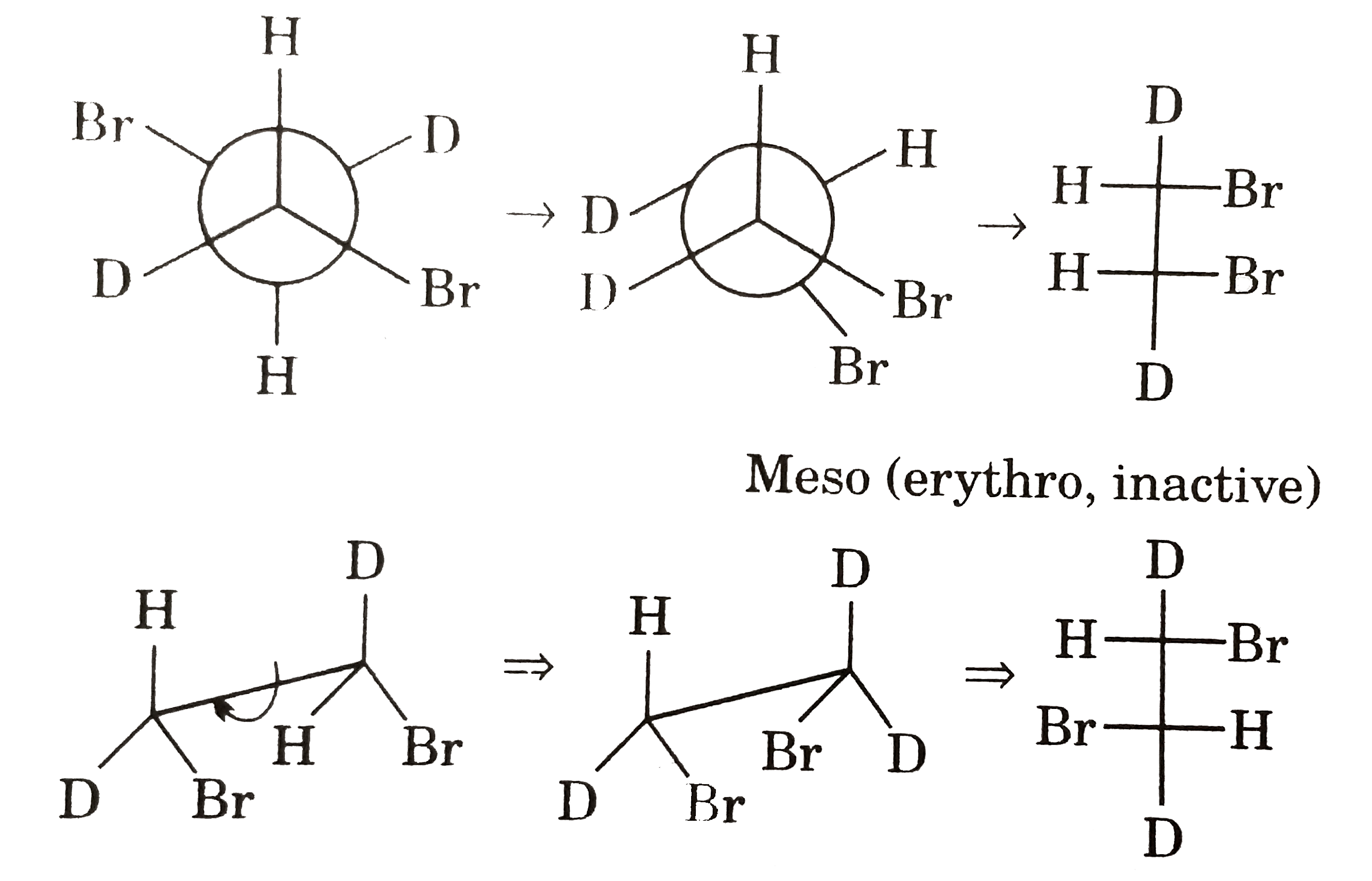
|
|
| 26. |
Which of the following statement is true about photochemical smog? |
|
Answer» it is reducing in nature |
|
| 27. |
Which of the following isomer of the pentane gives four monochloropantane on chlorination? |
|
Answer» Pyrolysis |
|
| 28. |
What is the oxidation number of S in S_(2)CI_(2) |
| Answer» SOLUTION :The O.N of S in `S_(2)CI_(2)` is +1 shown : `OVERSET(-1)CI-overset(+1)S-overset(+1)S-overset(-1)CI` | |
| 29. |
What is atomic number which contain 2 electrons in 4-orbit ? |
|
Answer» 12 |
|
| 30. |
Which of the following pair of elements are from the same group of the periodic table |
| Answer» Answer :B | |
| 31. |
You are given 500 mL of 2 N HCl and 500 mL of 5 N HCl. What will be the maximum of 3 M HCl that you can make from these two solution? |
|
Answer» 250 mL |
|
| 32. |
Which type of molecules from miselles? |
|
Answer» Non-polar MOLECULES |
|
| 33. |
Which of the following compound gives red ppt with CuCl_(2)//NH_(4)OH ? |
|
Answer» `CH_(3)-C-=C-CH_(3)` |
|
| 34. |
Volume strength of H_(2)O_(2) = ___________. |
|
Answer» `11.2 XX "NORMALITY"` |
|
| 35. |
The wavelengths of which series lie in the ultraviolet region ? |
|
Answer» Lyman |
|
| 36. |
Which of the following transitions are allowed in the normal electronic emission spectrum of an atom? |
|
Answer» `2s to 1S` |
|
| 37. |
What is the SI unit of density? |
|
Answer» `gm^(-3)` SI unit of density=`(kg)/(m^(3))` or kg`m^(-3)` |
|
| 38. |
Whichamong thefollowingtransition andwhichare innertransitionelement ? Sg, Bk, Er, Fm , Fe, Pb, Cr, Ca, Ar, Zr, Ce |
|
Answer» SOLUTION :Transitionelement `: Sg , Fe , CR, Zr` Innertransitionelement `: BK , ER , FM , Ce` |
|
| 39. |
The wrong statement regarding the solutions of alkali metals in liquid ammonia |
|
Answer» They are blue coloured because they absorb ENERGY in the visible region . |
|
| 40. |
Which of the following property does not possess of Carbon atom. |
|
Answer» It can FORM 4 COVALENT bond. |
|
| 41. |
When one s and three p orbitals hybridise, |
|
Answer» four EQUIVALENT orbitals at `90^@` to each other will be formed |
|
| 42. |
Write the relationship between equilibrium constant and enthapy. |
|
Answer» Solution :The value of equilibrium constant changes with CHANGE in TEMPERATURE. If `K_(1)` and `K_(2)` are equilibrium constants at temperature `T_(1)` and `T_(2)`. `DeltaH` - Heat of reaction at constant pressure. Then, `"log"(K_(2))/(K_(1))=(DeltaH)/(2.303R)[(T_(2)-T_(1))/(T_(1)T_(2))]` |
|
| 43. |
Which complex forms when carbon monoxide enters into body and binds with hemoglobin ? |
|
Answer» CARBOXYHEMOGLOBIN COMPLEX |
|
| 44. |
Which of the following statements is//are correct? |
|
Answer» `H_(2)O_(2)` is reduced to `O_(2)` by a strong reducing agent |
|
| 45. |
The sodium extract of an organic compound on treatement with FeSO_(4) solution, FeCl_(3) and HCl gives red solution. The Red colour of |
| Answer» Answer :C | |
| 46. |
What is avogram? |
|
Answer» Solution :AVOGRAM is reciprocal of Avogadro constant in gram Avogram `=(1)/(6.022)xx10^(23)g =1.66xx10^(-24)g` The REAL value of unified mass is avogram 1 amu= `1.66 xx10^(-24)g=1.66xx10^(-27)KG` |
|
| 47. |
Which prefix used in SI sysytem has coefficient10^(6) ? |
|
Answer» Micro 
|
|
| 48. |
The velocity of neutrons in nuclear reactor is slowed down by |
|
Answer» `H_2O` |
|
| 49. |
Which of the following forms a toxic substance in blood by combining with haemoglobin? |
|
Answer» Carbon DIOXIDE |
|
| 50. |
What is allotropy ? Give examples of allotropes. |
|
Answer» SOLUTION :Two or more forms of the same elements in same physical STATE which differ in their physical properties but have same chemical properties are called allotropic forms or ( allotropes ) and the phenomenon is called allotropy. (i) Diamod and graphite are allotropic forms of CARBON . (ii) Red phosphorous and white phosphorous are allotropes of phoshorous. (iii) Rhombic sulphur, monoclinic sulphur and plastic sulphur are allotropic forms of sulphur . |
|



