Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the empirical formula of the following? (i) Fructose (C_(6)H_(12)O_(6) ) found in honey (ii) Caffeine (C_(8)H_(10)N_(4)O_(2)) a substance found in tea and coffee. |
|
Answer» Solution :FRUCTOSE (`C_(6)H_(12)O_(6)`) Empirical FORMULA is the simplest formula. So it is DIVIDED by 6 and so empirical formula is `CH_(2)O`. Caffeine (`C_(8)H_(10)N_(4)O_(2)`) Simplified formula = `("molecular formula")/2` Empirical formula = `C_(4)H_(5)N_(2)O`. |
|
| 2. |
What are the various methods you suggest to protect our environment from pollution? |
|
Answer» Solution :Methods to control environmental pollution (i) Waste MANAGEMENT: Environmental pollution can be controlled by proper disposal of wastes. (ii) Recycling: A large amount of disposed waste materials can be reused by recycling the waste, THUS it reduces the landfill. (iii) Bysubstitution of less toxic SOLVENTS for highly toxic ones are used in industrial PROCESSES. (IV) By growing more trees. (v) By using fuels with lower sulphur content. (vi) By control measures in vehicle emissions which are adequate. |
|
| 3. |
Which is the best description of the behavior of bromine in the reaction given below ? H_(2)O+Br_(2)toHOBr+HBr |
|
Answer» PROTON ACCEPTOR only |
|
| 4. |
Which one of the following statements is false ? |
|
Answer» Raolt's law states that vapour pressure of a component over a SOLUTION is proportional to mole fraction. |
|
| 5. |
Which of the following is an acidic salt that can form an alkaline solution ? |
|
Answer» `NaHSO_4` |
|
| 6. |
Which mixture is used as Lindlar's catalyst ? |
|
Answer» Pt+Charcoal |
|
| 7. |
What happens when alcoholic KOH is treated with (i) Ethylidene dichloride (ii) Ethylene dichloride? |
|
Answer» Solution :(i) Gem-DIHALIDES and vicinal dihalides both on treatment with alcoholic KOH GIVE ALKYNES. (II) `H-underset(H)underset(|)overset(H)overset(|)C -underset(H)underset(|)overset(H)overset(|) - H + 2KOH underset(Delta)overset("ETHANOL")to underset("Acetylene")(HC-=CH) + 2KCl + H_2O` (iii) `H - underset(H)underset(|)overset(Cl)overset(|)C - underset(Cl)underset(|)overset(H)overset(|)C - H + 2KOH underset(Delta)overset("Ethanol")to underset("Acetylene")(HC-=CH) + 2KCl + H_2O` |
|
| 8. |
What transition in the hydrogen spectrum would have thesamewavelength as the Balmer transition n=4 to n=2 of He^(+) spectrum ? |
|
Answer» `n=4` to n=1 |
|
| 9. |
What is the conclusion of alpha-ray scattering experiment? |
|
Answer» Solution :The conclusion of `alpha` -ray scattering experiment is atoms inside gold are closely packed, `alpha` particles must have PASSED through the atoms. (a) Since most of the `alpha` - particles SIMPLY passed through, most part of the atom should be empty space. (b) Since a few of the`alpha` particles got DEFLECTED by small angles, they must have COME across a positive part which would repel them. (c) Since only one out of 20000 `alpha` particles retracted then path, the size of the positive part in the atom should be extremely small, only those particles which MADE a head on collision with the nucleus would retrace their path. |
|
| 10. |
Which of the following are analgesics ? |
|
Answer» Diclofen |
|
| 11. |
Which of the following methods is used for obtaining Aluminium metal ? |
|
Answer» ELECTROLYSIS of food `Al_(2)O_3` and cryolite |
|
| 12. |
What is the percentage of H in CH_(3)COOH ? |
|
Answer» 40 `% H = (4xx100)/(60) = 6.66` |
|
| 13. |
Which one of the following is used to remove silicon dioxide in the Serpeck's process of purification of bauxite ? |
Answer» Solution :`SiO_(2)+2C rarr Si UARR + 2CO uarr` 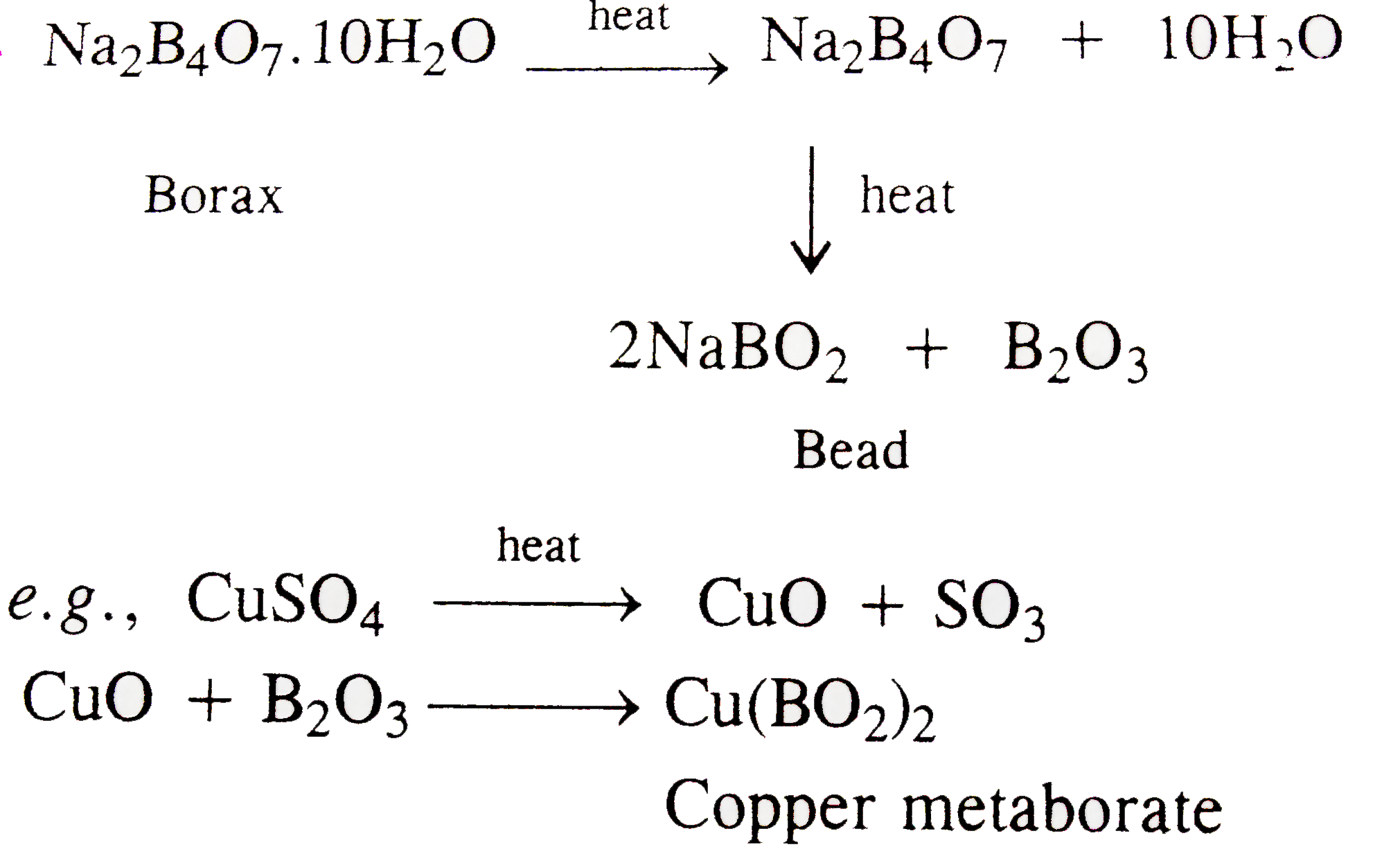 In Serpeck's process of purification of bauxite `SiO_(2)` is removed by carbon as volatile PRODUCTS. |
|
| 14. |
The volume of 40 vol. H_(2)O_(2) solution required to prepare 2L. of O_(2) at STP is |
|
Answer» 40 m.L |
|
| 15. |
The types of hybrid orbitals of nitrogen in NO_(2)^(+) , NO_(3)^(-) and NH_(4)^(+)respectively are expected to be |
|
Answer» `sp, sp^(3) and sp^(2)` The type of HYBRID orbitals of nitrogen can be calculated by using VSEPR THEORY counting bp and as Ip in `NO_(2)^(+) `= 2bp + 0lp= linear = sp hybridised `NO_(2)^(-) ` = 3bp + 0lp `rArr` linear `sp^(2)` hybridised `NH_(4)^(+)` = 4dp + 0lp `rArr` linear `hsp^(3)` hybridised |
|
| 16. |
When Lithium vapour is taken in a discharge tube and the potential difference between the electrodes is 5.4 ev, there is a sudden increase in the flow of current. The ionisation energy of Lithium is |
|
Answer» 54 EV |
|
| 17. |
Which one of the following is the strongest base in aqueous solution ? |
|
Answer» Dimethylamine |
|
| 18. |
Which element of every group shows anomalous properties? |
|
Answer» First |
|
| 19. |
The salt of an alkali metal gives yellow colour in the flame test . Also its aqueous solution gives an insolublewhite precipitate with barium chloride in acid medium . The salt is |
|
Answer» NACL `Na_(2)SO_(4) + BaCl_(2) to underset("White ppt.")(BaSO_(4)) darr + 2 NaCl` |
|
| 20. |
Out of O_(2) and O_(3), which is paramagnetic ? |
| Answer» SOLUTION :`O_(2)^(-)` is PARAMAGNETIC and `O^(2)^(2-)` is DIAMAGNETIC | |
| 21. |
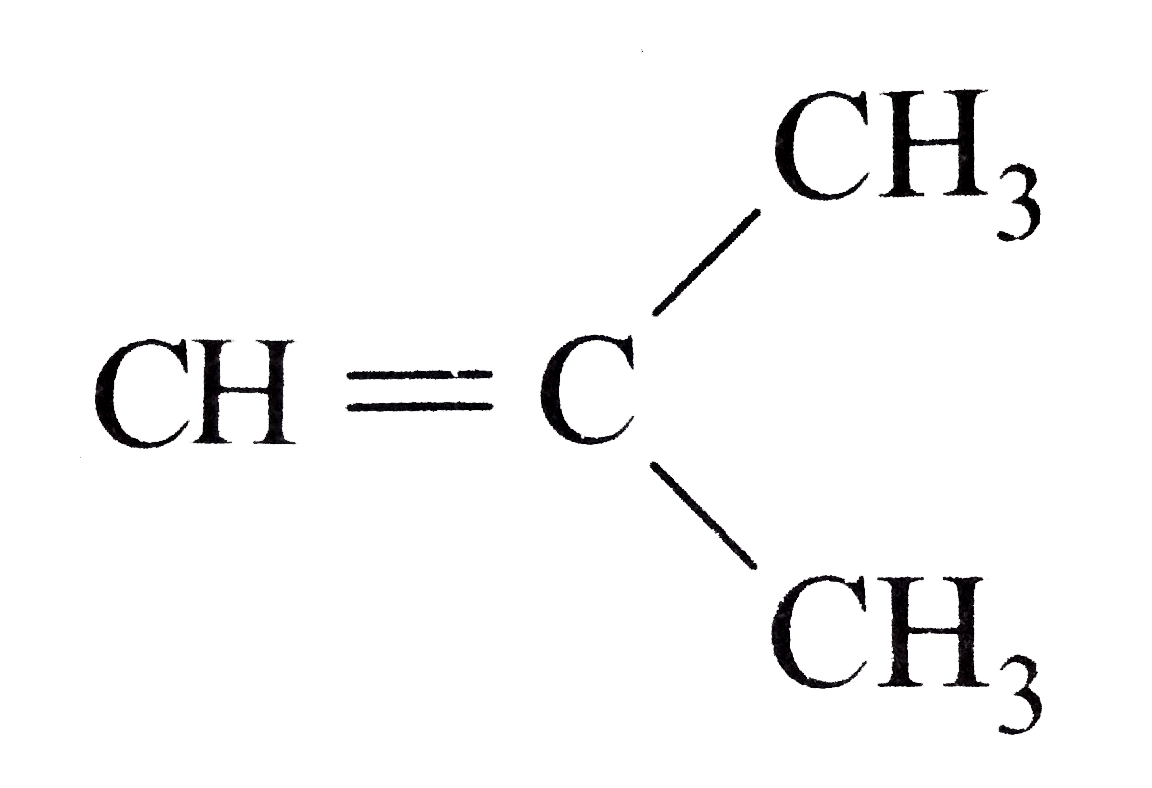
Which alkene on ozonolysis gives CH_(3)CH_(2)CHO and CH_(3)COCH_(3)? |
|
Answer»
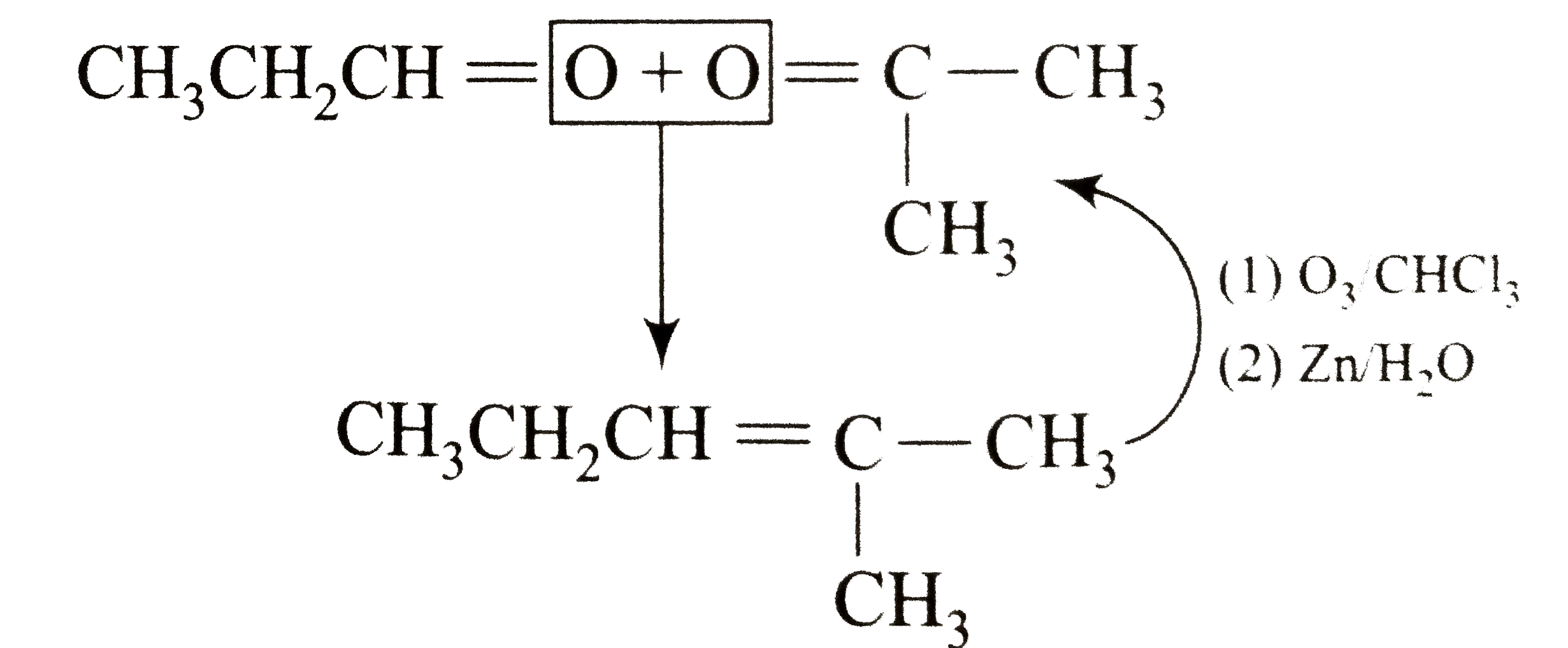
|
|
| 22. |
What is screening effect ? |
|
Answer» Solution :The repulsive FORCE between INNER shell electrons and the valence electrons leads to a decreases in theelectrostatic attractive forces acting on the valence electrons ACTS as a shield between the nucleus and the valence electrons Pauling 's scale : (i) Electronegativity is the relative tendency of an element present in a covalently bonded molecule to attract the shared pair of electrons towards itself. (ii)Pauling assigned arbitrary value of electro negativities for hydrogen and fluorine as 2.2 and 4 respectively . (iii)Based on this the electro negativity values for other ELEMENTS can be calculated using the following expression . `(X_(A)-X_(B))=0.182sqrt(E_(AB)-(E_(A A)xxE_(BB))^(1//2))` Where`E_(AB),E_(aa)andE_(bb)`are the bond dissociation energies of `AB,A_(2)andB_(2)` molecule respectively`X_(A)andX_(B)`are elctro negativity values of A and B. |
|
| 23. |
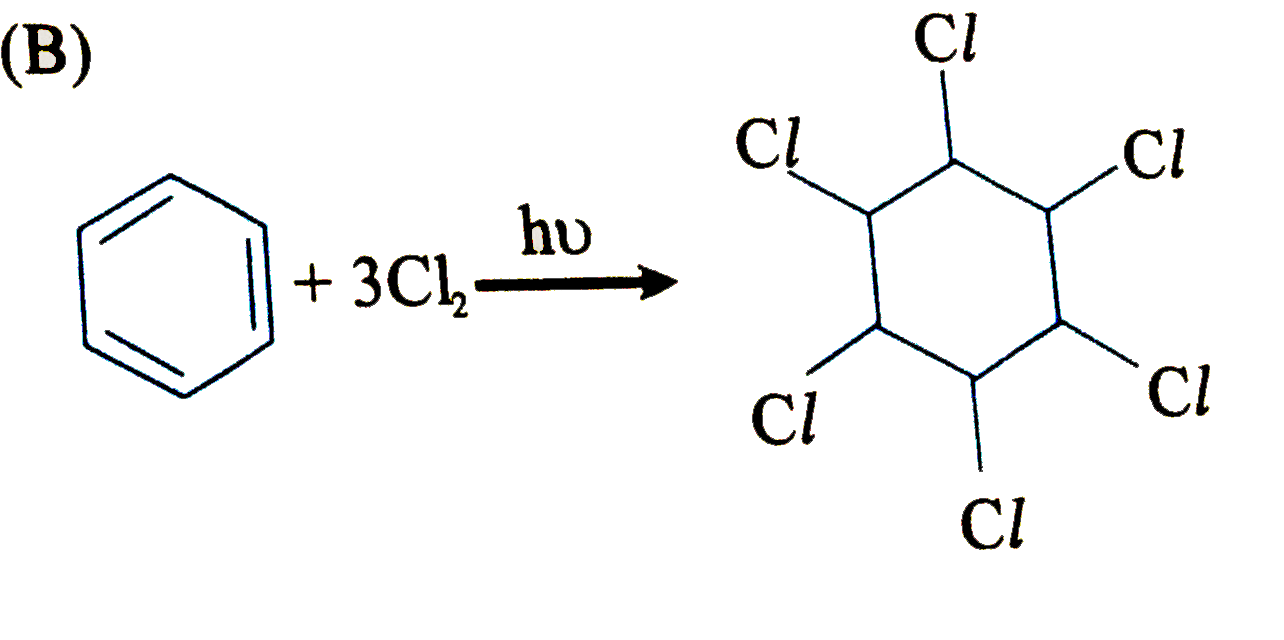
Which of the following is an electrophilic substitution reaction? |
|
Answer»
(C). Is electrophilic substitution reaction because `overset(o+)(N)O_(2)` is an electrophile. (D). Is friedel-crafts ALKYLATION i.e., electrophilic substitutio reactionb ecause `CH_(3)-overset(o+)(CH)-CH_(3)` is an electrophile. (B) is a free RADICAL ADDITION reaction.. |
|
| 24. |
Whichof thefollowingisCORRECTregardinghumanbloodplas,a ? |
|
Answer» Highconcentrationof`K^(+)` |
|
| 25. |
Write the formal charges on atoms in (i) and carbonate ion (ii) nitrite ion. |
Answer» Solution : (i) Lewis structure of ` CO_(3)""^(2-)` ions is 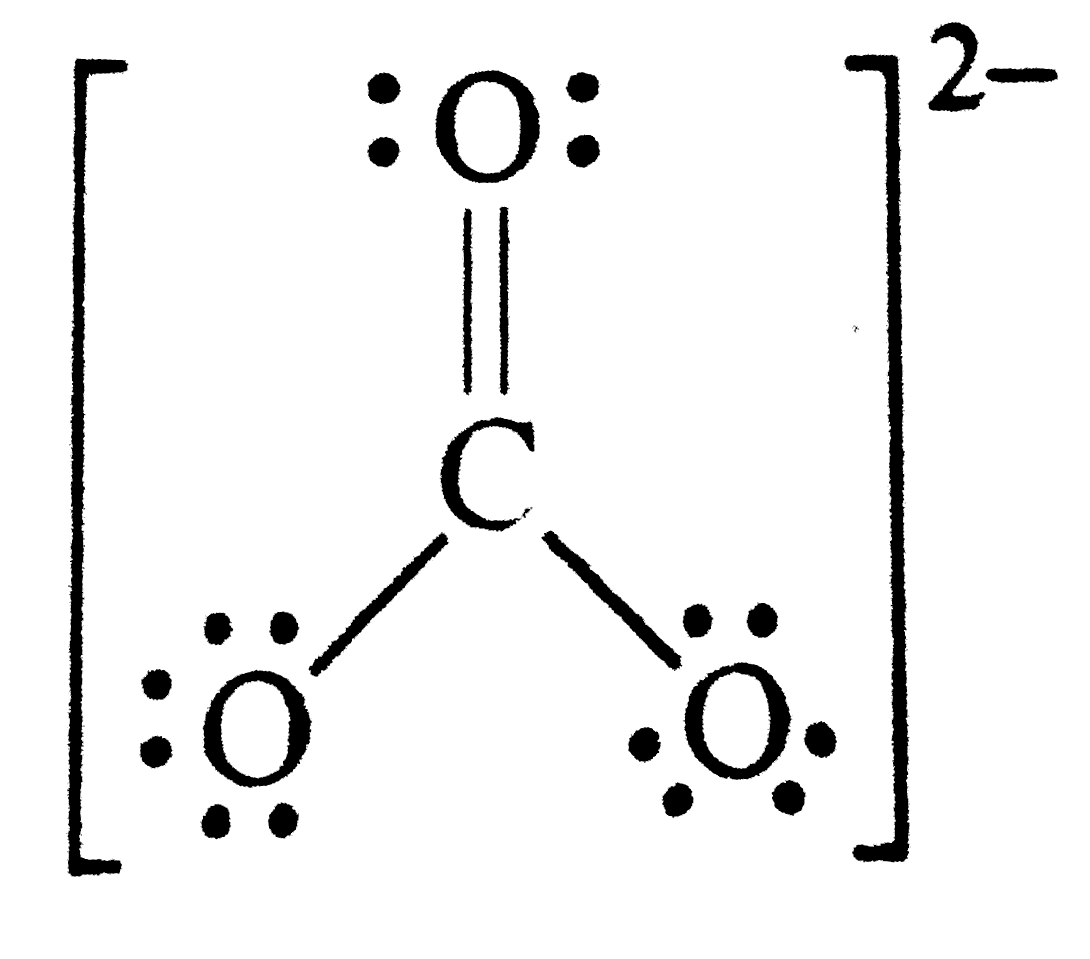 Formal charge on C atom ` = 4 - 0 - (1)/(2) (8) = 0 ` , formal charge on double BONDED O atom = ` 6 - 4 - (1)/(2) (4) = 0 ` Formal charge on single bonded O atom ` = 6 - 6 - (1)/(2) (2) = - ` (ii) Lewis structure of ` NO_(2)^(-)`ions is  Formal charge on N atom = ` 5 - 2 - (1)/(2) (6) = 0 ` , Formal charge on double bonded O atom ` = 6 - 4 - (1)/(2) (4) = 0 ` Formal charge on single bonded O atom `= 6 - 6 - (1)/(2)= - 1 ` Singnificance of formalcharge . The MAIN advantage of the calculation of formal charges is that it helps to select the most stable structure, i.e., the one with least energy out of the different POSSIBILE Lewis structure. The most stable is the one which has the SMALLEST formal charges on the atoms. |
|
| 26. |
Which of the following pairs of species have the same bond order? |
|
Answer» `CN^(-)` and `CN^(+)` |
|
| 27. |
Write formulas for the following compounds: a) Mercury (II) chloride |
| Answer» SOLUTION :HG (II) `Cl_2` | |
| 28. |
When a standard solution of NaOH is left in air for a few hours |
|
Answer» A PRECIPITATE will form |
|
| 29. |
Which of the following compound used for metal cleaning solvent? |
|
Answer» METHYLENE chloride |
|
| 30. |
Which of the following has higher mass |
|
Answer» electrons |
|
| 31. |
What will be the number of hydrogen atoms (Y) replaced by D on prolonged treatment with D^(o+)//D_(2)O ? |
|
Answer» |
|
| 32. |
Which of the following is an intermediate in the reaction of benzene with Br_2 and AlBr_3 ? |
|
Answer»
|
|
| 33. |
Which pairs of the salts should have identical solubilities in methanol? |
|
Answer» P and D |
|
| 34. |
Which of the following species is not stable? |
|
Answer» `[SN (OH)_6]^(2-)` |
|
| 35. |
What are hybridisation states of each carbon atom in the following compounds? CH_2 = C = O, CH_3CH=CH_2(CH_3)_2CO, CH_2= CHCN, C_6H_6 |
|
Answer» |
|
| 36. |
Which gas has contribution of 8% in globl warming ? |
|
Answer» CFC |
|
| 37. |
What are the oxidation numbers of : (a) S in H_(2)SO_(4), (b) Mn in MnO_(4)^(-) (c) N in NH_(4)^(+) and (d) Al in A1O_(2)^(-) (e) Mn in MnO_(4)^(-2) |
|
Answer» Solution :(a) 2 (Oxidation number of H)+(Oxidation number of S)+ 4(-2)=0 Oxidation number of S=-2+8=+6 (b) (Oxidation number of Mn)+4(-2)=-1 Oxidation number of Mn =-1+8=+7 (c ) (Oxidation number of N) +4(+1)=+1 Oxidation number of N =+1-4=-3 (d) (Oxidation number of AL)+2(-2)=-1 Oxidation number of Al =-1+4=+3. (E) (Oxidation number of Mn) +4(oxidation number of oxygem)=-2 Oxidation number of Mn=-2+8=+6 |
|
| 38. |
Write the cell reaction for each of the following cells. Ni(s)|Ni^(2+)(aq)||Cu^(2+)(aq)|Cu(s). |
| Answer» Solution :`NI(s)+CU^(2+)(AQ)TONI^(2+)(aq)+Cu(s)` | |
| 39. |
Which acid will not form hydrocarbon on soda lime decarboxylation ? |
|
Answer» Cinnamic acid 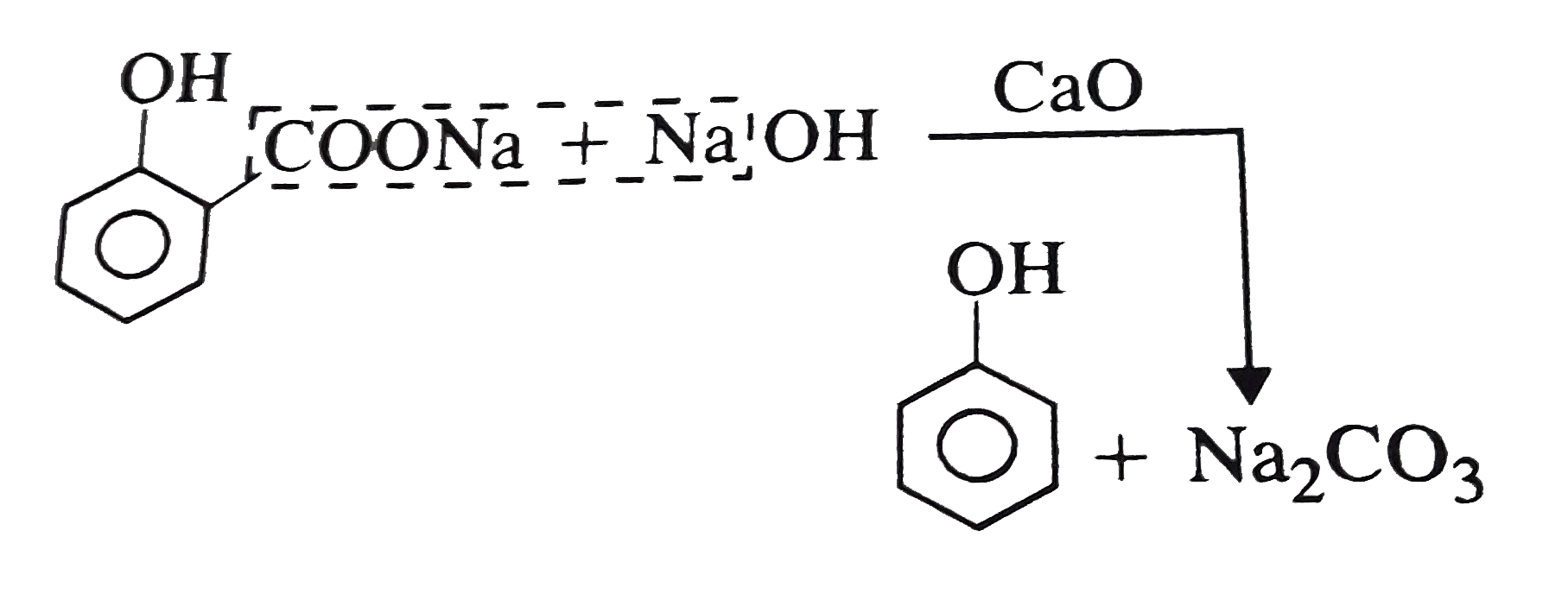
|
|
| 40. |
Which of the following species contains three bond pairs and one lone pair around the central atom- |
|
Answer» `H_(2)O` 
|
|
| 41. |
Write the significance/applications of dipole moment. |
|
Answer» Solution :In determining the POLARITY of bond As `mu = a xx d` obviously greater the magnitude of dipole moment the polarity of bond will be more. For one polar bond A - B TYPE molecule. H - F `gt H - Cl gt H - Br gt H - I and " if" mu = 0 ` then `mu= 1.78` D, HCL `mu` = 1.07 , H - Br = 13.79 D, H- I = 0.38 D molecule is non polar. e.g. `O_(2), N_(2), F_(2),Cl_(2), Br_(2), Z_(1), H_(1) ` etc. `BeF_(2) , CO_(2), BeCl_(2)` forsuch molecule `mu` = 0 so they are linear do, but `H_(2)`O. `SO_(2) mu ne 0` So they must have angular shapeSimilarly `BF_(3), CH_(4), "CCl"_(4)` non polar but `NF_(3),NH_(3), CH_(3)Cl` are polar so their shape can be determine. In CALCULATION of ionic or covalent characters : Ionic character`prop` Difference of electronegativity. ln HI, HBr, HCl, HF ionic character increase by using felon rules ionic character can be calculated. To distinguish between cis & trans isomers for cis isomers `mu ne 0` for trans isomer `mu = 0 `. To distinguish between ortho, META and para isomers for paraisomers `mu` = 0 and dipole moment of ortho is greater than meta. |
|
| 42. |
Which of the following options reprsents the correct bond order ? |
|
Answer» `O_(2)^(-) GT O_(2) LT O_(2)^(+)` |
|
| 43. |
What are conformers |
| Answer» SOLUTION :Therotationabout L `C-C` singlebondaxisyielding severalarrangementsof ahydrocacalledconformers. | |
| 44. |
Which will be the least stable resonating structure ? |
|
Answer» `CH_(2) = CH - overset(OPLUS)CH - overset(Theta)CH-NH_(2)` |
|
| 45. |
Which of the following statement is incorrect regarding the complete hydrolu=ysis of Marshall's acid? |
|
Answer» CARO's acid is an intermediate product |
|
| 46. |
Which of the following acts as reducing as well as oxidising agent ? |
|
Answer» `NaNO_(3)` |
|
| 47. |
The total number of compounds having atleast one bridging oxo group among the molecules given below is ……….. . N_(2)O_(5),N_(2)O_(3),P_(4)O_(6),P_(4)O_(7),H_(4),P_(2),O_(5),H_(5)P_(3)O_(10),H_(2)S_(2)O_(3),H_(2)S_(2)O_(5) |
Answer»  If `N_(2)O_(3)` is ALSO INCLUDED, then total number of compounds is |
|
| 48. |
What is the vapour pressure of water at 373 K ? |
|
Answer» SOLUTION :373 K is the boiling point of water. At the boiling point, the vapour pressure of any liquid is EQUAL to the ATMOSPHERIC pressure. The vapour pressure of water at 373 K is 760 mm Hg. |
|
| 49. |
Which of the following statement is incorrect for compounds of graup 2 meters? |
|
Answer» The number of MOLECULES of water of crystllistoion INCREASES with size of the metal IONS. |
|
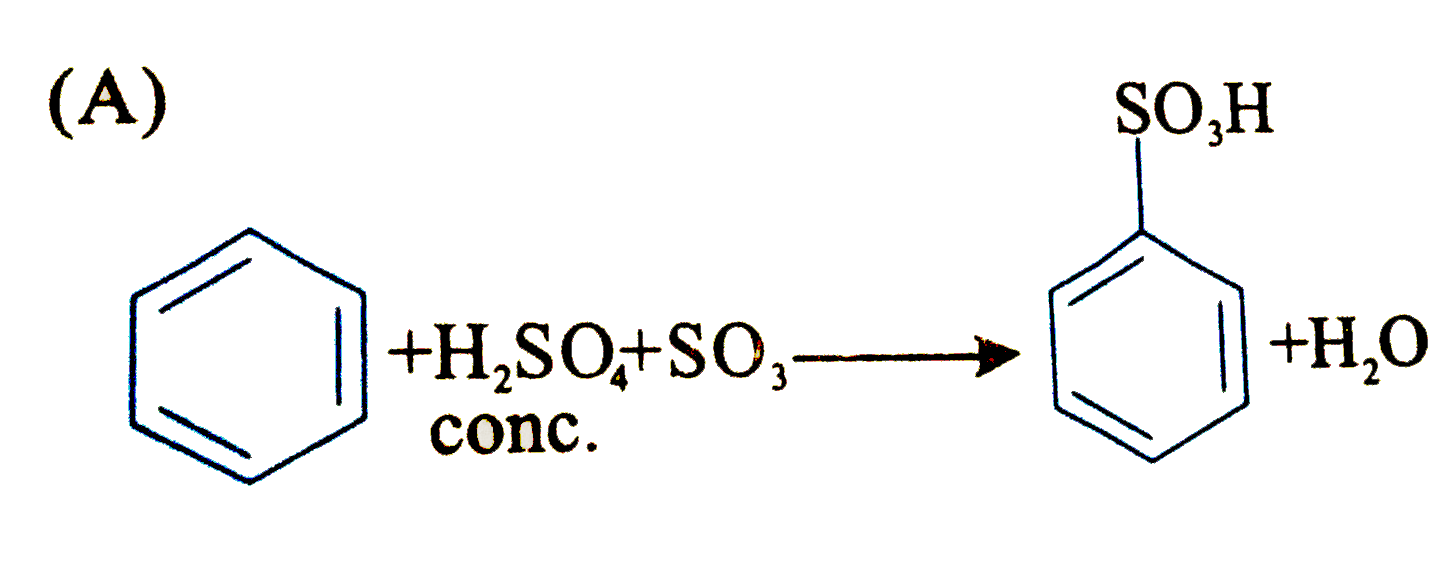
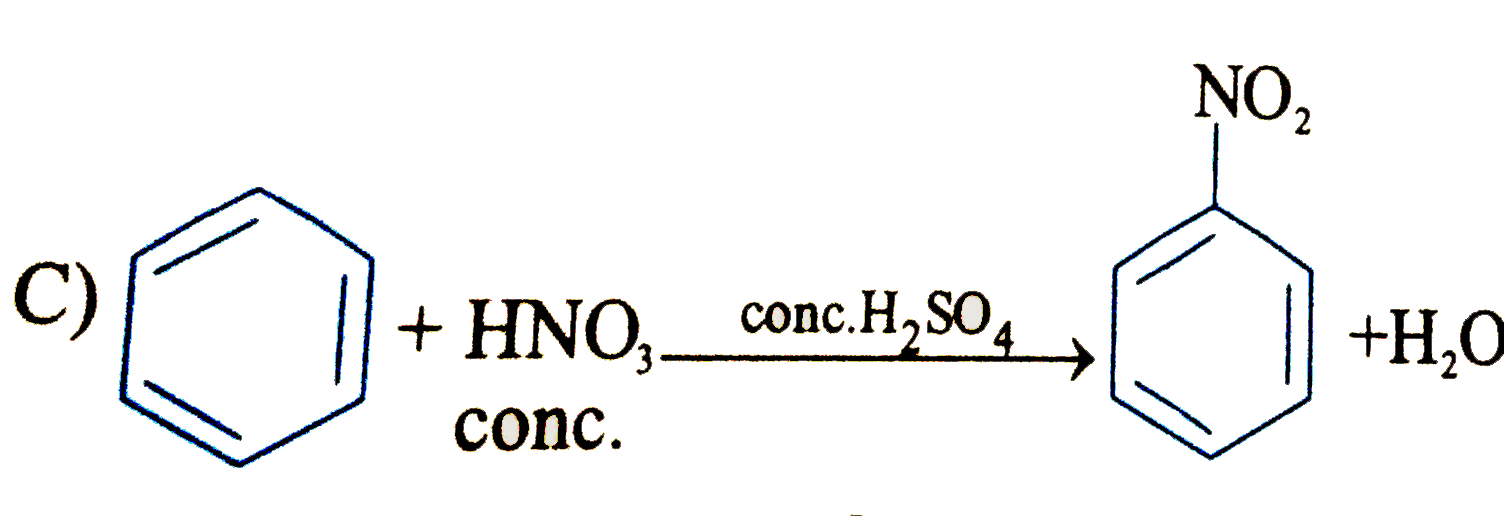

 <
<

