Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is name of K_(2)Cr_(2)O_(7) according to stock notation method ? |
| Answer» SOLUTION :POTASSIUM DICHROMATE (VI) | |
| 2. |
Which of the following halides are stable |
|
Answer» `SiCl_(4)` |
|
| 3. |
Two solids X and Y dissociate into gaseous products at a certain temperature as follows: i. X(s) hArr A(g)+C(g) and ii. Y(s) hArr B(g)+C(g) At a given temperature, pressure over excess solid 'X' is 40 mm of Hg and total pressure over solid 'Y(s)' is 60 mm of Hg. Now, answer the following questions: The total pressure of gaseous over a mixture of solids X and Y is: |
|
Answer» `100` mm `K_(p_(2))=p_(2)(p_(1)+p_(2))` On solveing `p_(1)=23.09, p_(2)=51.75` `:. p_(1)+p_(2)=74.84 mm` Hence, (b) is the correct ANSWER. |
|
| 4. |
Vacuum distillation is used to purify liquids which |
|
Answer» Are HIGHLY volatile |
|
| 5. |
What is the direction of flow of electrons in a galvanic cell? |
| Answer» SOLUTION :From ANODE to CATHODE | |
| 6. |
Weak acids dissociate partially in aqueous medium. If any common ion is present in the solution, the degree of dissociation of the acid is suppressed however the dissociation constant value remains constant. What is the degree of dissociation of water is 0.0IM HCOOH (K_(a) = 10^(-6)) at 25^(@) C? |
|
Answer» ` 1.8 xx 10 ^(-12) ` ` " at "25^(@) C ,K_W =[H^(+) ] [OH^(-) ]` ` 10 ^(-14) =(10 ^(-14))[OH^(-)]` ` [OH^(-) ]=10 ^(-10) M` , In water `[H^(+) ] =10 ^(-10)M` ` alpha =( 10 ^(-10))/( 55.5 )rArr alpha = 1.8 xx 10 ^(-12) ` |
|
| 7. |
What happens if a liquid is heated to the critical temperature of its vapour? |
| Answer» SOLUTION :`P=dRT//M` | |
| 8. |
Weak acids dissociate partially in aqueous medium. If any common ion is present in the solution, the degree of dissociation of the acid is suppressed however the dissociation constant value remains constant. In which of the following the degree of dissociation of water is maximum '? |
|
Answer» `0.1 M NH_3` |
|
| 9. |
Which elements properties are almost same ? |
| Answer» SOLUTION :f-block ELEMENTS [INNER TRANSITION elements] | |
| 10. |
Which method can be used to find out strength of reductant / oxidant in a solution ? Explain with an example |
| Answer» Solution :The METHOD used to find out the strength of REDUCTANT / oxidant INSOLUTION is to connect the REDOX couple of as the reductant and if it is negative it ACTS an oxidant compare the values and determine their comparative strength as an oxidant or reductant | |
| 11. |
The wave nature of an electron was first given by |
|
Answer» de-Broglie |
|
| 12. |
Which one is buffer solution ? |
|
Answer» NAOH and NaCl |
|
| 13. |
Write the correct orbital notations for each of the following sets of quantum numbers: (i) n = 2, l = 1, (ii) n = 3, l = 0 (iii) n = 5, l = 3 and (iv) n = 4, l = 2 ? |
|
Answer» <P> SOLUTION :(i) 1s (II) `P_(y)` (iii) `3 d_(xz)` |
|
| 14. |
Which group has higher priority order than CHO group? |
|
Answer» Amide |
|
| 15. |
Which of the following statements are correct with respect to bond order ? (i)Bond order of N_(2) " is " 3.5 (ii) Bond order of O_(2) " is " 2 (iii) Bond order of NO^(+)" is " 3.0 (iv) Bond order of NO is 2.5 |
|
Answer» Only (i) |
|
| 16. |
Which quantum number determines the (a) shape (b) orientation and (c) size of the orbital ? |
| Answer» SOLUTION :(a) AZIMUTHAL (b) MAGNETIC (C) PRINCIPAL | |
| 17. |
Which of the following acids is most abundant in acid rain ? |
|
Answer» HCI |
|
| 18. |
This section contains 2 questions. Each question contains statement given in two columns which have to be matched. Statements (a, b, c and d) in Column-I have to be matched with statement (p, q, r and s) in Column-II. The answers to these questions have to be appropriately bubbled as illustrated in the following examples: If the correct matches are (a-p, s), (c-p, q) and (d-s), then correct bubbled 4 xx 4 matrix should be as follows: Match the Column-I with Column -II |
|
Answer» <P> |
|
| 19. |
Which of the following substituted benzene derivative would furnish three isomers when one more electrophilic substituent is introduced? Select the correct answer using the codes given below. |
|
Answer» (i) |
|
| 20. |
This section contains 2 questions. Each question contains statement given in two columns which have to be matched. Statements (a, b, c and d) in Column-I have to be matched with statement (p, q, r and s) in Column-II. The answers to these questions have to be appropriately bubbled as illustrated in the following examples: If the correct matches are (a-p, s), (c-p, q) and (d-s), then correct bubbled 4 xx 4 matrix should be as follows: Match the Column -I with Column -II |
|
Answer» <P> |
|
| 21. |
When the chemical bond is formed ..... |
|
Answer» ELECTRON and Nucleus repulsion increases. Bond formation is always EXOTHERMIC so when the bond from energy decreases. |
|
| 22. |
Which one of the following compounds would not be easilty oxidised by K_(2)Cr_(2)O_(7) in dil. H_(2)SO_(4) ? |
|
Answer» `CH_(3)OH` |
|
| 23. |
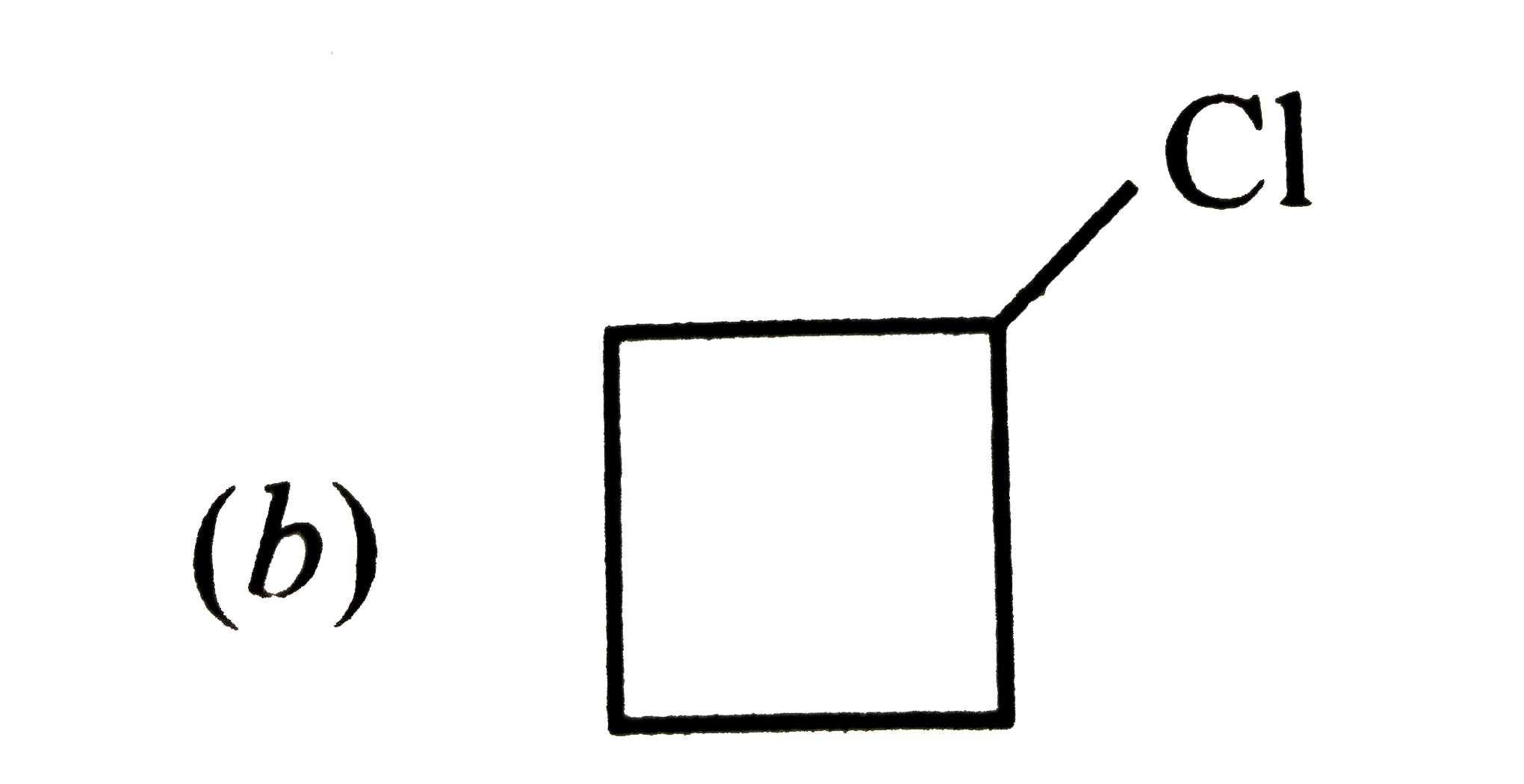
What will be the product formed when 1-bromo-3-chlorocyclobutane reacts with two equivalents of metallic sodiumin ether ? |
|
Answer»
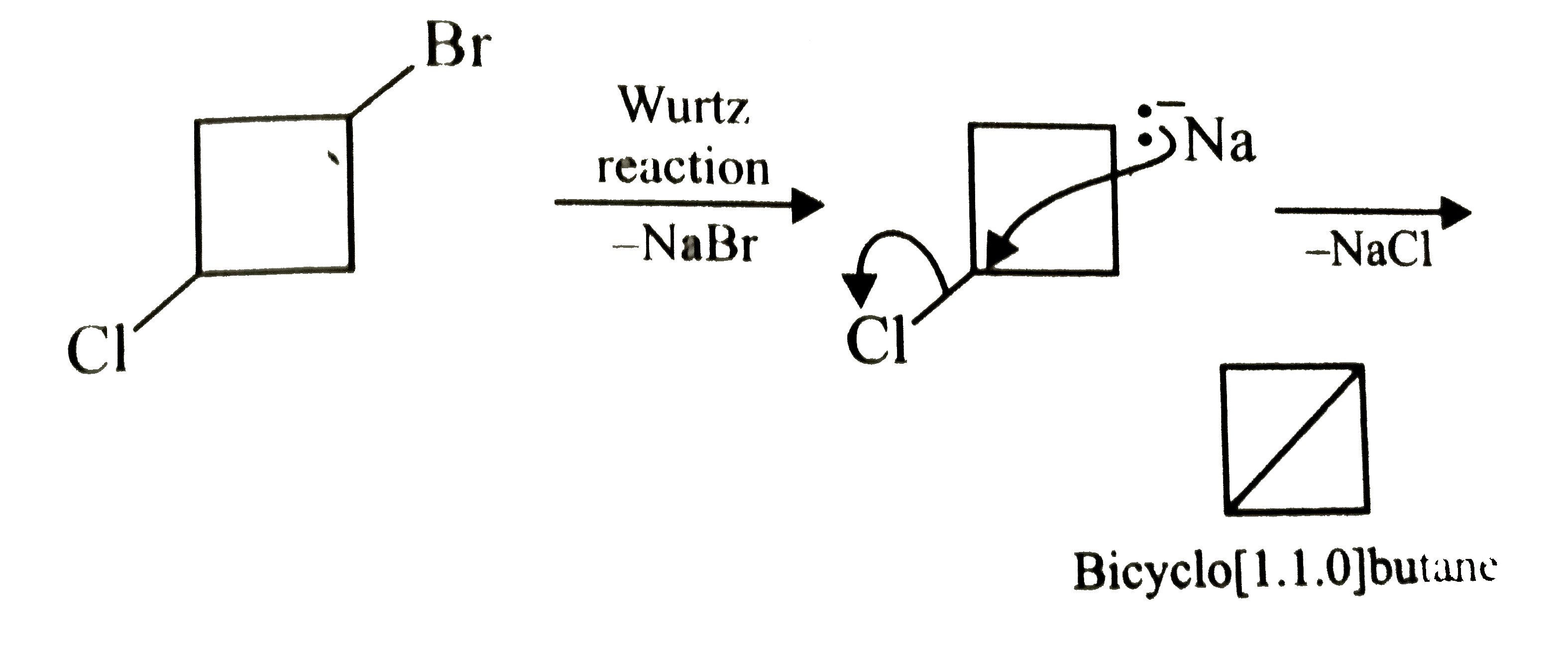
|
|
| 24. |
Which of the following sets of compounds correctly illustrate the law of reciprocal proportions ? (1) P_2O_3. PH_3, H_2O (2) P_2O_5, PH_3, H_2O (3) N_2O_5, NH_3, H_2O (4) N_2O, NH_3, H_2O (5) NO_2, NH_3, H_2O |
|
Answer» `N_(2)O_(5),NH_(3),H_(2)O` |
|
| 25. |
Which statement is not correct ? |
|
Answer» `sigma` -bond is WEAK than `PI` - bond |
|
| 27. |
Two flasks of equal volume connected by a narrow tube of negliglible volume are filled with N_(2) gas. When both are immersed in boiling water, the gas pressure inside the system is 0.5 atm. Calculate the pressure of the system when one of the flasks is immersed in an ice-water mixture keeping the other in boiling water. |
|
Answer» Solution :TEMP. of the gas when both the flasks are immersed in boiling WATER`=100^(@)C=373" K"`. Pressure=0.5 atm Average temp. of the gas when one flask is immersed in ice and the other in boiling water `=(0+100)/(2)=50^(@)C=323" K"` As VOLUME remains constant, `(P_(1))/(T_(1))=(P_(2))/(T_(2))` `(0.5)/(373)=(P_(2))/(323) "or" P_(2)=(323)/(373)xx0.5=0.433" atm"`. |
|
| 28. |
Which of the following statement is not true about the long form of periodic table ? |
|
Answer» It reflects the sequence of FILLING of electrons in order of sub-energy LEVELS. s, p, d and f. |
|
| 29. |
Which one of the followingelementsis unable toform MF_(6)^(3-) ion ? |
|
Answer» Ga |
|
| 30. |
Thesplittingof thespectrallinesunder theinfluenceof magneticfieldis called ……. |
|
Answer» Photoelectriceffect |
|
| 31. |
Which will have enantiomers |
|
Answer» n-BUTYL CHLORIDE |
|
| 32. |
What is Isothermal Curve ? Give isothermal curve presenting Boyle.s law. |
|
Answer» SOLUTION :Pressure (p) `to` Volume (V) isothermal graph : Graph of pV = constant at constant temperature according to (Eq. -iii) is given below : This graph is CURVE. When temperature (T) changes (200K, 400K, 600K) different types of curve obtained. Value of `k_(1)` changes with different tempe-rature. ..Temperature of fixed amount (n) of any gas changes than value of `k_(1)` changes... Isotherm graph : EVERY curve of constant temperature is known as isothermal curve. Isothermal graph means, Pressure (p) `to` Volume at constant (V) temperature. This isothermal curve is according to high temperature. Graph of `p to (1)/(V)` : At constant temperature graph amount of any gas is give below :It is a straight line and passing through origin. Plot of graph increases value of K increases of `T_(3)gt T_(2)gt T_(1). therefore` The graph is isothermal. |
|
| 33. |
The vapour pressure of a liquid in a closed vessel |
|
Answer» depandes upon the amount of the liquid taken s |
|
| 34. |
Which alkali metal forms nitride by direct reaction with nitrogen of air ? |
|
Answer» SOLUTION :`6Li +N_(2) to 2Li_(3)N` `3Mg+N_(2) to Mg_(3)N_(2)` |
|
| 35. |
Which kind of defect are introduced by doping ? |
|
Answer» Dislocation defect |
|
| 36. |
X reacts with aqueous NaOH solution to form Y and H_(2). Aqueous solution of Y is heated to 323-33 K and on passing CO_(2) into it,Na_(2)CO_(3) and Z were formed. When Z is heated to 1200^(@)C, Al_(2)O_(3) is formed. X, Y and Z respectively are |
|
Answer» `Al, AlCl_(3), NaAlO_(2)` (Hall's process for purificationof bauxite) |
|
| 37. |
Which of the following statements related to the modern periodictable is incorrect ? (a)The p-blockhas sic columns because a maximum of6 electronscan occupy alltheorbitals ina p - subshell. ( b)The d-blockhas 8columns becausea maximum of 8 electrons canoccupy all theorbitals in ad- subshell. ( c) Eachblockcontain a numberof columnsequal to the numberof electronsthat canoccupy thatsubshell. ( d) The blockindicatesvalue ofazimuthal quantum number(l)for thelastsubshellthatreceivedelectrons in buildingup theelectronic configuration . |
| Answer» Solution :STATEMENT ( B)is incorrectwhileotherstatement are correct. The CORRECTSTATEMENT( b) is : thed- blockhas10 columns because a maximum of 10electrons canoccupyall THEORBITALS in a d- SUBSHELL. | |
| 38. |
What is the average velocity of H_(2) molecules at 100K relative to their velocity at 50K? |
|
Answer» 2.00 TIMES the VELOCITY at 50K |
|
| 39. |
X+2KOHoverset("Alcohol")rarrH-C-=C-H Here the reactant ''X' is |
|
Answer» 1,1-Dibromoethane |
|
| 40. |
When formic acid is treatedwith conc.H_(2)SO_(4), the gas evolved is : |
| Answer» SOLUTION :`HCOOH overset("CONC."H_(2)SO_(4))rarr CO + H_(2)O` | |
| 41. |
Which one of the following reacts with CH_3Mgl followed by hydrolysis to yield tert. butyl alchol? |
|
Answer» `CH_3CHO` |
|
| 42. |
Which of the following statements regarding spectral series is correct? |
|
Answer» The lines in the BALMER series CORRESPOND to the electronic transition from higher energy level to n = 1 energy level. |
|
| 43. |
What is London force ? Give its characteristics ? |
|
Answer» Solution :London force : Attraction FORCES present between symmetric atoms or molecules are knows as London forces or dispersion forces. This force of attraction was first proposed by the GERMAN physicist Fritz London, and for this reasson force of attraction between two temporary dipoles is KNOWN as London force. Characteristics : Another name for this force is dispersion force. It is present between symmetric atom or molecules. e.g., `H_(2),Cl_(2),P_(4),CH_(4),C Cl_(4), C_(6)H_(6)` There is always attraction forces. These forces are always asttractive and INTERACTION energy is inversely proportional to the sixth POWER of the distance between two interacting particles. (`1//r^(6)` where r is the distance between two particles) These forces are important only at short distances (~ 500 pm) and their magnitude depends on the polarisability of the particle. |
|
| 44. |
What is the colour imparts when calcium, Barium and strontium undergoes glame test? |
|
Answer» Solution :CALCIUM = Brick red BARIUM = APPLE green STRONTIUM = Crimson red |
|
| 45. |
Which of the following is incorrect statement about S_(N^(2)) reaction mechanism? |
|
Answer» It FOLLOWS second ORDER kinetics |
|
| 46. |
Which of the followingcompounds give white ppt. with dil. HCI? |
|
Answer» `PB(OAC)_(2)` |
|
| 47. |
Which of the following reactions is expected to radily give a hydrocarbon product in good yield? |
|
Answer» `(CH_3)_3C-CIoverset(C2H_5OH)(to)` |
|