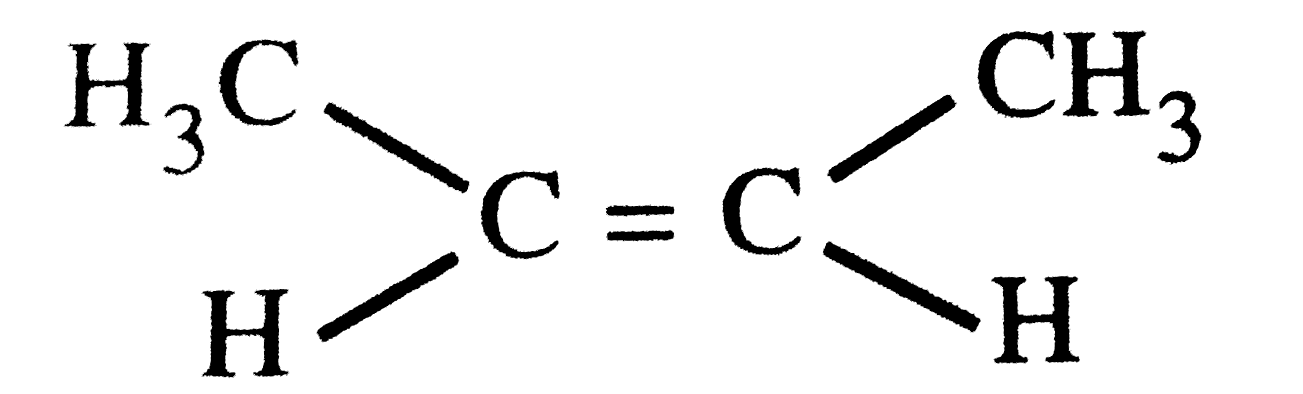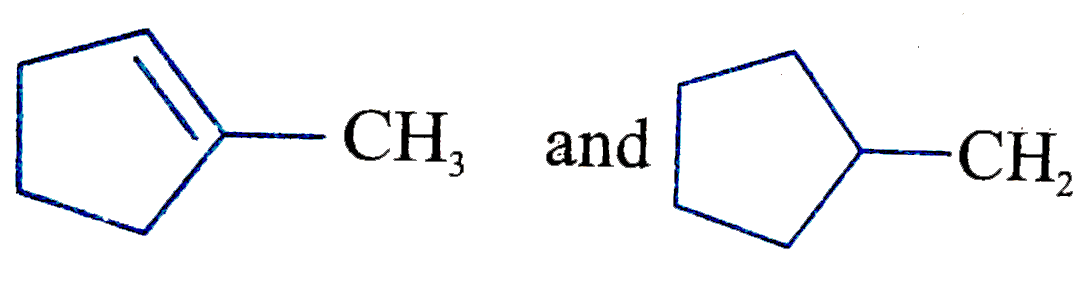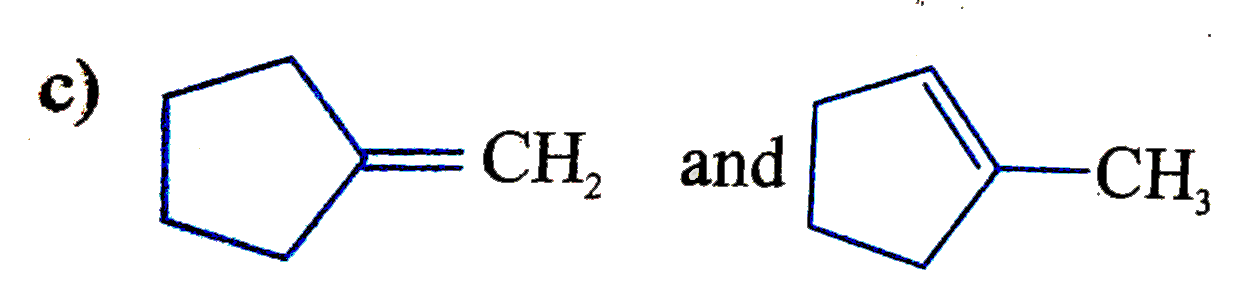Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the pH of a solution obtained by dissolving 0.0005 mole of the strong electrolyte, calcium hydroxide, Ca(OH)_(2) , to form 100 ml of a saturated solution (aqueous)? (K_(w)=1.0xx10^(-14) "mole"^(2) "litre"^(-2)) |
|
Answer» `9.8` ` THEREFORE N= M xx n = 0.01 M` ` [OH^(-)] =10 ^(-2)N rArr pOH =2rArr pH =12` |
|
| 2. |
What indicate the value of K_a ? What is its dimension ? |
| Answer» Solution :`K_a` is KNOWN as DISSOCIATION constant of weak ACID. `K_a` is dimension LESS. | |
| 3. |
What do you understand by bond pairs and lone pairs of electrons ? illustrate by giving one example of each type. |
|
Answer» Solution :Bonding ELECTRON pair : In molecule the electron pair which FORM bond between two atom is CALLED bonding electron pair. Nonbondlng electron pair : The electron pair which does not take PART in bond formation in molecule is called non bonding electron pair : eg. Lewis structure of `H_(2)O, ` 
|
|
| 4. |
What type of isomerism is shown by the But-1-yne and But-2-yne? |
|
Answer» |
|
| 5. |
Which of the following ions will cause hardness in water ? |
| Answer» Answer :A | |
| 6. |
What is temporary hardness of water? How is it removed? |
|
Answer» Solution :Temporary hardness of WATER is due to the presence of soluble bicarbonates of magnesium and calcium. By heating / boiling, these salts decomposed into insoluble carbonate and hydroxide, respectively. The RESULTING precipitates can be removed by filtration. `Ca(HCO_(3))_(2)_(aq)toCaCO_(3(s))+H_(2)O_((l))+CO_(2(g))` `Mg(HCO_(3))_(2)_(aq)toMg(OH)_(2(s))+CO_(2(g))` |
|
| 7. |
Two isomer of butane are n-butane and iso butane……… is the relation of no. of sigma and pi bond in it. |
|
Answer» |
|
| 8. |
Which of thefollowing will produce a buffer solution when mixed in equal volumes ? |
|
Answer» 0.1 MOL `dm^(-3) NH_(4)OH` and 0.1 mol `dm^(-3)` HCl |
|
| 9. |
What is buffer solutions ? Explain types of buffer solutions by examples. |
|
Answer» Solution :DEFINATION : The solutions which resist change in pH on dilutionor with the addition of small amounts of acid or ALKALI are called Buffer solutions. Buffer solutions are two types like acidic and basic. (A)Acidic buffer solutions : The pH value of this solutions is less than 7.0 . The examples of acidic buffer as under.  pH of a acidic buffer is CALCULATE as following : pH=`pK_a + log "[Salt]"/"[Acid]"`...(Eq.-i) `pH=pK_a + "log" (["Conjugate base" A^-])/"[Acid HA]"`....(Eq.-ii) In it `[H_3O^+]=K_a ([HA])/([A^-])` ....(Eq.-iii) This equation is KNOWN as Henderson Hasselback equation . Here weak acid HA is ionised in less amount. (B) Basic buffer solutions : The pH value of buffer solutions is more than 7.0 is known as basic buffer solution . Basic buffer is formed by mixing of weak base and its salt of strong acid. Examples of basic buffer :  pH of basic buffer is calculate by following : `pH=pK_w`-(`pK_b` + log salt/base) ...(Eq.-i) pH=`pK_a - "log" (["Conjugate acid" BH^+])/"[Base B]"`....(Eq.-ii) pOH=`pK_b+"log" (["Conjugate acid" BH^+])/"[Base B]"` ...(Eq.-iii) `=pK_b+ "log" "[Salt]"/"[Base]"` ...(Eq.-iv) The pH of buffer solution after dilution does not change as the ratio of exponent factor remain unchange. |
|
| 10. |
Which of the thermodynamic properties out ofE,S,T,P,V,H and G are intensive propertiesand why ? |
| Answer» SOLUTION :T and PARE intensive propertiesbecause they DEPEND only UPON the NATURE of the substance. | |
| 11. |
Which of the following redox reaction is oxidation and which is reduction? Cl_2+2e^(-)to2Cl^(-) |
| Answer» SOLUTION :REDUCTION | |
| 12. |
Which one of the following exhibits geometrical isomerism ? |
|
Answer» 1,2-dibromopropene |
|
| 13. |
Why a non- spontaneous reaction becomes spontaneous when coupled with a suitable spontaneous reaction ? |
| Answer» Solution :The OVERALL FREE ENERGY change for the coupledreaction is NEGATIVE. | |
| 14. |
What is the osmotic pressure of the solution obtained by mixing 300 cm^(3) of 2% (mass-volume) solution of urea with 300 cm^(3) of 3.43 % solution of sucrose of 20^(@)C ? (R = 0.082 "L atm K"^(-1) "mol"^(-1)) |
|
Answer» 5 ATM `pixx(600)/(1000)=[(6)/(60)+(3xx3.42)/(342)]0.082xx293` `pixx0.6=(0.1+0.03)xx0.082xx293` `pi=5.2`atm] |
|
| 15. |
Typical features of E2 involve : |
|
Answer» TWO step reaction |
|
| 16. |
When reaction is at standard state at equilibrium, then |
|
Answer» `80%` `CH_(3)OH(l)+(3)/(2)O_(2)(g)rarrCO_(2)+2H_(2)O(l)` `DeltaG_(r)[DeltaG_(f)(CO_(2),g)+2DeltaG_(f)(H_(2)O,l)]` `[-DeltaG_(f)(CH_(3)OH,l)-(3)/(2)DeltaG_(f)(O_(2),g)]` `=-394.4+2(-237.2)-(-166.2)-0` `=-394.4-474.4+166.2=-702.6KJmol^(-1)` Percentage effieiency `=(702.6)/(762)xx100=96.78%=97%` |
|
| 17. |
When a solution of alkaline earth metalin liquid ammonia is evaporated: |
|
Answer» They from hexammoniates, `M(NH_(3))_(6)` |
|
| 18. |
Which out of Na, K, Mg occurs as oxide in nature? |
| Answer» Solution :AI OCCURS as oxide in NATURE as BAUXITE `AI_(2)O_(3). 2H_(2)O` | |
| 19. |
The standard free energy of a reaction is found to be zero. What is its equilibrium constant ? |
| Answer» SOLUTION :`DeltaG^(@)= - 2.303 RT LOGK `. Hence, log `K = 0` or`K =1 `. | |
| 20. |
Which of the following pairs of ions is isoelectronic as well as isostructural ? |
|
Answer» `CO_(3)^(2-),NO_(3)^(-)` `CO_(3)^(-)` ionhas 42 electrons `(17+3xx8+1)` and Cl is `sp^(3)`-hybridized, while `SO_(3)^(2-)` also has 42 electrons and S is `sp^(3)`-hybridized. Thus, `ClO_(3)^(-)andSO_(3)^(2-)` are also isoelectronic and isostructural, i.e., option (d) is correct. |
|
| 21. |
Which of the following hydrocarbons has thelowest dipole menent ? |
|
Answer»
|
|
| 22. |
Which of the following reaction is expected to give readily a hydrocarbon product in good yields ? |
|
Answer» `RCOOK overset("Electrolysis")RARR` `overset("Electrolysis")rarr underset("Alkane")(R - R) + CO_(2) + H_(2) + 2KOH` |
|
| 23. |
The total vapoure pressure of a 4 mole% solutionof NH_(3) in water at 293 K is 50.0 torr. The vapour pressure of pure water is 17.0 torr at this temperature . Applying Henry's and Raoult's laws, calcilate the total vapour pressure for a 5 mole%solution: |
|
Answer» 58.25 torr |
|
| 24. |
What are organic reactions ? |
| Answer» Solution :Substrate is an ORGANIC MOLECULE REACTS with reagent , which may be an organic , in organic or any agnet like heat PHOTON etc , that brings about the chemical change to form a PRODUCT, this is known as organic reactions . | |
| 25. |
What is the acidity order of x,y & z ? |
|
Answer» ` X GT y gt Z` |
|
| 26. |
Which is the hybridisation of 1, 2, 3 carbon in overset(3)(C )H_(3)- overset(2)(C )-= overset(1)(C )H ? |
|
Answer» `SP^(3), sp^(2), sp` |
|
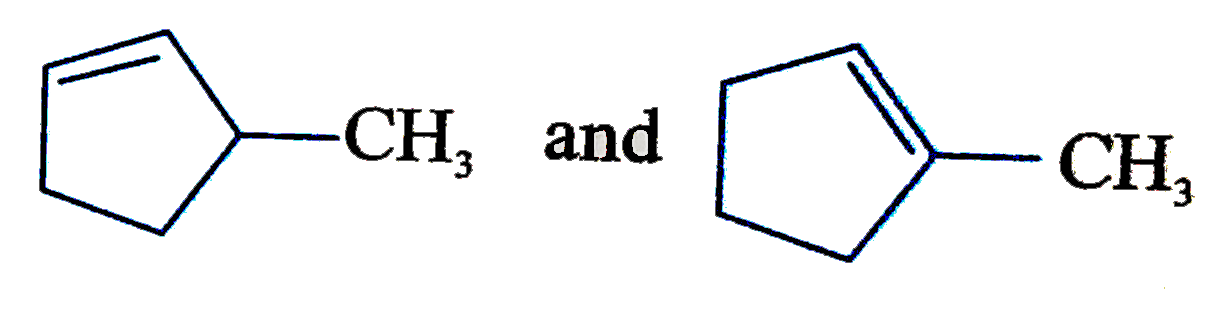
| 27. |
Two alkenes X(91%"yield") and Y(9% yield) are formed when the following compound is heated The structures of X and Y, respectively are |
|
Answer»
|
|
| 28. |
Three samples of water A, Band C have the D.O levels of 4 ppm, and 3.8 ppm and 2.1 ppm respectively the most polluted sample of water is |
| Answer» Answer :C | |
| 29. |
Which of the following is not the effect of acid rain |
|
Answer» The GLOSSY NATURE of Tajmahal is affected |
|
| 30. |
Which of the following statements is/are correct ? |
|
Answer» Spiro compounds contain fused rings at quarternary carbon |
|
| 31. |
The total no. of possible dichloro derivatives of isobutene are : |
|
Answer» 2 
|
|
| 32. |
Write resonance structures of CH_(3)COO^(-) and show the movement of electrons by curved arrows |
|
Answer» Solution :(i) First write the structure and put unshared pairs of valence electrons on appropriate atoms. (ii) Then draw the arrow ONE at a TIME MOVING the electrons to get the other structures. (III) `CH_(3)COO^(-)` is a `CH_(3) - overset(overset(O)(||))(C )- O^(-)` and its two resonance structure are as under 
|
|
| 33. |
Which of the following statement are correct for give reaction. CH_3-undersetunderset(CH_3)(|)C=CH-CH_3+HBr overset("Peroxide")toCH_3-undersetunderset(CH_3)(|)oversetoverset(Br)(|)C-CH_2-CH_3 " " CH_3-undersetunderset(CH_3)(|)CH-oversetoverset(Br)(|)CH-CH_3 |
|
Answer» MAJOR PRODUCT is mixture of two enantiomers. |
|
| 34. |
Which buffer solution has maximum pH? |
|
Answer» mixture which is 0.1 M in `CH_3 COOH and 0.1 Mis CH_3 COONa [pK_a (CH_3COOH )= 4.74` ( B ) ` pH =pKa +log ""(0.2)/(0.2)=4.74` ( C ) ` pH =pKa +log ""(0.1)/(0.1)=9.26` |
|
| 35. |
Which of the following is formed when lithium is heated in air ? |
|
Answer» Only `Li_(2)O` |
|
| 36. |
The uncertaintyin positionis 10^(-10)m andin velocityis 5.27 xx 10^(-24) ms^(-1)calculatemass of particle . |
| Answer» SOLUTION :`1XX 10^(10) M` | |
| 37. |
The relation between K_(p) and K_(c) is K_(p)=K_(c)(RT)^(Deltan) unit of K_(p)=(atm)^(Deltan), unit of K_(c)=(mol L^(-1))^(Deltan) Consider the two reaction: XeF_(6)(g)+H_(2)O(g) hArr XeO_(3)F_(4)(g)+2HF(g), K_(1) XeO_(4)(g)+XeF_(6)(g) hArr XeOF_(4)(g)+XeO_(3)F_(2)(g), K_(2) Then the equilibrium constant for the following reaction XeO_(4)(g)+2HF(g) hArr XeO_(3)F_(2)(g) is given by: |
|
Answer» `K_(1)//K_(2)^(2)` |
|
| 38. |
The total number of fundamental particles in normal hydrogen molecule |
|
Answer» 1 |
|
| 39. |
Write the coordination number of each ion in the following crystals .(i) NaCl (ii) CsCl (iii) ZnS (iv)CaF_(2) ( v)Na_(2)O |
| Answer» SOLUTION :`Na^(+) = 6, CL^(-) =6 , (ii) CS^(+) = 8, Cl^(-) = 8 (iii) Zn^(2+) = 4, S^(2-) = 4(iv) Ca^(2+) = 8, F^(-) = 4 , (V) Na^(+) = 4, O^(2-) = 8 ` | |
| 40. |
Which elements are exception in classification of periodic table. |
|
Answer» Solution :Two elements like hydrogen and helium are observed as an exception . Place of hydrogen in periodic table : Hydrogen contain le in ls orbital so it must be situated in group-1. Hydrogen ACCEPT `1 e^(-)`and from stable structure like helium. So, it must be situated in group-17 But there is same DIFFERENCE observed between hydrogen and ist group and hydrogen and `17^(th)`group so hydrogen atom is situated in separately in periodic table. Place of helium in periodic table . Helium element having `1 ^(ST)`electron configuration so it must be present in s-block. But its positioning in the p-block along with other group 18 elements is Justified. Because it has a completely filled valence shell `(1 s^(2))` and as a RESULT, exhibits properties CHARACTERISTIC of other noble gases. |
|
| 41. |
What is oxidation number of N inunderlineNH_(4)underlineNO_(3) respectively ? |
Answer» SOLUTION :`NH_(4)NO_(3)toNH_(4)^(+)+NO_(3)^(-)` 
|
|
| 42. |
Thermodynamically the most stable form of carbon is …… |
|
Answer» DIAMOND |
|
| 43. |
What is ionic equilibrium ? Discuss the relation of types of substance and ionic equilibrium in solution. |
|
Answer» Solution :The ions of aqueous solution in equilibrium its known as ionic equilibrium. e.g. `Fe_((aq))^(3+) + SCN_((aq))^(-) hArr [Fe(SCN)]_((aq))^(2+)` The aqueous solution of sugar does not conduct electricity and sugar is .non-electrolytic.. The ionic equilibrium is not occurs in non electrolytic substance. However, when common salt is added to water it conducts electricity. The substances conduct electricity in their aqueous solutions are called electrolytes STRONG electrolytes on dissolution in water are ionized almost completely. The salt which is almost 100% ionization in water is called strong ELECTROLYTE. e.g. NaCl is strong electrolyte. This type of solution ionic concentration and conductivity is more. The substance less ionised (less than 5%) in solution is known as weak electrolyte. e.g., Acetic ACID. "In weak electrolytes, equilibrium is established between ions and the unionized molecules." e.g. `CH_3COOH_((aq))+aq hArr CH_3COO_((aq))^(-) + H_((aq))^(+)` Ionic equilibrium is established in Acid, Base and SALTS. |
|
| 44. |
What happens when calcium nitrate is heated ? |
|
Answer» Solution : Calcium nitrate, on HEATING, decomposes to GIVE calcium OXIDE. `2CA(NO_(3))_(2(s)) overset(Delta)to 2CaO_((s)) +4NO_(2(g))+O_(2(g))` |
|
| 45. |
Which of the following does not represent the resonating structure of biphenyl |
|
Answer»
|
|
| 46. |
Which of the following is nature ore name of calcium |
|
Answer» HYDROLITH Hydrolith `CaH_(2)` Colamanite `2Cao.3Ba_(2).5H_(2)O` Phosphorite `Ca3(PO_(4))_(2)` |
|
| 47. |
Which among the following is the heaviest? |
|
Answer» One MOLE of oxygen |
|
| 48. |
What is mean by stock solution (or) standard solution ? what is meant by working standard? |
|
Answer» Solution :(i) A standard solution or a STOCK solution whose concentration is accurately KNOWN. (II) At the time of experiment, the solution with required concentration is prepared by DILUTING the stock solution. This diluted solution is called working standard. |
|
| 49. |
Which of the following is not ferromagnetic ? |
|
Answer» Cobalt |
|
| 50. |
Write the IUPAC names of the following compounds. (i) CH_(3)-CH_(2)-CH_(2)-NH-CH_(3) (ii) CH_(3)-underset(CH_(3))underset(|)(CH)-NH-CH_(3) (iii) CH_(3)-CH_(2)-CH_(2)-underset(CH_(3))underset(|)N-CH_(3) |
|
Answer» SOLUTION :(i) N-methylpropan-l-amine (II) N-methylpropan-2-amine (III) N,N-dimethylpropan-1-amine |
|