Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Two reactions are given below: (i) H_(2)O(g)+(1)/(2)O_(2)(g) rarrH_(2)O(l) (ii) H_(2)(g)+(1)/(2)O_(2)(g) rarr H_(2)O(g) Which one will have greater value of DeltaH and why ? |
| Answer» Solution :Reaction(i) will have greater value of `DELTAH`because when `H_(2)O(G)` CONDENSES to form`H_(2)O(l)` , some heatis EVOLVED( called heatof CONDENSATION). | |
| 2. |
What is basic difference between the terms electron gain enthalpy and electronegativity? |
| Answer» Solution :ELECTRON gain enthalpy refers to TENDENCY of an isolated gaseous atom to accept an additional electron to FORM a negative ion. Whereas ELECTRONEGATIVITY refers to tendency of the atom of an element to attract shared pair of electrons towards it in a covalent bond. | |
| 3. |
What is diagonal relation? Give one pair of elements, that have this relation. |
|
Answer» |
|
| 4. |
The volume of CO_(2)that can be obtaind at STP from 60 g. of 70% pure MgCO_(3) is |
|
Answer» 16L `MgCO_(3)rarrMgO=CO_(2)` `84g ______ UNDERSET("22.4 L")underset(DARR)(44g)` `42g______11.2L` |
|
| 5. |
Which of the following is not common component of photochemical smog? |
|
Answer» Ozone |
|
| 7. |
Write the detail about hydrogenation of unsaturates hydrocarbon. |
|
Answer» Solution :Defination of HYDROGENATION : Dihydrogen gas adds to alkenes and alkynes in the presene of finely divided catalysts like platinum, palladium or nickel to form alkanes. This process is called hydrogenation. * Work of catalyst : These metals absorb dihydrogen gas on their surfaces and activate the hydrogen-hydrogen bond. Platinum and palladium CATALYSE the reaction at room temperature but RELATIVELY higher temperatureand pressure are REQUIRED with nickel catalysts. * Some examples of hydrogenation reaction : When hydrogen is added to unsaturated hydrocarbons like alkene and alkyne and alkane is formed, such type of reaction is known as alkane. (i) `underset("Ethene")(CH_(2))=CH_(2)+H_(2)underset(or Ni, Delta)overset(Pt//Pd)rarrunderset("Ethane")(CH_(3))-CH_(3)` (ii) `underset("Propane")(CH_(3))-CH=CH_(2)+H_(2)underset(or Ni, Delta)overset(Pt//Pd)rarr underset("Propane")(CH_(3))-CH_(2)-CH_(3)` (iii) `underset("PROPYNE")(CH_(3))-C-=C-H+2H_(2)underset(or Ni, Delta)overset(Pt//Pd)rarrunderset("Propane")(CH_(3))-CH_(2)-CH_(3)` Note : Rany nickel is used as catalyst. It is a working nickel catalyst and used in the form of powder. |
|
| 8. |
Which of the following is a characteristic feature of a free radical? |
|
Answer» It has a POSITIVE charge |
|
| 9. |
Write history and devlopement of organic chemistry |
|
Answer» Solution :Science of organic chemistry is about two hundred years old. 1828: In 1828F, Wohler synthesised an organic compound, UREA from an inorganic compound, ammonium cyanate, `UNDERSET("Ammonium cyanate")(NH_(4)CNO) overset("Heat")RARR underset("Urea")(NH_(2)CONH_(2))` The pioneering synthesis of acetic acid by Kolbe (1845) and that of methane by BERTHELOT (1856) showed conclusively that organic compounds could be synthesised from inorganic sources in a laboratory. |
|
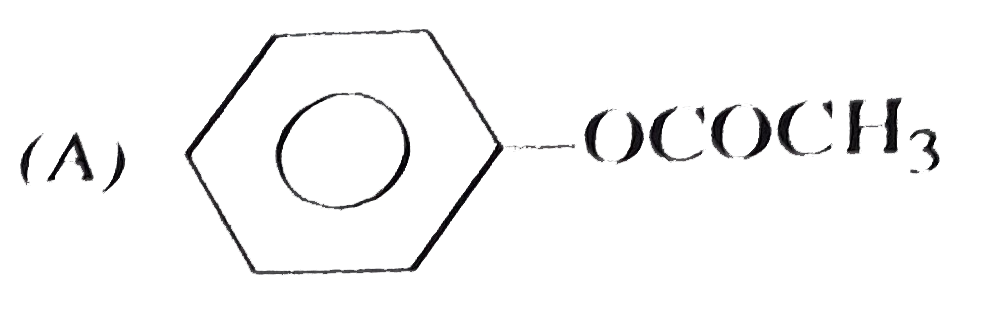
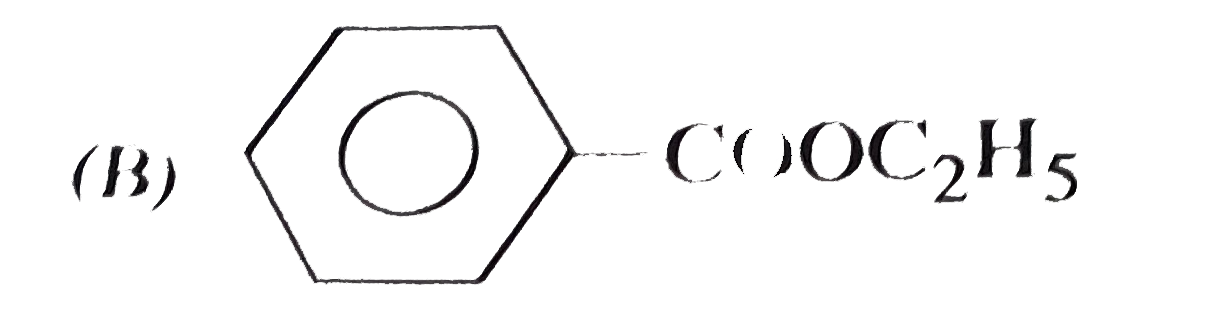
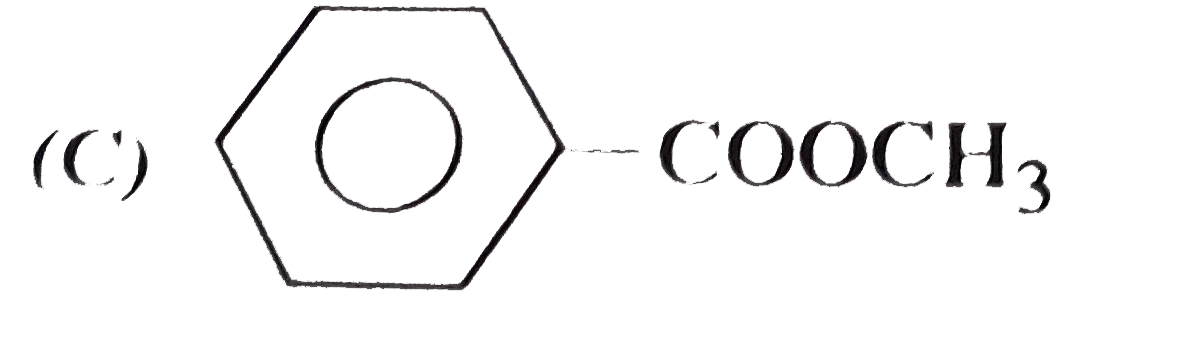
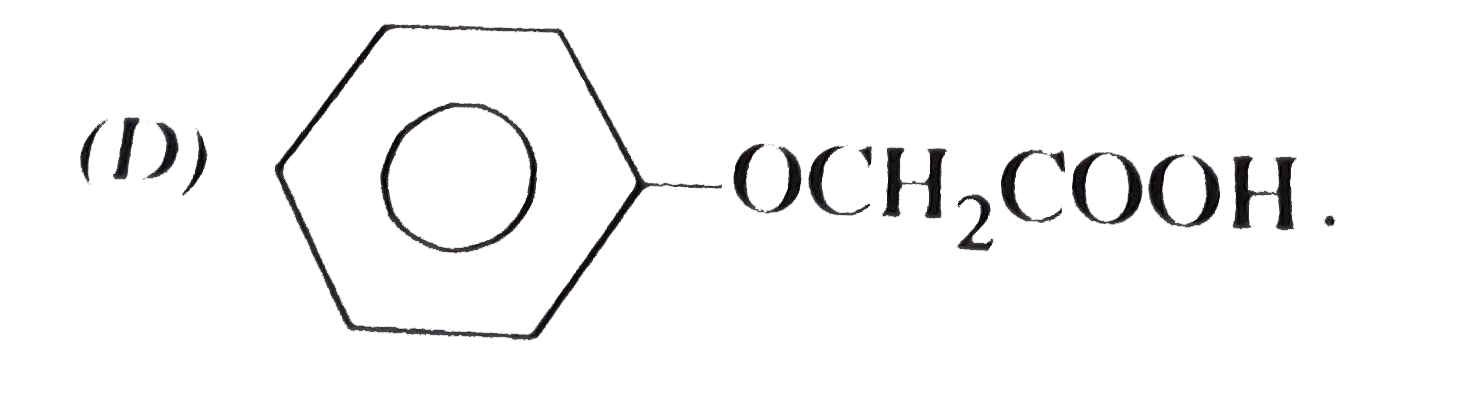
| 11. |
Which of the following is phenyl ethanoate ? |
|
Answer»
|
|
| 12. |
The screening effect of inner electrons of an atom causes |
|
Answer» DECREASE in IONISATION energy |
|
| 13. |
The reagent (s) that can selectively precipitate S^(2-) from a mixture of S^(2-) and SO_(4)^(2-) in aqueous solution is (are) |
|
Answer» `CuCl_(2)` `Cu^(2+) + S^(2-) rarr underset(("Black ppt."))(CuS)` `CuSO_(4)` formed remains soluble in AQUEOUS solution |
|
| 14. |
What is the main limiatation of the first law of thermodynamics ? |
| Answer» SOLUTION :It cannot PREDICT the SPONTANEITY of a PROCESS. | |
| 15. |
Which of the following statements is incorrect for PCI_5 ? |
|
Answer» Its all P-Cl bond lengths are equal |
|
| 16. |
Which of the following is correct order of the stability of carbocations? |
|
Answer» `""^(+)CH_(3) GT""^(+)CH_(2)CH_(3) gt ""^(+)CH(CH_(3))_(2) gt ""^(+)C(CH_(3))_(3)` |
|
| 17. |
Which of the following statements is incorrect in relation to ionization enthalpy? |
|
Answer» lonization ENTHALPY increases for each successive ELECTRON |
|
| 18. |
The state of hybridization of C_(2), C_(3), C_(5) and C_(6) of the hydrocarbon is in the following sequence…… underset(7)(C )H_(3)- underset(underset(CH_(3))(6|))overset(overset(CH_(3))(|))(C )-underset(5)(C H)= underset(4)overset(overset(CH_(3))(|))(C )- underset(3)(C )H_(2)- underset(2)(C )-= underset(1)(C )H |
|
Answer» `SP, sp^(3), sp^(2) and sp^(3)` |
|
| 19. |
Which forces of attraction are responsible for liquification of hydrogen chloride |
|
Answer» COULOMBIC FORCES |
|
| 20. |
Which one of the following is used as water softener? |
|
Answer» ZEOLITES |
|
| 21. |
What are intersitials in a crystal ? |
| Answer» Solution :Atoms or ions that FILL the NORMAL vacent intersitial VOIDS in a crystal are called interstitials. | |
| 22. |
Which of the following is a constituent of baking powder ? |
| Answer» Solution :Cream of tartar | |
| 23. |
Which of the following are true ? |
|
Answer» In NaCl crystals , `Na^+` ions are present in all the OCTAHEDRAL voids |
|
| 24. |
The solubility product of Al (OH)_3 is 2.7 xx 10^(-11). Calculate the solubility in g L^(-1)and also find pH of this solution. (Atomic mass of Al = 27 u). |
|
Answer» SOLUTION :LET S be the solubility of `AL(OH)_3` `{:(Al(OH)_3 hArr, Al_((aq))^(3+) + , 3OH_((aq))^(-)),(1,0,0),(1-s,s,3s):}` Concentration to species at t=0 Concentration of various species at EQUILIBRIUM `K_(sp)=[Al^(3+)] [OH^-]^3 =(S) (3s)^3 = 27S^4` `S^4=K_(sp)/27=(2.7+10^(-11))/27=1xx10^(-12)` `S=1xx10^(-3) "mol L"^(-1)` (i) Solubility of `Al(OH)_3` Molar mass of `Al(OH)_3` is 78 g. Therefore , Solubilityof `Al(OH)_3` in g `L^(-1)` `=1xx10^(-3) xx 78 g L^(-1) 78xx10^(-3) g L^(-1)` `=7.8xx10^(-2) g L^(-1)` (ii) pH of the solution `S=1xx10^(-3) "mol L"^(-1)` `[OH^-]=3S=3xx1xx10^(-3)=3xx10^(-3)` pOH=3-log 3 pH=14-pOH =11+log 3 = 11.4771 |
|
| 26. |
What is Visocity ? |
| Answer» SOLUTION : ..Viscosity is a MEASURE of resistance to flow which arises due to the INTERNAL FRICTION between layers of fluid or they slip PART one another while liquid flows... | |
| 27. |
When 20 mL of (M)/(10) NaOH are added to 10 mL of (M)/(10)HCl, the resulting solution will : |
|
Answer» TURN blue litmus RED |
|
| 28. |
Water always flows from higher level to lower level on its own. Which of the following statement is correct for this phenomena ? |
|
Answer» FREE ENERGY increases |
|
| 29. |
.What are A,B,C and D in these conversions ? |
|
Answer» Solution :`BeO+C+Cl_(2) to BeCl_(2) +CO` `BeCl_(2) +LiAlH_(4) to LiCl+AlCl_(3)+2BeH_(2)` `BeCl_(2)+2H_(2)O to Be(OH)_(2)+2HCL` `Be(OH)_(2) +2OH^(-) to [Be(OH)_(4)]^(2-)` `A-BeCl_(2) , B - BeH_(2) , C-Be(OH)_(2) and D[Be(OH)_(4)]^(2-)` |
|
| 30. |
Which of the following represents the composition of carnallite mineral ? |
|
Answer» `K_(2)O.Al_(2)O_(3).6SiO_(2)` |
|
| 31. |
Which one of the following is not electrophile? |
|
Answer» `NH_(3)` |
|
| 32. |
Write the value forCH_3COOH and NaCl . |
|
Answer» SOLUTION :` i_(CH_3 COOH)- 0.5 (CH_3 COOH "in " C_6H_6 ) ` ` i_(NACL) -2 (NaCl "in " H_2O) ` |
|
| 33. |
The set representing the correct order of ionic radius is |
|
Answer» `LI ^(+) gt Be ^(2+) gt Na ^(+) gt Mg ^(2+)` |
|
| 34. |
Which of the following metals rendered passive with Conc.HNO_(3) |
|
Answer» `Be` |
|
| 35. |
Which hydride is called alante? |
|
Answer» `CaH_(2)` |
|
| 36. |
Which of the following is/are +vely charged colloid |
|
Answer» `Al(OH)_(3)` (d) Gold is -vely charged colloid due to capture of electrons in Bredig's Arc method. |
|
| 37. |
Which of the following is electron deficient ? |
|
Answer» `PH_(3)` `CH_(3) - CH_(3) -` COVALENT neutral MOLECULE, `BH_(3)` - electron deficient |
|
| 38. |
Which of the following compounds possesses the C-H bond with the lowest bond dissociation energy ? |
|
Answer» TOLUENE `C_(6)H_(5)OVERSET(*)(C)H_(2) gt (CH_(3))_(3)overset(*)(C) gt CH_(3)overset(*)(C)HCH_(2)CH_(2)CH_(3) gt overset(*)(C_(6))H_(5)`, therefore, the C-H bond of toluene has the LOWEST bond dissociation energy. |
|
| 39. |
Writenotes on isomerisaton |
|
Answer» Solution :Isomerisatin is achemicalprocessby whicha compoundis transformedintoany toisomeric form (ii)Normalalkanescan beconverted intobranched alkanesin THEPRESENT of `AiCI_(2)` ANDAT 298 K (ii) Thisprocess is fogreatindustrialimportanehte quantityof gasolineisimprovesisomerisingtis components |
|
| 40. |
Which important property did Mendeleev use to classify the elements in his periodic table and did he stick to that? |
| Answer» SOLUTION :He classified elements according to atomic weight. He could`N^(+)` stick for this and he accept that physical and CHEMICAL PROPERTIES are basic for that. | |
| 41. |
Write structure of different isomers corresponding to the 5^(th) member of alkyne serie. Also awrite IUPAC names of all the isomers. What type of isomerism is exhibited by different pairs of isomers ? |
|
Answer» Solution :`5^(th)` member of alkyne has the molecular formula `C_(6)H_(10)`. It has seven possible alkaline isomers. The possible isomers are : Position isomers : (i) `underset("Hex-1-yne")(overset(6)(C)H_(3)-overset(5)(C)CH_(2)-overset(4)(C)H_(2)-overset(3)(C)H_(2)-overset(2)(C)-=overset(1)(C)H)` (ii) `underset("Hex-2-yne")(overset(6)(C)H_(3)-overset(5)(C)H_(2)-overset(4)(C)H_(2)-overset(3)(C)-=overset(2)(C)-overset(1)(C)H_(3))` (III) `underset("Hex-3-yne")(overset(1)(C)H_(3)-overset(2)(C)H_(2)-overset(3)(C)-=overset(4)(C)-overset(5)(C)H-overset(6)(C)H_(3))` Chain isomers: (iv) `underset("3-methylpent-1-yne")(H-overset(1)(C)-=overset(2)(C)-overset(3)(C)H_(2)-underset(CH_(3))underset(|)overset(4)(CH)-overset(5)(C)H_(3))` (V) `underset("4-methylpent-1-yne")(Hoverset(1)(C)-=overset(2)(C)-underset(CH_(3))underset(|)overset(3)(CH)-overset(4)(C)H_(2)-overset(5)(C)H_(3))` (vi) `underset("4-methylpent-2-yne")(H_(3)overset(1)-overset(2)(C)-=overset(3)(C)-underset(CH_(3))underset(|)overset(4)(CH)-overset(5)(C)H_(3))` (vii) `underset("3,3-methylbut-1-yne")(H-overset(1)(C)-=overset(2)(C)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-overset(4)(C)H_(3))` Different isomers pairs have position and chain isomers. |
|
| 42. |
What are the properties of electromagnetic radiations? |
|
Answer» Solution :(a) The radiant energy is emitted from any source continuously in the form of radiations (or) waves. Radiation is the EMISSION and transmission of energy through space in the form of waves. (b) The electromagnetic waves consist of oscillating ELECTRIC and magnetic fields. These two filed components have the same wavelengths and frequency and travel in planes PERPENDICULAR to each other and also perpendicular to the direction of propagation of electromagnetic wave. (c) The electromagnetic waves do not require any material medium for their propagation. They can travel in vacuum also. (d) All electromagnetic radiations travel with the same velocity, namely, with the velocity of light `(3XX 10^(8) ms^(-1))` (E) The electromagnetic radiations consist of difterent radiations which differ in their wavelength (or frequencies) and constitue "electromagnetic spectrum". Visible light is a small portion of electromagnetic spectrum. |
|
| 43. |
The solid state of matter is converted inot gas by |
|
Answer» SUBLIMATION |
|
| 44. |
What electronic transition in Li^(2+) produces the radiation of the same wave length as the first line in the Lyman series of hydrogen? |
|
Answer» N = 4 to n = 2 `""Li^(2+) ""H` `(3)^2 (1/(n_1^2) - 1/(n_2^2)) = (1)^2 (1/((1)^2) - 1/((2)^2))` on solving`n_1 =2 , n_2 = 4` |
|
| 45. |
Which of the following salt is not obtained from lithium and magnesium ? |
|
Answer» Oxide |
|
| 46. |
Which of the following species acts as Bronsted base but not as acid ? |
|
Answer» `CH_3COOH` |
|
| 47. |
When the first element of the periodic table is treated with dioxygen , it gives a compound whose solid state floats on its liquid state. This compound has an ability to act as an acid a well as a base. What product will be formed when this compound undergoes autoionisation ? |
|
Answer» Solution :The first element of the periodic table is H and its molecular FORM is dihydrogen `(H_(2))` . When dihydrogen reacts with dioxygen , water is formed. Water is a liquid at room temperature . When liquid water freezes,it expnads to form ice . In other words, density of ice is lower than that of liquid wter and hence ice floats over water. Water is amphoteric in nature, i.e., it acts as an ACID in presence of strong bases and a base in presence of strong acids. `UNDERSET("Acid"_(1))(H_(2)O(l))+ underset("Base"_(2))(NH_(3)(aq)) to underset("Acids"_(2))(NH_(4)^(+)(aq))+ underset("Base"_(1))(OH^(-)(aq))` `underset("Base"_(1))(H_(2)O(l))+ underset("Acid"_(2))(H_(2)S(aq)) to underset("Acids"_(1))(H_(2)O^(+)(aq))+ underset("Base"_(2))(HS^(-)(aq))` Due to amphoteric CHARACTER, water undergoes self ionization as shown below : `underset("Acid"_(1))(H_(2)O(l))+ underset("Base"_(2))(H_(2)O("base")) to underset("(CONJUGATE acid)")underset("Acids"_(2))(H_(3)O^(+)(aq))+ underset("(Comjugate base)")underset("Base"_(2))(OH^(-)(aq))` This self ionization of water is called auto-protolysis. |
|
| 48. |
Which of the following pairs of species have the same bond order- |
|
Answer» `O_(2),NO^(+)` Total no. of electrons present in `CO=14` MO electronic configuration of `CO-` `(sigma_(1s))^(2)(sigma_(1s)^(**))^(2)(sigma_(2s))^(2)(sigma_(2p_(z)))^(2)(sigma_(2s)^(**))^(2)(pi_(2p_(X)))^(2)(pi_(2p_(y)))^(2)` MO electronic configuration of `CN^(-)`- `(sigma_(1s))^(2)(sigma_(1s)^(**))^(2)(sigma_(2s))^(2)(sigma_(2s)^(**))^(2)(pi_(2p_(x)))^(2)(pi_(2p_(y)))^(2)(sigma_(2p_(z)))^(2)` ltbgt bond order of `CO=(1)/(2)(8-2)=3` Bond order of `CN^(-)=(1)/(2)(8-2)=3` |
|
| 49. |
Which compound is 2,2,3-trimethyl hexane? |
|
Answer» `CH_(3)- underset(CH_(3))underset(|)overset(CH_(3))overset(|)C-overset(CH_(3)) overset(|) CH-CH_(2)-CH_(3)` |
|
| 50. |
What weight of zinc would be required to produce enough hydrogen to reduce completely 7.95 g of CuO. |
|
Answer» Solution :The corresponding equations are : `underset(65.38 g)(Zn) + H_(2)SO_(4) to underset(2.016 g)(ZnSO_(4)) + H_(2)` and `underset(79.55 g)(CuO) + H_(2) to underset(2.016 g)(Cu) + H_(2)O` `therefore 79.55 g` of CuO REQUIRE for REDUCTION, hydrogen = 2.016 g. `therefore 7.95 g` of CuO will require for reduction, hydrogen `=(2.016)/(79.55) xx 7.95 = 0.201 g` Moreover, `therefore 2.016 g` of `H_(2)` is produced by ZINC = 65.38 g `therefore 0.201` g of `H_(2)` will be produced by zinc `=(65.38)/(2.016) xx 0.201 = 6.52 g` Hence, the amount of zinc required to REDUCE 5 g of CuO is 6.52 g |
|