Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write the structures and names of products obtained in the reaction of sodium with a mixture of 1-iodo-2-methylpropane and 2-iodopropane. |
Answer» Solution :WURTZ REACTION between 1-iodo-2-methylpropane and 2-iodopropane gives the following THREE PRODUCTS : 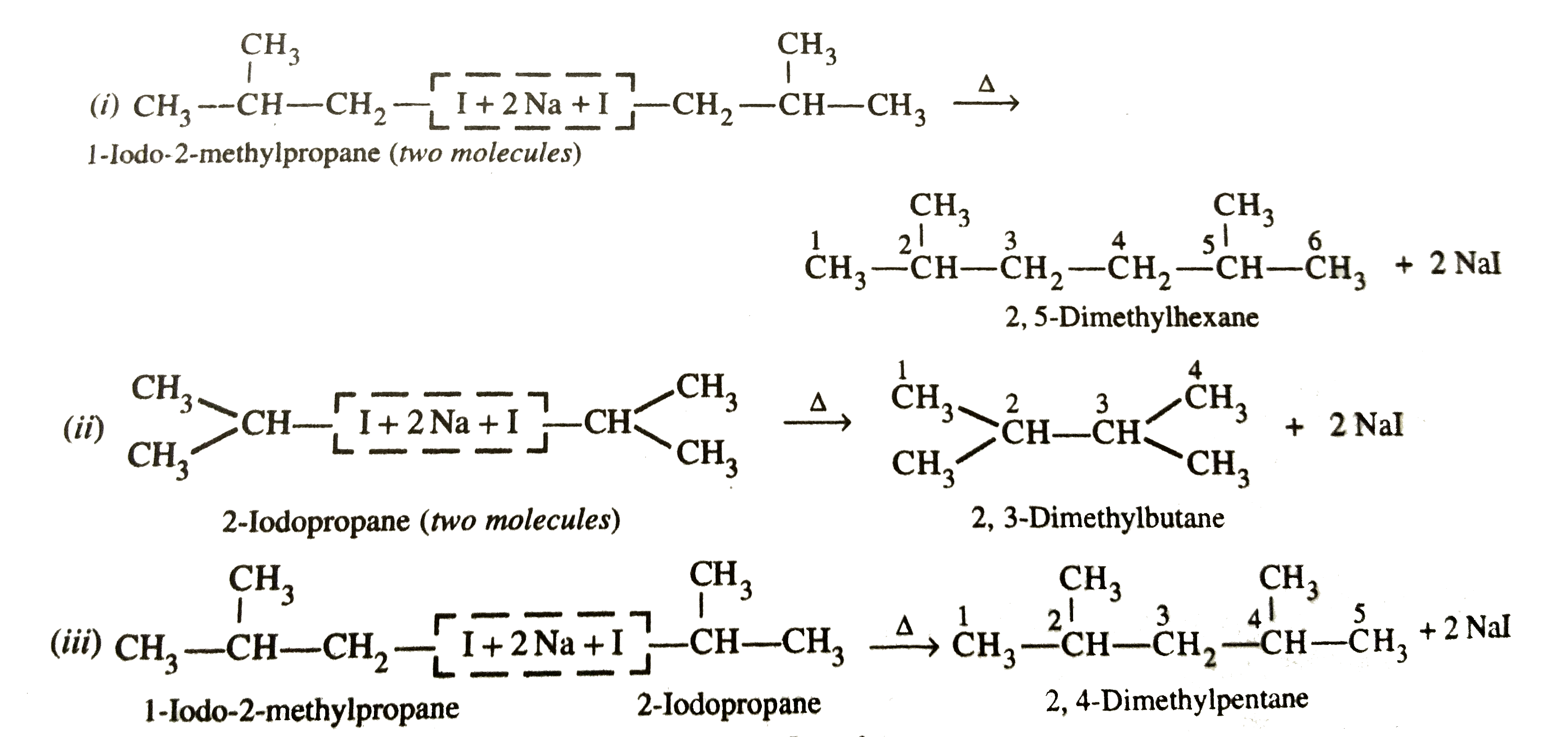
|
|
| 2. |
Two structures of anilinium are given. Choose the correct statement |
|
Answer» II is not an ACCEPTABLE canonical structure because it is non-aromatic |
|
| 3. |
The Rydberg's constant is 1.097373177 xx 10^(7)m^(-1). It can be expressed to three significant figures as |
|
Answer» `1.0974 XX 10^(7)m^(-1)` |
|
| 4. |
Why an aqueous borax solution is alkaline to litmus? |
|
Answer» Solution :`B_4 O_(7)^(2-)` is CONJUGATE base of weak ACID tetraboric acid. Due to anion hydrolysis, the AQUEOUS solution of borax is basic. ` B_(4)O_(7)^(2-) +7H_2 O hArr2OH^(-) +4H_3 BO_(3)`. |
|
| 5. |
The solubility of KCIO_(3) at several temperature is shown in the accoupanying diagram. A student mixed 10.0g of KCIO_(3) with 45.0g of H_(2)O and stires it for a long time at 60^(@)C until the solution is completely clear then allows it to cool slowly to 20^(@)C where it remains clear. which statement about the final clear mixture at 20^(@)C is correct ? |
|
Answer» It is a SATURATED SOLUTION |
|
| 7. |
The temperature of 5 moles of a gas is decreased by 2K at constant pressure of 1 atm. Indicate the correct statement |
|
Answer» <P>Work done by GAS is = 5R `pv_(1) = 5RT` For 5 moles of gas at temperature T-2 `PV_(2) = 5R (T-2)` `:. P(V_(2) - V_(1)) = 5R (T-2-T), P DELTA V = -10R, - P Delta V = 10R` When `DeltaV= 10R` When `Delta V` is negative, W is +ve |
|
| 8. |
Which of the following represent the given mode of hybridisation sp2, sp2, sp, sp from left to right ? |
|
Answer» `H_(2)C=CH-C -= N` |
|
| 10. |
Which of the following sets of quantum numbers are not permitted? (i) n = 2, l = 2, m = -1, s = +1//2 (ii) n = 2, l = 1, m = -1, 2 = -1//2 (iii) n=2, l = 0, m = 0, s = 0 (iv) n = 2, l = 1, m = 2, s = +1//2 |
|
Answer» Solution :(i) This set of QUANTUM numbers is not permitted since the value of L cannot be equal to N. (ii) This set of quantum number is permitted. (iii) This set of quantum number is not permitted because the value of SPIN quantum number cannot by zero. (iv) This set of quantum numbers is also not permitted since the vlaue of 'm' cannot be GREATER than. 1. |
|
| 11. |
Which one of the following curves represents the graph pH during the titration of NaOH and HCl(aq) |
|
Answer»
|
|
| 12. |
What will be the mole fraction of solution if solution cantain 0.1 mole of each A, B & C ? |
|
Answer» `1.0` |
|
| 13. |
Three gases A,B and C have values of van der Waal's constnat, a (in units of" litre"^(3) "mol"^(-2) atm) of 1.38,6.70 and 4.00 respectively. The ease of liquefaction of gases decreases in the order: |
|
Answer» `BgtAgtC` |
|
| 14. |
What are ternary hydrides? Give example? |
| Answer» Solution :Ternary HYDRIDES are COMPOUNDS in which the molecule is CONSTITUTED by HYDROGEN and two types of elements, EG.. LIBH, or LiAlH | |
| 15. |
What is the difference between diffusion and effusion ? Which law is applicable to the process of effusion ? |
| Answer» SOLUTION :Graham.s LAW. | |
| 16. |
Which effect provides the experimental justification for magnetic quantum number? |
| Answer» Answer :D | |
| 17. |
Which of the following is the most acidic ? |
|
Answer» HCOOH |
|
| 18. |
What is similar between (i) Na and Na^(+) (ii) F and F^(-) |
|
Answer» Solution :(i) Atomic number of Na and `Na^(+)` having same (Z = 11). (II) Atomic NUMBERS of F and `F^(-)` having same (Z = 9). |
|
| 19. |
Which of the following statements about hydrogen are correct ? |
|
Answer» Hydrogen has three isotopes of which protium is the most common. |
|
| 20. |
Which of the following processes can be used for the purification of colloids? |
|
Answer» Coagulation |
|
| 21. |
What is the pH of 0.005 M Ba(OH)_2 ? |
|
Answer» SOLUTION :`[OH^-]`=(0.005) x 2 = 0.01 = `1xx10^(-2)` THEREFORE pOH=2 and pH=12 |
|
| 22. |
What do you understand by the terms" |
|
Answer» Solution :(i) Hydrogen econom:Consult SECTION 9:5 (ii)Hydrogenation:Consult section 9:4 (III) Syn gas: Consult section 9:3 (iv) Water gas shift reaction: Consult section 9:3 (V) Fuel cell: Consult section 9:5 |
|
| 23. |
What is d-block elements and transition elements ? Explain characteristic of d-block elements. |
|
Answer» Solution :"Elements of group 3 to 12 are known as d-block elements." General electronic configuration of these elements are `(n -1) d^(1-10) ns^(0-2)`. These elements are known as d-block elements because their last electron is filled in d-orbital. Characteristic of d-block elements : They are all metals. They mostly form coloured ions, exhibit variable valence (oxidation states), paramagnetism and oftenly used as catalysts. However, Zn, CD and Hg which have the electronic configuration, (n - 1) `d^(10) ns^(2)`do not show most of the properties of TRANSITION elements. Transition Elements : In a way, transition metals form a bridge between the chemically active metals of s-block elements and the less active elements of Groups 13 and 14 and thus take their familiar NAME "Transition Elements". |
|
| 24. |
Which of the following reaction causes depletion of ozone layer in summer season. |
|
Answer» `ClONO_(2(g))+H_2 OrarrHCOl_((g))+NHO_(3(g))` |
|
| 25. |
What is the dominant intermolecular force or bond that must be overcome in converting liquid methanol to a gas? |
|
Answer» LONDON DISPERSION force |
|
| 26. |
Which statement is correct? |
|
Answer» Both are AROMATIC compounds. |
|
| 27. |
The region which is extended upto 500 km above sea level is known as ..... |
|
Answer» ATMOSPHERE |
|
| 28. |
Which of the following free radicals is the least stable ? |
|
Answer» `*CH_(3)` |
|
| 29. |
Which of the following is most volatile |
|
Answer» Diamond |
|
| 30. |
When water is added to compound (A) of calcium , solution of compound (B) is formed . When carbon dioxide is passed into the solution , it turns milky due to the formation of compound (C) . If excess of carbon dioxide is passed into the solution , milkiness disappears due to the formation of compound D. Identify the compounds A , B , C and D . Explain why the milkiness disappears in the last step . |
|
Answer» Solution :When `CO_(2)` is passed through solution of COMPOUND (B) , it TURNS milky , therefore , solution of compound (B) must be lime water , i.e., `Ca(OH)_(2)` solution and milkiness must be due to the formation of insoluble `CaCO_(3)` (C) . Further since , compound (B) is obtained by dissolving compound (A) in water , therefore , compound (A) must be quick lime , CAO `underset((A)) underset("Calcium OXIDE")(CaO) + H_(2)O underset("Lime water (B)")(Ca(OH)_(2)) , CO_(2) to underset("Milkiness)")underset("carbonate (C)")underset("Calcium")(CaCO_(3)) + H_(2)O` When EXCESS of `CO_(2)` is passed milkiness disappears due to formation of soluble calcium bicarbonate (D) `underset("Milkiness (C)")(CaCO_(3)) + CO_(2) + H_(2)O to underset("(Soluble in" H_(2)O")") underset("Calcium bicarbonate")(Ca(HCO_(3))_(2))` Thus , compound (A) = CaO , Compound (B) = `Ca(OH)_(2)` , Compound (C) = `CaCO_(3)` and compound (D) = `Ca(HCO_(3))_(2)`. |
|
| 31. |
Write the possible isomers for the formula C_(5)H_(10)O with their name indicating position isomerism. |
| Answer» SOLUTION :(i) `UNDERSET("pent-2-one")(CH_(3)-underset(O)underset(||)C-CH_(2)-CH_(2)-CH_(3))` (II) `underset("pent-3-one")(CH_(3)-CH_(2)-underset(O)underset(||)C-CH_(2)-CH_(3)` | |
| 32. |
Which of the following acidic radicals produce coloured volatile product with conc. H_(2)SO_(4)? NO_(2)^(-),NO_(3)^(-),Br^(-),I^(-),CI^(-),S_(2)O_(3)^(2-),B_(3)^(3-),CrO_(4)^(2-) |
|
Answer» <BR> SOLUTION :`NO_(2)^(-),NO_(3)^(-),Br^(-),I^(-)` |
|
| 33. |
Which of the following reagents can be used to distinguished phenol and benzoic acid ? |
|
Answer» TOLLEN's reagent |
|
| 34. |
What are the general conditions for liquification of a gas? |
|
Answer» Solution :Conditions for LIQUIFICATION of a gas are: 1) LOW TEMPERATURE and 2) HIGH pressure. |
|
| 35. |
Which is the basic building unit of all silicates ? |
| Answer» SOLUTION :`SiO_(4)^(4-)`is the basicbuilding UNIT of all SILICATES . | |
| 36. |
Which of the following pairs will form the most stable ionic bond? |
|
Answer» NA and F |
|
| 37. |
Which of the following statement are not correct ? |
|
Answer» The standard value of absolute entropy of elementarysubstances is taken as zero. |
|
| 38. |
Which is the most stable carbonium ion? |
|
Answer» `CH_(3)CH_(2)^(+)` |
|
| 39. |
Which of the following transitions will have minimum wavelength ? |
|
Answer» `n_4 to n_1` |
|
| 41. |
The solubility product for silver choride is 1.2xx10^(-10) at 298 K. Calculate the solubility of silver chloride at 298 K. |
|
Answer» Solution :Silver chloride dissociates according to theequation : `AgCl(s) hArr AgCl(aq) hArr AG^(+) (aq) + CL^(-)(aq)` LET s be the solubility of AgCl in moles per litre. Consequently, the molar concentration of `Ag^(+) and Cl^(-)` will also be s each. Substituting in the expression for solubilityproduct of AgCl, `K_(sp)=[Ag^(+)][Cl^(-)]=s xx s =s^(2)` But `K_(sp)= 1.2xx10^(-10) ` (Given) `:. s^(2)=1.2xx10^(-10) or s = sqrt(1.2xx10^(-19))=1.1xx10^(-5) "mol " L^(-1)` |
|
| 42. |
Which of the following oxidation states are the most characteristic for lead and tin. respectively ? |
| Answer» ANSWER :A | |
| 43. |
Which of the following species are not known ? |
|
Answer» `[SiCl_(6)]^(2-)` |
|
| 44. |
Write the favourable factors for the formation of ionic bond . |
|
Answer» Solution :(i) Low ionization enthalpy of the metal atom. (II)HIGH electron GAIN enthalpy of the non-metal atom. (iii) High lattic enthalpy of the COMPOUND formed . |
|
| 45. |
What would have happened to the gas if the molecular collisions were not elastic ? |
| Answer» Solution :On every COLLISION, there would have been loss of energy. As a RESULT, the molecules would have slowed down and ultimately settle down in the VESSEL. Moreover, the pressure would have gradually reduced to ZERO. | |
| 46. |
Which of the following statement is/are true 1) PH_5 and BiCl_5 donot exist 2) p - d bond is present in SO_2 3) I_3^(+) has geometry 4) SeF_4 and CH_4 have same shape |
|
Answer» 1,2,3 |
|
| 47. |
Which one is known as oleum ? |
|
Answer» `H_2SO_5` |
|
| 48. |
What happens when CH_3Cl is heated at 570K in presence of Cu powder and Si ? |
| Answer» | |
| 49. |
Write redox reaction when zinc rod is kept in H_(2)SO_(4) solution. |
| Answer» SOLUTION :`Zn_((s))^(+)+2H_((aq))^(+)toZN_((aq))^(+2)+H_(2(G))` | |



