Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write equation of Dipole moment. |
|
Answer» SOLUTION :Dipole moment = CHARGE `xx` distance R = Q (coulomb `xx` r (METER) `thereforer = Q xx r " Cm" `here, Cm = coulomb meter `therefore r = Q xx r "Cm " `here, Cm = coulomb meter |
|
| 2. |
what is the nature of the reaction for the following ? DeltaGgt0 |
| Answer» Solution :`DELTAGGT0` : The process in NON -spontaneous and non - FEASIBLE | |
| 3. |
Which alkali metal emits longest wavelength light in flame test ? |
|
Answer» Na |
|
| 4. |
Total number of stereoisomeric products obtained by addition of bromine to 2-pentene. |
|
Answer» |
|
| 5. |
Which of the following is used as a moderator in nuclear reactors ? |
|
Answer» HARD water |
|
| 6. |
The vant Hoff factor (i) for a dilute solution of potassium aluminium sulphate is |
|
Answer» 4 No. of IONS PRODUCED `=4 THEREFORE i=4.0` |
|
| 7. |
Two elements X and Y have atomic weight of 14 and 16 . They form a series of compounds , A , B , C with fixed mass of X , Y is present in the ratio 1 :2 : 3 : 4 : 5 . If the compound A has 28 parts by weight of X and 16 parts by weight of Y , then the compound C will have 28 parts by weight of X and |
|
Answer» 32 parts by weight of Y |
|
| 8. |
Which of the following activates the benzene ring most towards electrophilic substitution ? |
|
Answer» `-CHO` |
|
| 9. |
Which pair of isomers given below are positional isomers |
|
Answer» Propanal and propanone 
|
|
| 10. |
Water and hydrogen peroxide illustrate the law of |
|
Answer» A. CONSTANT PROPORTIONS |
|
| 11. |
What is the product obtained when chlorine reacts with ethyl alcohol in the presence of NaoH? |
|
Answer» `CH_(3)Cl` `UNDERSET("Ethanol")(CH_(3)CH_(2)OH)+Cl_(2)overset("Oxidation")rarrunderset("Acetaldehyde")(CH_(3)CHO+2HCl)` `CH_(3)CHO+3Cl_(2)overset("CHLORINATION")rarr underset("CHLORAL")(C Cl_(3)CHO+3HCl)` `C Cl_(3)CHO+NaOH overset("Hydrolysis")rarr underset("Chloroform")(CHCl_(3))+underset("Sod. formate".)(HCOONA)` |
|
| 12. |
What are representative elements? Give their valence shell configuration |
|
Answer» <P> |
|
| 13. |
What will be the pressure of the gaseous mixture when 0.5 L of H_(2) at 0.8 bar and 2.0 L of dioxygen at 0.7 bar are introduced in a 1L vessel at 27^(@)C? |
|
Answer» SOLUTION :SUPPOSE the partial pressures of `H_2 and O_2` in the GAS mixture are `p_1 and P_2` respectively. For `H_2 " gas : " P_1 V_1 = P_2 V_2` `0.8 xx 0.5 = P_1 xx 1 or P_1 = (0.8 xx 0.5)/1 = 0.4` bar For `O_2 " gas : " 0.7 xx 2.0 = P_2 xx 1` or `P_2 = (0.70 xx 2.0)/1 = 1.4` bar Hence, the total pressure of the gas mixture = `0.4 +1.4 = 1.8` bar |
|
| 14. |
Which one is more closerto nucleusin 4s^(2)and 3d^(10)4s^(0)and 3d^(10) |
| Answer» SOLUTION :`3D^(10)`and `4S^(0)` is morecloseto nucleusthen `4s^(2)` and`3d^(10)` | |
| 15. |
What is DDT? How is it prepared? |
|
Answer» Solution :(i) DDT is p, p.-dichloro-diphenyl-trichloroethane (ii) DDT can be prepared by HEATING a mixture of chloro BENZENE with CHLORAL in the presence of CONC. `H_2SO_4` 
|
|
| 16. |
What is the colour change at the end point in the titration of oxalate ions against MnO_4^- |
| Answer» SOLUTION :Colourless solutions TURNS PALE PINK. | |
| 17. |
Which one of the following is not a colloidal solution:- |
|
Answer» Smoke |
|
| 18. |
What percentage of solution of H_(2) O_(2)is called as "100 - Volume "H_(2) O_(2) |
|
Answer» 0.15 |
|
| 19. |
Using MO theory, explain why cyclobutadiene is antiaromatic. Strategy: Use the polygon-and-circle method for deriving the relative energies pi MOs. |
Answer» Solution : The MO ELECTRON DISTRIBUTION is Since the nonbonding `MOs` are half-filled, CYCLOBUTADIENE isantiaromatic. |
|
| 20. |
Which of the following is not the use to calcium ? |
|
Answer» Used in chewing gum |
|
| 21. |
What is the symbol for SI unit for electric current ? |
|
Answer» s |
|
| 22. |
Which of the following alkyl groups is more powerful electron donating group when they are attached to unsaturated carbon |
|
Answer» `-CH(CH_(3))_(2)` |
|
| 23. |
Which diseases cause by SO_2 ? |
|
Answer» Asthma |
|
| 24. |
Use the following data to calculate Delta_("lattice") H^( Theta ) for NaBr. Delta_("sub") H^( Theta ) for sodium metal = 108.4 "kJ mol"^(-1), ionization enthalpy of sodium =496 "kJ mol"^(-1), electron gain enthalpy of bromine = - 325 "kJ mol"^(-1), bond dissociation enthalpy of bromine = 192 "kJ mol"^(-1) Delta_(f) H^( Theta )for NaBr_((s)) = - 360.1 "kJ mol"^(-1). |
|
Answer» SOLUTION :Given that, `Delta_("sub") H^( THETA )` for `Na` metal `= 108.4 "kJ mol"^(-1)" IE"` of `Na= 496 "kJ mol"^(-1) , Delta_("eg") H^( Theta )` of `BR = - 325 "kJ mol"^(-1), Delta_("DISS") H^( Theta )` of `Br = 192 "kJ mol"^(-1) , Delta H^( Theta )` for `NaBr = - 360.1 "kJ mol"^(-1)` Born-Haber cycle for the formation of NaBr is as  By applying Hess.s law, `Delta_(f) H^( Theta )= Delta_("sub") H^( Theta ) + "IE" +Delta_("diss") H^( Theta ) + Delta_("eg") H^( Theta ) + U - 360.1 = 108.4 + 496 + 96 + (-325) - U` `U= + 735.5 "kJ mol"^(-1)` |
|
| 25. |
What do you understand by bond pairs and lone pairs of electrons ? Illustrate by giving one example of each type . |
|
Answer» Solution :Covalent BONDS are formed by mutual sharing of elecrons between the two atoms . The shared pairs of electrons THUS present between the BONDED atoms are called bond pairs. All the electrons of an atom MAY not participate in the bonding . The electron pairs present on the atoms which do not take in bonding are called lone paris of electrons . For example ,in` CH_(4)(H:overset(H)overset(. .)underset(. .)C:H)`, there are only 4 bond pairs in ` H_(2) OCH_(4)(H:overset(. .)underset(. .)C:H)` , there are two bond pairs and two lone pairs . |
|
| 26. |
The types of hybridiration on the five carbon atom from right to left in the , 2,3 pentadiene. |
|
Answer» `SP^(3),sp^(2),sp,sp^(2),sp^(3)` |
|
| 27. |
What is meant by positive and negative deviations from Raoult's law and how is the sign of Delta H _(mix) related to positive and negative deviations from Raoult's law? |
|
Answer» Solution :NEGATIVE deviations: In these type of deviations, the partial vapour pressure of each component A and B of solution is HIGHER than the vapour pressure calculated from Raoult.s law. For example: Water and ethanol, chloroform and water. POSITIVE deviations: In case of positive deviation A-B interactions are weaker than those between A-A or B-B. This means that in such solutions molecules or A (or B) will find it EASIER to positive deviation fromRaoult.s law. |
|
| 28. |
Which of the following can adsorb largest volume of hydrogen gas ? |
|
Answer» Finely DIVIDED PLATINUM |
|
| 29. |
Total stereoisomer of the compound Cortisone is : |
|
Answer» 32 |
|
| 30. |
Total stereoisomer of the compound Cortisone is : |
|
Answer» 4 |
|
| 31. |
What is the difference between resonance structure of CH_(3)CH_(2)COO^(-) and CH_(3)COOH ? |
| Answer» Solution :In DIFFERENT structure of `CH_(3)CH_(2)COO^(-)` the DISPERSION of NEGATIVE charge and all are negative ion. In resonance structure of `CH_(3) COOH`, the POLARITY is produced by separation of charge. | |
| 33. |
Write resonance structures of CH_(3)COO^(-) and show the movement of electrons by curved arrows. |
Answer» SOLUTION :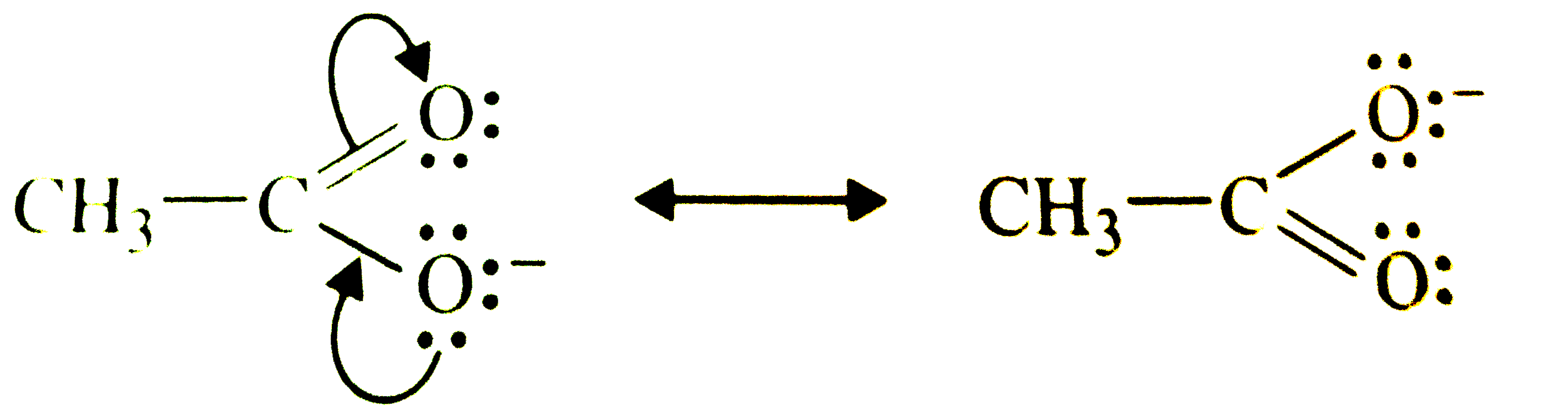 These are RESONANCE STRUCTURES SINCE these differ in the position of electrons and not atoms. |
|
| 34. |
Which of the two is expected to be more stable and why ? O_(2) NCH_(2) CH_(2) overset (bar)O " (or )" CH_(3) CH_(2) overset(bar)O |
|
Answer» SOLUTION :`square O_(2) NCH_(2) CH_(2) overset (bar)O ` is expaected to be more stable than ` CH_(3) -CH_(2) - overset(bar) O ` ` square -NO_(2) ` has -I effect , this leads to the DISPERSAL of NEGATIVE charge. ` square ` On the other hand `-CH_(3)` group has +I effect which intensity the negative charge . ` square therefore ` Dispersalof charge leads to the stability of ion and intensification of negative charge leads to unstailiy of ion. `{:(O_(2)N LARR CH_(2) larr CH_(2) - overset (bar)O ,"More stable "),(CH_(3)rarrCH_(3) rarr overset (bar)O,"Less stable "):}` |
|
| 35. |
Whichof thefollows indicatesdeficiencyofpotassiumin human body ? |
|
Answer» Paralysisof MUSCLES |
|
| 36. |
What happens when (a) Borax is heatedstrongly, (b) Boric acid is added to water, (c ) Aluminium is treated wi th dilute NaOH, (d) BF_(3) is reacted with ammonia ? |
|
Answer» Solution :(a) When borax is heatedstrongly, a transparentglassybead which consis of sodium METABORATE `(NaBO_(2))` and boric anhydride is formed `UNDERSET(" Borax ")(Na_(2)B_(4)O_(7).10H_(2))overset("Heat")rarrNa_(2)B_(4)O_(7) + 10H_(2)O`  (b) Boric act as a weak LEWIS ACID by accepting a hydroxideion of waterand releasing a proton the solution.  (c ) Dihydrogen is evolved. `2Al (s) + 2NaOH(aq) + 6H_(2)O(l) rarr 2Na^(+) [Al(OH)_(4)]^(-) + 3H_(2)(g)` (d) `BF_(3)` beinga Lewisacid accepts a PAIR of electrons from `NH_(3)`to form the corresponding complex. 
|
|
| 37. |
Whichone fo the follownigis not an ortho para director |
| Answer» Answer :a | |
| 38. |
Which is helpful in the formation of ionic bond |
|
Answer» Only SMALL cation |
|
| 39. |
Two moles of an ideal gas is expanded isothermally and reversibly from 1 litre to 10 litre at 300 K. The internal energy change (in kJ) for the process (R = 8.3J) |
|
Answer» 11.4 KJ |
|
| 40. |
Zinc dust in alcoholic solution is a specific reagent in the preparation of alkenes as a |
|
Answer» Dehydrohalogenating AGENT |
|
| 41. |
When ethanol dissolves in water, which of the following is observed? |
|
Answer» Absorption of heat and DECREASE in volume |
|
| 42. |
Which one of the following has lowest ionisation energy- |
|
Answer» `1s^(2)2s^(2)2p^(6)` |
|
| 43. |
Which has the highest melting point ? |
|
Answer» Si |
|
| 44. |
Which of the following is not hydrolysed easily |
|
Answer» `C Cl_(4)` |
|
| 45. |
What is CHN in organic estimation? |
| Answer» Solution :It is a AUTOMATIC EXPERIENTIAL technique in which the ELEMENT, carbon hydrogen and nitrogen present in a compound are DETERMINED by and apparatus known as CHNelements of analysis | |
| 46. |
What is the mass in grams of a hydrogen chloride, HCl? |
|
Answer» Solution :The molecular weight of HCL equal to the atomic weight of H, plus the atomic weight of Cl, (ie) (1.01 + 35.5) amu = 36.5 amu. Therefore 1 mol of HCl contains 36.5 G HCl Mass of HCl MOLECULE `=(36.5 g)/( 6.02 xx 10^(23))` `= 6.06 xx 10^(-23)g` |
|
| 47. |
Write the structure of an anion which is isostructural with BF_(3)and the structure of a cation which is Isostural with CH_(4) |
| Answer» SOLUTION :(i)` NO_(3)^(-)` (Triangular PLANAR) (ii) ` NH_(4) ^(+)` (terachedral ) | |
| 48. |
The wavelength of a blue light is 4800Å. Calculate the frequency and wave number of this light. |
|
Answer» Solution :Given that `c = 3 xx 10^(8)ms^(-1)` `lambda=4800Å=4800xx10^(-10)m=4.8xx10^(-7)m` `"V = frequency = ? "` `"Using the RELATION"upsilon=(c )/(lambda)=(3xx10^(8))/(4.8xx10^(-7)m)` `=6.25xx10^(14)s^(-1) or 6.25xx10^(14)Hz` `"The wave number is the reciprocal ofwavelength "=bar(upsilon)=(1)/(lambda)=(1)/(4.8xx10^(-7)m)=2.0833xx10^(6)m^(-1)` `"Thus frequency of blue light is "6.25 xx 10^(14) Hz` `"Waveumber of blue light is "2.0833 xx 10^(6) ml` |
|
| 49. |
Which of the following statements is true for a catalyst |
|
Answer» It increases the ENERGY of the reactant |
|
| 50. |
Which of the following is an intensive property : Surface tension, mass, volume, enthalpy, density? |
| Answer» SOLUTION :SURFACE tension and density are INTENSIVE PROPERTIES. | |