Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Wite the resonance strutures for NO_(2) and NO_(3)^(-). |
Answer» SOLUTION :
|
|
| 2. |
Three elements, X,Y and Z belong to the same period. Their oxides are acidic, amphoteric and basic respectively. The order of these elements in the periodic table is |
| Answer» Answer :D | |
| 3. |
Which of the following reagents cannot be used for differentiation between CH_(3)CHO and CH_(3)-underset(O)underset(||)(C)-Ph? |
|
Answer» NaOI |
|
| 4. |
Write the combustion of n-hexane with equation . |
|
Answer» Solution :COMBUSTION of n-hexane: `2C_(6)H_(14)+19O_(2)to12CO_(2)+14H_(2)O` |
|
| 5. |
What happens when a ferromgnetic or anti - ferromagnetic or a ferrimagnetic solid is heated ? |
| Answer» SOLUTION :It CHANGES INOT PARAMAGNETIC at some TEMPERATURE | |
| 6. |
What is anhydrone? |
| Answer» Solution :Magnesium PERCHLORATE, `MG( ClO_4)_2`is known as ANHYDRONE. | |
| 7. |
Which one of the following pairs of atoms/ ions have identical ground state configurations? |
|
Answer» `Li^(+) and He^(+)` |
|
| 8. |
What is green fuel ? |
| Answer» SOLUTION :FUEL obtained from PLASTIC waste has high octane rating. It CONTAINS no lead and is known as "GREEN fuel". | |
| 9. |
What is valency ? Explain. |
|
Answer» Solution :Valency is CHARACTERISTIC properties of the elements can be understood in terms of their electronic configurations. The valence of representative elements is usually EQUAL to the NUMBER of ELECTRONS in the outermost orbitals or equal to eight minus the number of outermost electrons as SHOWN below. 
|
|
| 10. |
When benzoic acid is treated with LiAlH_(4), it forms |
|
Answer» Banzaldehyde |
|
| 11. |
The relative rates of mononitration of C_6H_5 - Z , " where " Z = CH_3 - , - NO, - OH , Clare |
|
Answer» `CH_3 GT OH gt NO_2 gt CL` |
|
| 12. |
The translational kinetic energy of 10^(20) molecules of nitrogen at a certain temperature is 0.629J. What is the temperature in .^(@)C? |
|
Answer» `43.3^(@)C` |
|
| 13. |
Write the Lewis dot symbol for Si and P. |
|
Answer» Solution :The NUMBER of valence electons in Si is 4. LEWIS dot structure is `.UNDERSET(.)overset(.)(Si).` The number of electrons in the valence SHELL of P is 5. The Lewis dot structure of P is `(.)underset(.)overset(.)(P).` |
|
| 14. |
List the uses of sodium hydroxide. |
|
Answer» Solution :SODIUM hydroxide is used as LABORATORY reagent. (ii) It is used in the purification of bauxite and petroleum refining. (III) It is used in the textile industries for mercerising cotton FABRICS. (iv) It is used in the manufacture of soap, paper, artificial silk and a number of CHEMICALS |
|
| 15. |
Thevapour pressureof waterat 300Kin aclosedcontaineris 0.4 atm. Ifthe volumeofthe containeris doubled , itsvapourpressureat300 Kwill be _____. |
|
Answer» 0.8 ATM |
|
| 16. |
What effect does branching of an alkane chain has on its boiling point ? |
| Answer» Solution :In ALKANE as the CHAIN INCREASES, boiling POINT decreases. | |
| 18. |
What is the correct order of decreasing stability of the following cations? underset((I))(CH_(3)- overset(oplus)(C )H-CH_(3)) " "underset((II))(CH_(3) - overset(oplus)(C )H-OCH_(3)) " "underset((III))(CH_(3) - overset(oplus)(C )H-CH_(2)-OCH_(3)) |
|
Answer» `II GT I gt III`  Thus jthe stability of carbocation DECREASES in the ORDER: `(II) gt (I) gt (III)` |
|
| 20. |
What is reduction reaction ? Give its examples. |
|
Answer» SOLUTION :.Removal of oxygen/electronegative element from a substance or addition of hydrogen/ electropositive element to a substance is known as rduction." `2HgO_((s))to2Hg_((l))+O_(2(g))` Oxygen removed from MERCURIC oxide. `2FeCl_(3(aq))+H_(2(g))to2FeCl_(2(aq))+2HCl_((aq))` Chlorine remove from ferric chloride. `CH_(2)=CH_(2(g))+H_(2(g))toCH_(3)*CH_(3(g))` (Addition of hydrogen in ethene) `2HgCl_(2(aq))+SnCl_(2(aq))toHg_(2)Cl_(2(s))+SnCl_(4(aq))` (Addition of Hg in `HgCl_(2)`) |
|
| 21. |
Zinc can be coated on ironto produce galvanized iron but thereverse is not posible it is because |
|
Answer» zincis lighter than iron |
|
| 22. |
What is syn gas? How it is prepared? |
|
Answer» SOLUTION :The MIXTURE of CO and `H_2` GASES are called syn GAS. PREPARATION: `C+H_(2)O rarr CO +H_2` |
|
| 23. |
Which of the following compounds(s) is/are paramagnetic |
|
Answer» `KO_(2)` |
|
| 24. |
Under which of the following two condition applied together, a gas devites most from the ideal behaviour ? |
|
Answer» LOW pressure |
|
| 25. |
When maximum amount of solute is dissolved in a solvent at a given temperature, the solution is called……………… |
| Answer» SOLUTION :SATURATED solution | |
| 26. |
Which of the following molecules is polar and possesses zero dipole moment ? |
|
Answer» `Cl_(2)`  The shape is triangular planar , F - B - B = `120^(@), MU =0` D In `BF_(3)` molecule, there are three polar B - F bo nds PRESENT because the electronegativity of F(4 .0) is more then B(2.0) but hybridization of B in `BF_(3) ` is `sp^(2) ` so the shape is triangular planer. B - F bond is planar but it has regular shape . All vector component each other so it is NON polar. |
|
| 27. |
Which of the following has common formula C_(n)H_(2n) ? |
|
Answer» Alkane |
|
| 28. |
What is bond energy? Why is it called enthalpy of atomisation ? |
| Answer» Solution :Bond ENERGY is the AMOUNT of energy requiredto dissociate one moleof bonds PRESENT between the atoms in the gaesous MOLECULES. As the molecules dissociate completely into atoms in the gaseousstate, therefore, bond energy of a diatomic molecule is called enthalpy of ATOMISATION. | |
| 29. |
Write the conjugate acids for the following Bronsted bases : NH_(2)^(-), NH_(3) and HCO O^(-) |
|
Answer» SOLUTION :Conjugate ACID = Bronsted BASE + `H^(+)` `:.` Conjugate ACIDS of `NH_(2)^(-), NH_(3) and HCO O^(-) ` will be `NH_(3), NH_(4)^(+)` and HCOOH respectively. |
|
| 30. |
When the absolute temperature of a gas is doubled then the correct statements are a) The V of a gas increases by 4 times at constant P b) The P of a gas increases by 2 times at constant V c) The V of a gas increases by 2 times at constant P d) The P of a gas increases by 4 times at constant V |
|
Answer» b,d |
|
| 31. |
Which of the following carbocation is most stable ? (a) (CH_(3))_(3)C overset(+)(C)H_(2) (b) (CH_(3))_(3)overset(+)(C)(c) CH_(3)CH_(2)overset(+)(C)H_(2)(d) CH_(3)overset(+)(C)HCH_(2)CH_(3) |
|
Answer» Solution :The order of STABILITY of carbocation is : `3^(@) gt 2^(@)gt 1^(@)` (a) `UNDERSET(1^(@) " Carbocation")((CH_(3))_(3)C - overset(+)(C)H_(2))` (b) `underset(3^(@) "Carbocation")((CH_(3))_(3)overset(+)(C))` (c) `underset(1^(@) "Carbocation")(CH_(3)CH_(2)overset(+)(C)H_(2))`(d) `underset(2^(@) "Carbocation")(CH_(3)overset(+)(C)HCH_(2)CH_(3))` Since `3^(@)` carbocations are the most stable, therefore, option (b) is CORRECT. |
|
| 32. |
Which sysyematic diagram reprsents the correct sequence of Solvay process |
|
Answer»
`NH_(4)OH+_CO_(2)rarrMH_(4)HCO_(3)` `2NaAHCO_(3)UNDERSET(triangle)rarNa_(2)CO_(3)+H_(2)O+CO_(2)` |
|
| 33. |
Which one is a wrong statement? |
|
Answer» the uncertainty principle is `DeltaE xxDeltat ge(h)/(pi)` |
|
| 34. |
True statement about this compound cornane is/are : |
|
Answer» It is having `C_(3)` axis of symmetry 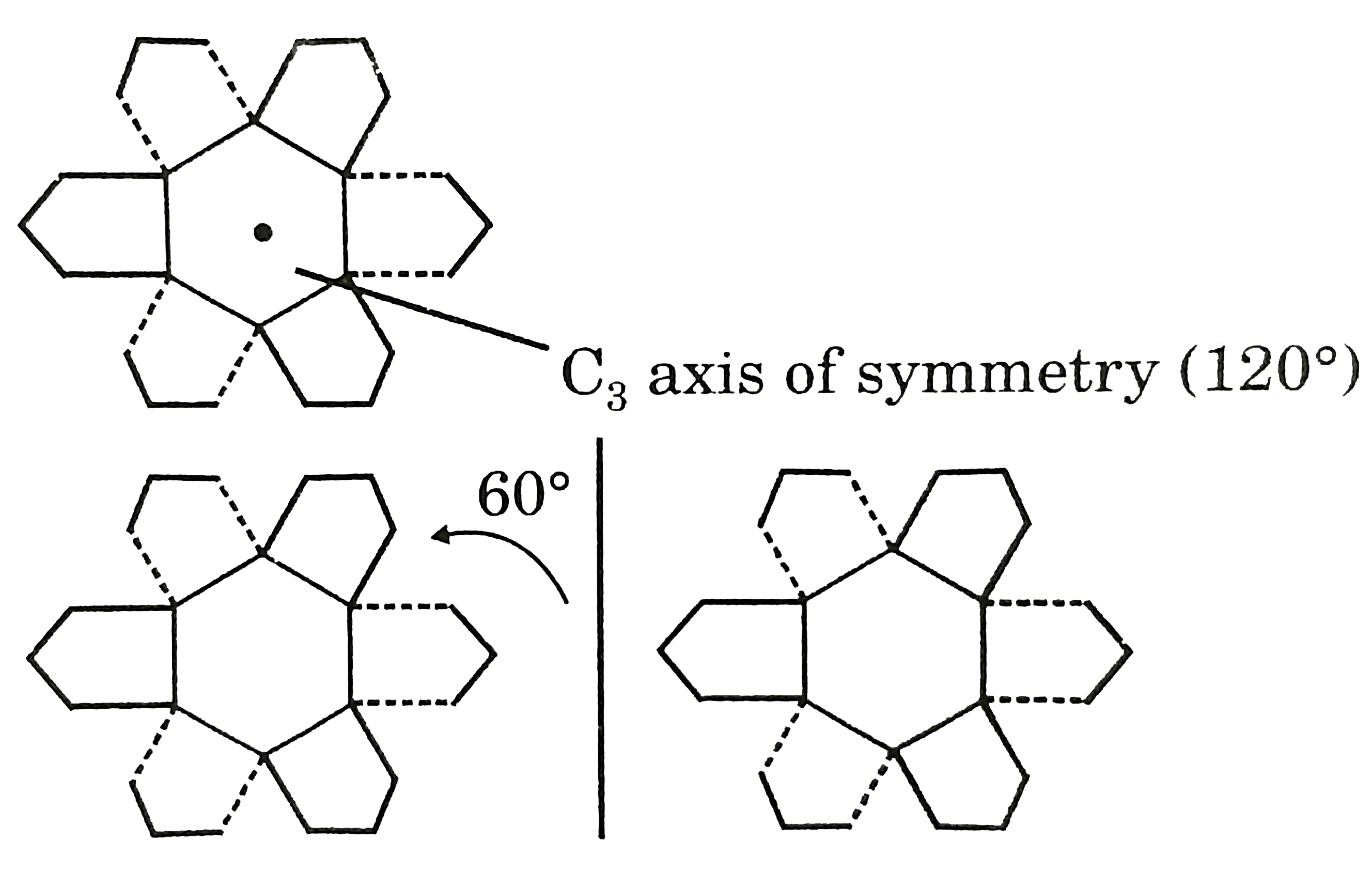 Rotate by `60^(@)` forms MIRROR IMAGE `(S_(6)).` |
|
| 35. |
Which of the following contains the least number of molecules ? |
|
Answer» 0.5 G atom of Zn |
|
| 36. |
The reagent used to convert Ethanoic acid to Ethanol is |
|
Answer» `LiAlH_(4)` |
|
| 37. |
Which of the following is Dobereiner triad |
| Answer» Answer :A | |
| 38. |
Which of the folowing statements are correct |
|
Answer» `H_(2)O_(2)` reduces `MnO_(4)^(-)` both in ACIDIC and basic media |
|
| 39. |
Using the standard electrode potentials given is the Table 1 predict if the reaction between the following is feasible : (a) Fe^(3+)(aq) and I^(-) (aq) (b) Ag^(+) (aq) and Cu(s) (c) Fe^(3+) (aq) and Cu(s) (d) Ag(s) and Fe^(3+)(aq) (e) Br_2(aq) and Fe^(2+) (aq) . |
|
Answer» Solution :(a) It is CLEAR from the table that electrode potential `Fe^(3+)|Fe` (0.77V) is more than that of `I_2|I^(-) (0.54V)`, therefore , `Fe^(3+)` will be READILY reduced and the following reaction is feasible. `Fe^(3+)(AQ) +I^(-) (aq) rarr Fe^(2+) (aq) +1/2I_2(s)` (b) The electrod potential of `AG^(+) |Ag` (0.80V) is more than that of `Cu^(2+)|Cu` (0.34V ) and therefore , `Ag^(+)`will be reduced by COPPER . The following reaction is feasible. `2Ag^(+) (aq) +Cu(s) rarr 2Ag(s) +Cu^(2+) (aq)` (c) The electrode potential of `Fe^(3+)|Fe^(2+)` (0.77V) is more than that of `Cu^(2+)|Cu` (0.34V), therefore , `Fe^(3+)` can be reduced . The following reaction is feasible . `2Fe^(3+)(aq) +Cu(s) rarr 2Fe^(2+)(aq) +Cu^(2+) (aq)` (d) The electrode potential of `Ag|Ag^(+)` (0.80V) is more than that of `Fe^(3+)|Fe` (0.77V) and therefore`Ag^+` will not be reduced by `Fe^(3+)` . Therefore , the reaction will not be feasible. `Ag(s) +Fe^(3+)(aq) rarr Ag^(+) (aq) +Fe^(2+) (aq)` (e) The electrode potential of `Br | Br^(-)` (1.09V) is more than that of `Fe^(3+)|Fe^(2+)` (0.77V) and therefore , Br will be able to reduce by `Fe^(3+)` . Therefore , the following reaction is feasible. `1/2 Br_2(aq) +Fe^(2+)(aq)rarrBr^(-) (aq) +Fe^(3+)(aq)` |
|
| 40. |
Which is the correct order of atomic sizes (At. No: Ce=58, Sn=50, Yb=70 and Lu=71) |
|
Answer» Ce GT Sn gt YB gt LU |
|
| 41. |
What is the magnetic character of the anion of KO_(2) ? |
| Answer» Solution : Anion of `KO_(2) = O_(2)^(-)`(superoxide ion) which has one UNPAIRED ELECTRON and HENCE in PARAMAGNETIC. | |
| 42. |
When salicylic acid is heated with Zn dust. What is the main product? |
|
Answer» Benzene 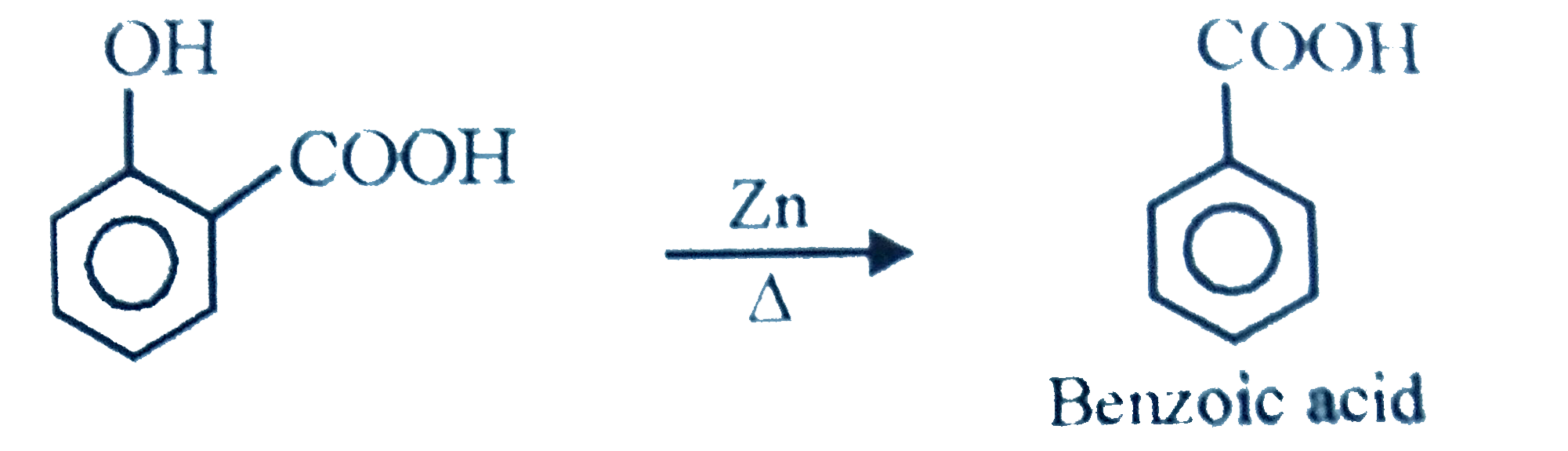
|
|
| 43. |
Which requires catalyst |
|
Answer» `S+O_(2)toSO_(2)` |
|
| 44. |
Which statement( s) is arefalse for theperiodicclassificationof element ? |
|
Answer» Theproperties of the element are theperioicfunctions of thetheiratomic number . inNi(Z= 28 ) , it is `d^(8)` , in Cu (Z=29)it is`d^(10)` andin Zn (Z= 30)it isalso `d^(10)`. Thusd- electrons are notfilledmonotonically.Thusoption(d ) iswrong . Option( c) isalsowrongbecause`Delta_(i)H_(1)` OFTHE elementsin aperiodvaryregularly . |
|
| 45. |
When sodium and chlorine react energy is |
|
Answer» Released and IONIC BOND is formed |
|
| 46. |
Which of the followingstatements are correct regarding hydrogen? (i) The largest single use of dihydrogen is in the synthesis of NH_(3) which is used in the manufacture of HNO_(3) and nitrogenous fertilizers. (ii) It is used to reduce heavy metal oxide (iii) It is used as rocket fuel. (iv) Atomic hydrogen and oxy hydrogen torches find use for cutting and welded to purpose. |
| Answer» Answer :D | |
| 47. |
Which one is most reactive towards S_(N) 1 reaction ? |
|
Answer» `C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br` `C_(6)H_(5) overset(+)CH_(2) lt C_(6)H_(5)overset(+)CHCH_(3) lt C_(6)H_(5)overset(+)CHC_(6)H_(5) lt C_(6)H_(6)overset(+)C(CH_(3))C_(6)H_(5)` Since (a) forms the most stable carbocation, it is most reactive. `underset("(Most reactive)")(C_(6)H_(5)-underset(CH_(3))underset(|)overset(C_(6)H_(5))overset(|)C-Br) overset(-Br^(-))to underset("(Most stable carbocation)")(C_(6)H_(5)-underset(CH_(3))underset(|)overset(C_(6)H_(5))overset(|)(C^(ox)))` |
|
| 49. |
Which of the following mixtures can act as buffer? |
|
Answer» `H_2CO_3+ NaOH (1.5 : 1` MOLAR ratio) guffer is FORMED (b)` {:( H_2CO_3 + , NaOH to ,NaHCO_3+H_2) , ( 1.5M , 2M, 0),( -, 1.5M , 1.5 M),( , 0.5 M , ):}` ` {:( NaHCO_3+ , NaOH to , Na_2CO_3+H_2O ),( 1.5 M, 0.5 M , 0 ),( 0.5 M ,- , 0.5 M),( 1.0 M,, ) :}` buffer is formed ( C) ` {:( NH_4 OH +, HCl to, NH_4Cl + H_2O ),( 5M, 4M, 0),(4M, -, 4M),( 1M,, ):}` (d)ACID is excess |
|
| 50. |
what is 'Z' above reaction |
|
Answer» Chloral |
|



