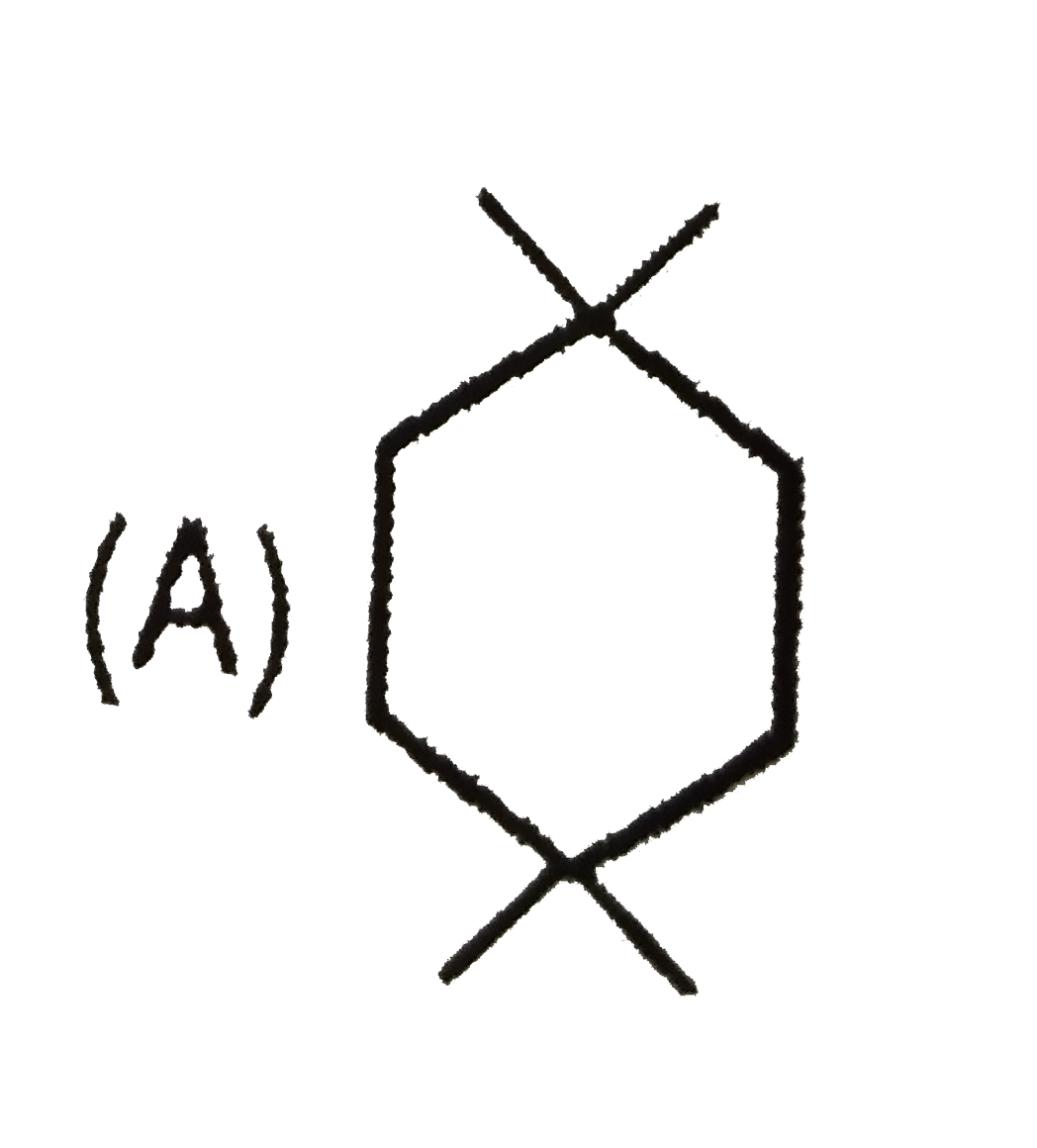Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 3. |
Which of the following compounds will produce the most stable carbonium ion when heterolytic cleavage of C-O bond take place ? |
|
Answer» `CH_(3)CH-UNDERSET(underset(CH_(3))(|))(CH_(2))-OH` |
|
| 4. |
The volume of H_(2)STP required to completely reduce 160 gms of Fe_(2)O_(3) is |
|
Answer» `3 xx 22.4L` |
|
| 5. |
The violent reaction between sodium and water is an example of "_________________". |
|
Answer» reduction |
|
| 6. |
Which of the following statements about hydrogen 1 is incorrect ? |
|
Answer» HYDROGEN ion, `H_(3)O^(+)` exists FREELY in solution. |
|
| 7. |
Which of the following pairs has almost same radii ? |
|
Answer» `Al, Ga` |
|
| 8. |
Which type of Na and K metal salts are more present in ash of plant ? |
|
Answer» `PO_(4)^(-3)` |
|
| 9. |
What doyou understand byexothermicreaction andendothermicreaction? Giveone exampleofeach type. |
|
Answer» Solution :Exothermicreactions . Reactions WHICHARE accompaniedby evolution ofheatare calledexothermic reactions. The quantityof heatproducedis showneitheralongwith theproducts WITHA plussignor intermsof `DeltaH`with anegativesign. forexample. `C ( s)+ O_(2)( g) toCO_(2)( g) +393. 5 kJ` ` H_(2) (g) + (1)/(2) O_(2) (g)toH_(2) O(l) , Delta H=-285.8 kJ MOL^(-1)` Endothermicreactions. Reactions whichproceed withabsorption ofheatare calledendothermicreactions . the quantityof heatabsorbed is shown eitheralong withthe PRODUCTS witha negativesign orintermsof `DeltaH`with apositivesign. Forexample ` C ( s)+ H_(2) O( g) toCO (g)+ H_(2)( g) -131 .4 kJ` `N_(2)( g) +3 H_(2)( g) to2 NH_(3)( g) Delta H=- 92 kJ mol^(-1)` |
|
| 10. |
Which of the following is a free radical? |
|
Answer» `NO_(2)^(-)` |
|
| 11. |
The standard molar enthalpies of formation of cyclohexane(l ) and benzen(l) at25^(@) are - 156 and + 49 kJ mol^(-1) respectively. The standard enthalpy of hydrogenationof cyclohexene( l) at 25^(@)C is-119 kJ mol^(-1) . Use these data to estimate the magnitudeof the resonanceenergy of benzene. |
Answer» Solution :Standard enthalpyof hydrogenation of cyclohexene`= -119 KJ mol^(-1)`. This is the observedenthalpy of hydrogenation ofone double bond ( PRESENT in cyclohexene) . If benezene is considered as cyclohexatriene, the observed ENTHALPY of the reaction.  wouldbe `= 3 xx(-119)=- 357 kJ mol^(-1)` wouldbe `= 3 xx(-119)=- 357 kJ mol^(-1)` Calculated enthalpy of the above reaction is `Delta_(r)H =DeltaH_(f)^(@)(C_(6)H_(14)) - [ DeltaH_(f)^(@) (C_(6)H_(6))+3DeltaH_(f)^(@)(H_(2))]= - 156 - ( 49+0) = - 2205 kJmol^(-1)` `:. `Resonance energy = Observed`Delta_(r)H^(@) -` Calculated`Delta_(r)H^(@)` `= ( - 357 ) - ( - 205) kJ mol^(-1) = -152 kJ mol^(-1)` |
|
| 12. |
Thermal decomposition temperature is more for |
|
Answer» `NaHCO_(3)` |
|
| 15. |
What is eluant? |
| Answer» Solution :ELUANT is mobile PHASE used in CHROMATOGRAPHY in which the compound move SLOWLY over the stationary phase | |
| 16. |
When CO_(2) is passed into Ca(OH)_(2) a white precipitate is formed due to the formation of |
| Answer» Answer :A | |
| 17. |
What is the spin of two electron in He ? |
| Answer» SOLUTION :Both are in OPPOSITEDIRECTION`+(1)/(2) ` and `(1)/(2)` | |
| 18. |
Which of the following is not correct with regard to the structure of diborane? |
|
Answer» FOUR TERMINAL hydrogen atoms and TWO bridged hydrogen atoms |
|
| 19. |
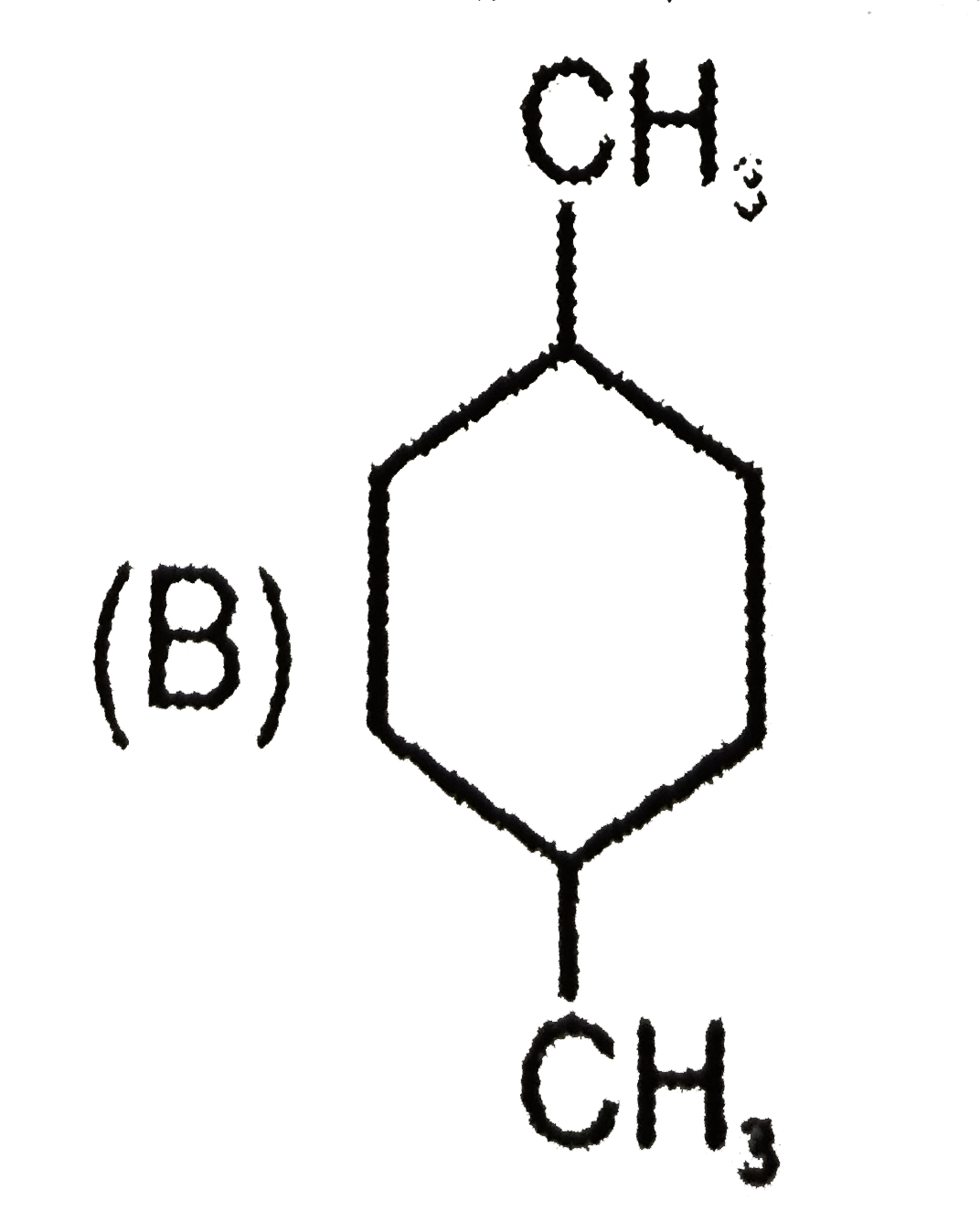
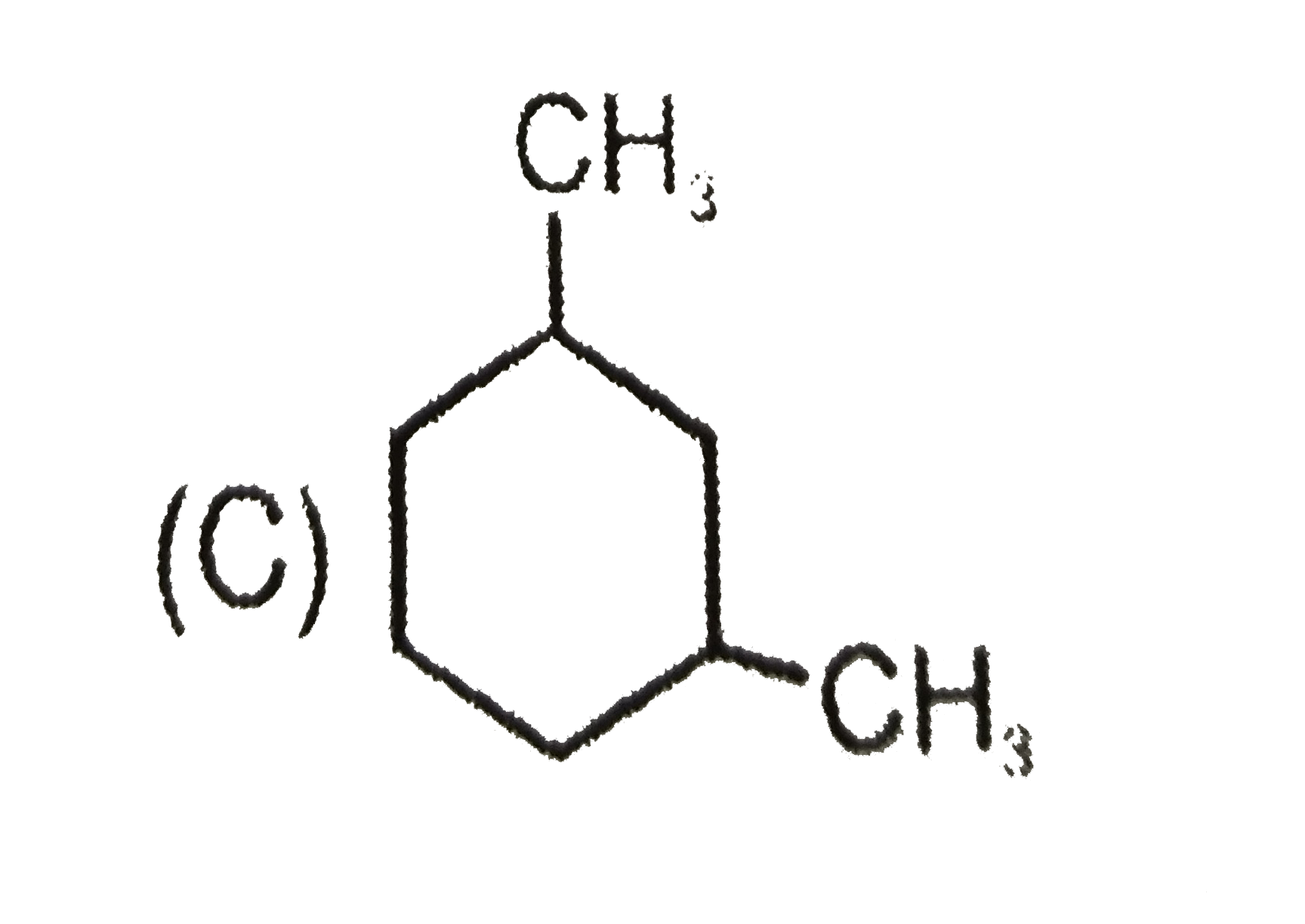
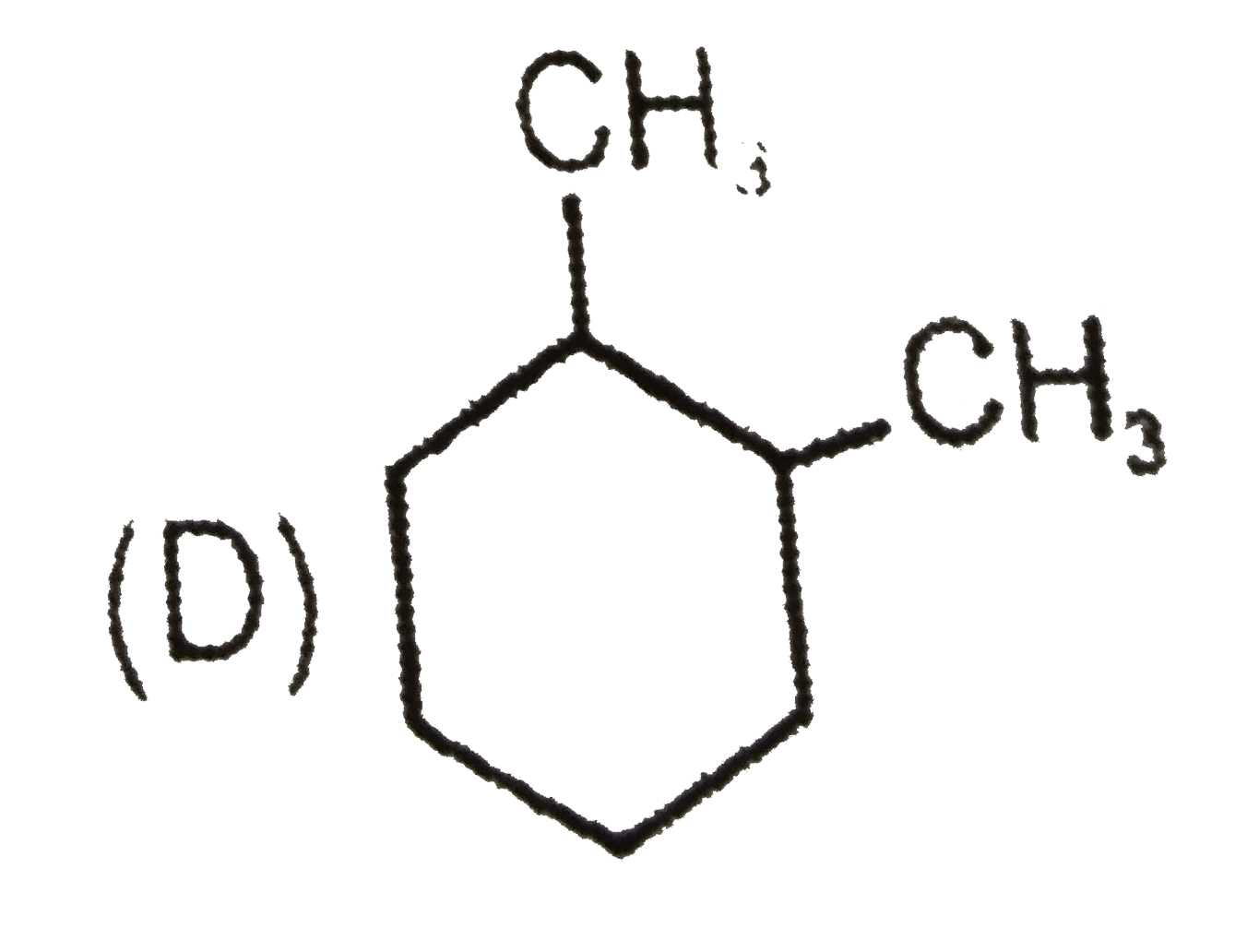
Which of the following compound will give four monochloro (structural) product on monochlorination. |
|
Answer»
|
|
| 20. |
Which of the following is/are primary pollutant,, (A) Ozone(B) SO_(2) ""(C) SO_(3) ""(D) NO_(2) "" PBN |
|
Answer» A,C and E |
|
| 21. |
Which of the following fcc structure contains cation in alternate tetrahedral voids ? |
|
Answer» NaCl |
|
| 22. |
Writeshort notes on corey housereaction |
| Answer» SOLUTION :An alkylhalideand lithiumdialkylcuprateare reactedto givehigheralkanes . THISREACTIONIS KNOWAS Corey -Housereaction. | |
| 23. |
Write the chemical formula of plaster of Paris. |
| Answer» SOLUTION :`(CaSO_4)_2 . H_2O` | |
| 24. |
Write equation of ionization constant of weak base MOH. |
|
Answer» Solution :`K_b=([M^+][OH^-])/"[MOH]"=(c ALPHA^2)/((1-alpha))` Here, `alpha`=dissociation constant of BASE c= initial concentration of base |
|
| 25. |
Zinc oxide is white but it turns yellow on heating. Explain. |
|
Answer» Solution :When ZnO is heated, it loses oxygen according to the reaction :`ZnO to Zn^(2+) + 1/2O_2 + 2e^(-)` `Zn^(2+)` ions are entrapped in the interstitial voids and electrons in the neighbouring interstitial voids to maintain electrical neutrality. This GIVES RISE to metal defect. Due to presence of electron in the interstitial voids (F-centres), the COLOUR is yellow as the REMAINING colours of white light are absorbed by these electrons. |
|
| 26. |
What is acid rain ? |
| Answer» SOLUTION :When the pH of the RAIN water DROPS below 5.6. It is called ACID rain. | |
| 27. |
Which one is the correct order of ionic radii of following ions ? |
|
Answer» <P>`Ti^(4+) LT Mn^(7+)` |
|
| 28. |
Which of the following is the most powerful oxidizing agent ? |
|
Answer» `F_(2)` |
|
| 29. |
Which of thefollowingpairscontainmetalloid elementsin the periodictable ? |
|
Answer» NA and K |
|
| 30. |
When metal X is treated with sodium hydroxide, a white precipitate (A) is obtained, which is soluble in excess of NaOH to give soluble complex (B). Compound (A) is soluble in dilute HCl to form compound (C). The compound (A) when heated strongly gives (D), which is used to extract metal. Identify (X), (A), (B), (C) and (D). Write suitable equations to support their identities. |
|
Answer» Solution :The given metal X gives a white precipitate with sodium hydroxide and the precipitate dissolves in excess of sodium hydroxide. Hence, X MUST be aluminium. The white precipitate (compound A) obtained is aluminium hydroxide. The compound B formed when an excess of the base is added is sodium tetrahydroxoaluminate (III). `underset"(X)"(2AL)+3NaOH to underset"(ppt)"(Al(OH)_3) darr + 3Na^(+)` `underset"(A)"(Al(OH)_3) + NaOH to underset"Sodium tetrahydroxo aluminate (III)"(NA^+ [Al(OH)_4]^(-))` Now, when dilute hydrochloric acid is added to aluminium hydroxide, aluminium chloride (compound C) is obtained. `underset"(A)"(Al(OH)_3) + 3HCl to underset"(C)"(AlCl_3) + 3H_2O` Also, when compound A is HEATED strongly, it gives compound D. This compound is used to extract metal X. Aluminium metal is extracted from alumina. Hence, compound D must be alumina. `underset"(A)"(2Al(OH)_3) oversetDeltato underset"(D)"(Al_2O_3) + 3H_2O` |
|
| 31. |
The solubility of gypsum in water increases of ammonium sulphate due to the formation os |
|
Answer» Mixed SALT |
|
| 33. |
Which of the following factor affects ionisation energy ? SSSon : S (2) s - sub orbit nearest to nucleus. |
|
Answer» Size of the ATOM |
|
| 34. |
Which one of the following statement is not true? |
|
Answer» CLEAN water would have a BOD value of less than 5 ppm |
|
| 35. |
Which of the following is not true in case of reaction with heated copper at 300^(@)C ? |
|
Answer» Phenol `RARR` BENZYL alcohol |
|
| 36. |
Write the common name for the following compounds. (i)CH_(3)OH (ii) C_(2)H_(5)OH (iii) C_(2)H_(5)-O-C_(2)H_(5) (iv) CH_(3)COOH |
|
Answer» Solution :(i) Methyl alcohol (ii) ETHYL alcohol (III) DI ethyl ETHER (iv) Acetic acid |
|
| 37. |
Which of the following not avaible in nature heated |
|
Answer» `BeCO_(3)` |
|
| 38. |
What is the solubility product (K_(sp)) ? |
| Answer» Solution :At definite TEMPERATURE the product or concentration of ions of sparingly soluble salt is called solubility product `K_(SP)`. | |
| 39. |
What is the maximum number of orbitals that can be identified with the following quantum numbers? n = 3,1 = 1, m_(l) = 0 |
|
Answer» 3d |
|
| 41. |
Which of the following on thermal decomposition yields a basic as well as an acidic oxide ? |
|
Answer» `KClO_(3)` |
|
| 42. |
Write the relation between standard free energy change and equilibrium constant K_(p) for a reversible reaction. |
| Answer» SOLUTION :Relationship between standard free energy change `Delta G^(@)` and the EQUILIBRIUM CONSTANT `K_(p)` is `Delta G^(@) = -2.303 RT log K_(p)`, where R is the gas constant `= 8.314 JK^(-1)` and T is the temperature in Kelvin. | |
| 43. |
Which of the following is the most stable carbocation |
|
Answer»
|
|
| 44. |
Which of the following pairs of elements would have a more negative electron gain enthalpy? (i) O or F (ii) F or Cl |
|
Answer» SOLUTION :(i) F (II) Cl `O (-141), F (-328) " and " Cl (-349) kJ "mol"^(-1) Delta_(eg) H`. |
|
| 45. |
Which of the following has zero electron density in xy plane ? |
|
Answer» <P>`d_(z^(2))` |
|
| 46. |
Which product is formed on chlorination of (CH_(3))_(3)CH ? Also give comparison of their composition. |
|
Answer» SOLUTION :`overset(1^(@))(C)H_(3)-underset(""^(1^(@))(CH_(3)))underset(|)overset(1^(@))(CH)-overset(1^(@))(CH_(3))underset(hv)overset(Cl_(2))rarrunderset((A))underset(3^(@)"butylchloride")(CH_(3)-underset(CH_(3))underset(|)overset(Cl)overset(|)(C)-CH_(3))+underset((B))underset(1^(@)-"chloride")underset(CH_(3))underset(|)(CH)-CH_(2)Cl` A and B both are obtained from the monochloroalkane, (A) proportion `gt` product (B) proportion. Because compound (A) formed which DEPEND on production RATE of `3^(@)` hydrogen `gt` (B) formed which depends on `1^(@)` hydrogen atoms. |
|
| 47. |
Which cations can be removed by ion exchange method ? |
|
Answer» Ca and Mg |
|
| 48. |
The type of isomerism not found in alkenes is : |
|
Answer» CHAIN isomerism |
|
| 49. |
What are the applications of Dalton's law of partial pressure? |
|
Answer» SOLUTION :(i) Physicians reports the pressure of the patient.s gases in BLOOD , analyzed by hospital lab, the values are reported as partial pressures. Gas `""` Normal range `P_(CO_(2)) ""` 35-45 mm of Hg `P_(O_(2)) ""` 80-100 mm of Hg (II) When gas is colleted by downward displacement of water , the pressure of dry vapour collected is computed USING Dalton.s law. `P_("dry gas colleted") = P_("Total") = P_("Water vapour")` `P_("water vapour")` = Aqueous tension |
|
| 50. |
When does Frenkel defect arise ? Give reason . |
| Answer» Solution :When some IONS (usually cations) are missing from the lattice sites and they occupy the interstitial sites so that electrical neutrality as WELL as stoichiometry is MAINTAINED , it is called FRENKEL defect. | |